Management of chest trauma
Management of chest trauma can be divided into three distinct levels of care; pre-hospital trauma life support, in-hospital or emergency room trauma life support and surgical trauma life support. At each level of care recognition of thoracic injury is crucial for the later outcome. Initial resuscitation and management of chest trauma patient is based upon protocols from the Advanced Trauma Life Support (ATLS) (1). After a primary survey immediately life-threatening injuries should be excluded or treated such as:
- Airway obstruction;
- Tension pneumothorax;
- Open pneumothorax;
- Massive haemothorax;
- Flail chest;
- Cardiac tamponade.
Secondary survey will provide information on potentially life-threatening injuries:
- Pulmonary contusion;
- Myocardial contusion;
- Aortic disruption;
- Traumatic diaphragmatic rupture;
- Tracheobronchial disruption;
- Oesophageal disruption.
The German Trauma Registry DGU® included 873 chest trauma patients between 1993 and 2008, of these 60% had an AIS (serious) score ≥3, expressing once again the potential severity of chest trauma. Nevertheless, thoracic surgeons are not part of the initial emergency room trauma team in level I trauma centres in Germany (2). In North America, the thoracic surgeon is present in 1 out of 16 level I trauma centres. This is due to the distribution of injuries, many of which do not require the specific knowledge of a thoracic surgeon in every patient (3). Kulshrestha et al. showed that diagnosis made in 1,359 chest trauma patients was in 49% 1–2 rib fractures, 20% pneumothorax, 12% lung contusion and 6% thoracic vascular injury. Treatment was in 18% a chest tube. Thoracic surgical intervention in the emergency room or operating theatre was necessary in only 2.6%. Mortality was 9.4% of which 56% in the first 24 hours. This paper demonstrates exactly why thoracic surgeons are seldom part of the trauma team in the emergency room. The very specific experience of thoracic surgeons, when available, should part of the extended trauma team. Many deaths can be prevented by prompt diagnosis and treatment coupled with an understanding of pathophysiologic factors associated with thoracic trauma.
In 2016, the German S3 guideline on treatment of polytrauma/severe injuries was revised involving representatives of all disciplines (4). With regards to chest trauma the several points were stressed, according to the recent available literature, for the three levels of trauma care.
Pre-hospital trauma life support
Assessment of breathing and clinical examination of the thorax (respiratory movements and quality of respiration) are necessary to recognize major thoracic injuries such as tension pneumothorax, open pneumothorax, fail chest, pulmonary contusion and massive haemothorax. Inspection, palpation, percussion and especially auscultation [sensitivity 90%, specificity 98% (5)] will provide information as to whether a tension pneumothorax is present. Clinical diagnosis of pneumothorax, may require immediate intervention, by initial needle decompression of the pleura space (6). Should this not be successful or there is evidence of pneumothorax, chest tube drainage is necessary. In the absence of hypoventilation on auscultation, or thoracic pain in a stable patient a major tension pneumothorax can be ruled out. Repeated examination is mandatory to avoid omission of progression of a pneumothorax. As tension pneumothorax is the most frequent reversible cause of death in trauma patients with cardiac arrest (7-9).
Emergency room trauma life support
Repetition of clinical examination in primary survey together with anamnestic information on the mechanism of thoracic trauma will provide information on potential severity of thoracic injury (10). When the extent of trauma cannot be defined it is recommend to perform contrast-enhanced CT scan (11). As the sensitivity of a chest X-ray in the emergency room is only 58.3% (12). Thoracic ultrasound examination is valid when CT scan is not necessary, in comparison with chest X-ray it shows equivalent sensitivity and specificity for diagnosis of pneumothorax (13-15). Ultrasonography in the emergency room is also a reliable method to exclude pleural/pericardial effusion (16,17).
Chest tube drainage is necessary when pneumothorax is relevant, progressive or when the patient is mechanically ventilated (18,19). Large bore chest tubes (Ch. 36–40) compared with smaller chest tube (Ch. 28–32) have no advantage in treatment of severely injured patients (20).
