Role of adjuvant therapy in early-stage small-cell lung cancer: comment on a population-based cohort study of patients with early-stage small-cell lung cancer
Small-cell lung cancer (SCLC) is a lethal disease for which there have been only small advances in diagnosis and treatment over the past decade. After a thorough invasive mediastinal stage evaluation, surgical resection is recommended over nonsurgical treatment in patients with clinical stage I SCLC. Although fewer than 5% of patients with SCLC present with stage I disease, the increasing use of computed tomography (CT) and minimally invasive surgery to remove suspicious lung nodules without prior biopsy may increase this percentage in the future. The role of surgery in SCLC has been analyzed using two large population databases. A study of data from 1988 to 2002 in the US SEER database identified 14,179 patients with SCLC, including 863 (6.1%) who underwent surgery (1). Surgery was more commonly performed in patients with T1/T2 disease (P<0.001). Patients who underwent surgery had better survival than those who did not undergo surgery for both localized (median survival, 42 months vs. 15 months, respectively; P<0.001) and regional disease (median survival, 22 months vs. 12 months, respectively; P<0.001). The 5-year overall survival (OS) rates postoperatively were 45% for localized disease and 26% for regional disease. Another comparative study from 1988 to 2004 using the SEER database analyzed patients treated for stage I SCLC (2). Patients who did not undergo surgery (n=51,161) were compared with those who underwent lobectomy (n=5,247, 15.8%), with 15% of patients receiving postoperative irradiation. The routine administration of adjuvant systemic therapy was assumed, but data were unavailable for confirmation. The 5-year OS for patients undergoing lobectomy with and without postoperative irradiation was 57% and 50%, respectively (P=0.9). In contrast, the 5-year OS for patients treated with external beam irradiation alone was 15%. Furthermore, many studies of patients with SCLC who underwent surgical resection reported that approximately 50% of the patients were not suspected of having SCLC prior to resection.
Adjuvant systemic therapy is also recommended for all stages of SCLC following complete resection. However, adjuvant chemotherapy has not been evaluated in prospective randomized trials. Only four prospective studies have evaluated the outcomes of patients receiving adjuvant therapy following surgery for SCLC. Table 1 summarizes these four studies (3-6), which are single-arm, prospective phase II trials, including various chemotherapy regimens, with or without irradiation. These adjuvant treatments were generally well tolerated with acceptable levels of toxicity. Collectively, 5- or 4-year survival rates ranged between 36% and 73%. Pathological stage, including tumor size or lymph node metastasis, clearly correlated with survival outcome and can be used as a powerful prognostic factor. Thus, these reports suggest an advantage of chemotherapy after complete surgical resection, yielding impressive long-term survival for stage I SCLC and subsequently leading to a level 2C recommendation in the third edition of the American College of Chest Physicians (ACCP) Lung Cancer Guidelines and a category 2A recommendation in the current NCCN guidelines.
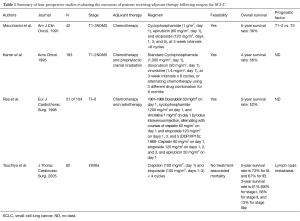
Full table
The current study is the first population-based study to examine the role of adjuvant therapy for patients who previously underwent resection for pathologic stage T1-2N0M0 SCLC. In univariable analysis, adjuvant chemotherapy, with or without radiation, was significantly associated with improved OS compared with no adjuvant therapy (P<0.01, 52.7% and 40.4% 5-year survival rates, respectively). In addition, multivariable Cox modeling demonstrated that adjuvant chemotherapy alone or with PCI were associated with improved survival when compared with no adjuvant therapy. These results provide further support for the use of adjuvant chemotherapy as recommended by the current ACCP and NCCN guidelines.
Interestingly, patients who underwent surgery with adjuvant chemotherapy and presumed PCI had much better OS when compared with other groups. The current NCCN guidelines recommended that in patients with limited-stage SCLC who have a good response to initial therapy, PCI decreases brain metastases and increases OS (Category 1) (7,8). This NCCN recommendation is based on trials that studied the impact of chemotherapy and radiation on patients who did not undergo surgery. Therefore, this is the first study to suggest a possible benefit for prophylactic cranial irradiation (PCI) after surgery and adjuvant chemotherapy, and further prospective, large sample-size studies are thus warranted.
The current study is the first population-based study to examine the role of adjuvant therapy in patients who underwent resection for pathologic stage T1-2N0M0 SCLC. However, there are several limitations to this study, including its retrospective nature and the confounding variables that were not considered during the analysis. Of note, the major limitation of population-based studies is that there is insufficient information on the irradiation settings and chemotherapy regimens; there may also have been selection bias for the particular regimen given. Moreover, there may have been a possible type II error because of the limited sample size. These findings should be further evaluated in prospective phase II or randomized controlled trials. JCOG1205/1206 is an ongoing phase III trial comparing irinotecan and cisplatin with etoposide and cisplatin for adjuvant chemotherapy in cases of completely resected, pulmonary high-grade neuroendocrine carcinoma, including SCLC.
Acknowledgements
Funding: The Grant-in-Aid for Scientific Research, Japan Society for the Promotion of Science (24592104), Ministry of Education, Culture, Sports, Science and Technology, Japan.
Footnote
Provenance: This is an invited Commentary commissioned by the Section Editor Gang Shen, MMSC (The Second Affiliated Hospital Zhejiang University School of Medicine, Hangzhou, China).
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
- Schreiber D, Rineer J, Weedon J, et al. Survival outcomes with the use of surgery in limited-stage small cell lung cancer: should its role be re-evaluated? Cancer 2010;116:1350-7. [Crossref] [PubMed]
- Yu JB, Decker RH, Detterbeck FC, et al. Surveillance epidemiology and end results evaluation of the role of surgery for stage I small cell lung cancer. J Thorac Oncol 2010;5:215-9. [Crossref] [PubMed]
- Macchiarini P, Hardin M, Basolo F, et al. Surgery plus adjuvant chemotherapy for T1-3N0M0 small-cell lung cancer. Rationale for current approach. Am J Clin Oncol 1991;14:218-24. [Crossref] [PubMed]
- Karrer K, Ulsperger E. Surgery for cure followed by chemotherapy in small cell carcinoma of the lung. For the ISC-Lung Cancer Study Group. Acta Oncol 1995;34:899-906. [Crossref] [PubMed]
- Rea F, Callegaro D, Favaretto A, et al. Long term results of surgery and chemotherapy in small cell lung cancer. Eur J Cardiothorac Surg 1998;14:398-402. [Crossref] [PubMed]
- Tsuchiya R, Suzuki K, Ichinose Y, et al. Phase II trial of postoperative adjuvant cisplatin and etoposide in patients with completely resected stage I-IIIa small cell lung cancer: the Japan Clinical Oncology Lung Cancer Study Group Trial (JCOG9101). J Thorac Cardiovasc Surg 2005;129:977-83. [Crossref] [PubMed]
- Aupérin A, Arriagada R, Pignon JP, et al. Prophylactic cranial irradiation for patients with small-cell lung cancer in complete remission. Prophylactic Cranial Irradiation Overview Collaborative Group. N Engl J Med 1999;341:476-84. [Crossref] [PubMed]
- Arriagada R, Le Chevalier T, Rivière A, et al. Patterns of failure after prophylactic cranial irradiation in small-cell lung cancer: analysis of 505 randomized patients. Ann Oncol 2002;13:748-54. [Crossref] [PubMed]