When should the thoracic surgeon definitely be involved?
According to the ATLS guideline this is recommended as follows (1):
- Blood loss over the chest TD >1,500 mL initially or >200 mL/hour over 2–4 hours;
- Haemoptysis;
- Massive subcutaneous emphysema;
- Important air-leakage over the chest tub;
- Uncertain images on the chest X-ray or CT thorax;
- Penetrating chest trauma.
Indications for an immediate thoracic surgical intervention are (1):
- Blood loss ≥1,500 mL initially/>200 mL/hour over 2–4 hours;
- Endobronchial blood loss; massive contusion with significant impairment of mechanical ventilation;
- Tracheobronchial tree injury (air-leakage/hemothorax);
- Injury of the heart or large vessels (blood loss/pericardial tamponade).
Surgical trauma life support
Which incision should be chosen for emergency thoracic surgical intervention? Anterolateral thoracotomy in the 4–6th intercostal space is usually recommended, although in 20% of the patients it is insufficient to visualize all lesions and must therefore modified (21). Clamshell (transverse sternotomy and bilateral anterolateral thoracotomy) or hemi-clamshell (longitudinal sternotomy and anterolateral thoracotomy) will permit better exposition of thoracic organs. The necessity for emergency room thoracotomy is extremely rare, anterolateral thoracotomy will permit a potentially lifesaving measure (clamping of a great vessel) in an extreme situation before proceeding to the operating theatre (3).
The role of minimal invasive surgery in management of chest trauma should not be under- or overestimated (22). The amount of randomized prospective data for video-assisted thoracoscopic surgery (VATS) management in chest-trauma patient is very sparse. Nevertheless the on-going acceptance and use of VATS for major thoracic resections has led to advanced techniques for management of major bleedings in the elective-surgery-patient. VATS as a procedure for pleural space management in the non-critical, non-massive-transfusion patients can be of great assistance (23). Its value in persistent non-major-vessel-bleeding haemothorax in terms of pleural space debridement is undisputable (24). In some cases VATS is considered to be related to lower ARDS-rates in comparison to open thoracotomy patients, whereby an obvious bias for the non-massive-injury-patients exist (25). Yu et al. demonstrated in 2016 a small series of VATS-treated patients with penetrating thoracic trauma (26). In all cases, the patients were haemodynamically stable and no major intrapericardial vessels were found. Even though major lung or intercostal injuries were documented, all patients could be treated successfully through minimal invasive surgery techniques. Jin et al. could prove a significant advantage for stable thoracic trauma patients treated through VATS in a randomized trial vs. open thoracotomy (27). Video-assisted thoracoscopy is also a useful explorative and eventually therapeutic tool for diaphragmatic lesions. Traumatic diaphragmatic lesions are more common than believed and can be misdiagnosed very easily, thus leading to delayed complications (28). A thoracoscopic evaluation of the pleural cavity can demonstrate easily misdiagnosed lesions and treat a possible persisting haemothorax. It is obvious that VATS can play a role in the treatment of chest trauma management so long the inclusion criteria are respected. In a haemodynamic unstable patient with severe chest wall or cardiac vessels injuries and massive transfusion there is no place for thoracoscopic efforts that only delay the unavoidable open approach and perhaps minimize the chances for a positive outcome. But in the stable haemodynamic state with small perforating wounds, VATS can be a valuable weapon of thoracic surgeons for fast recovery, minimized pain and perfect visualization of the entire pleural place. Indications for such an approach are as followed:
Indications for VATS in severely injured patients:
- (Penetrating) injury with little blood loss in a stable patient;
- Persistent hemothorax;
- Empyema;
- Persistent air-leakage;
- Suspicion of diaphragmatic rupture.
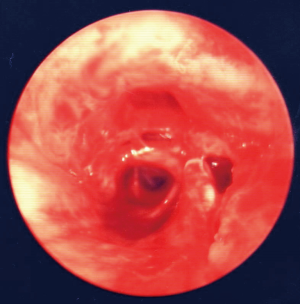
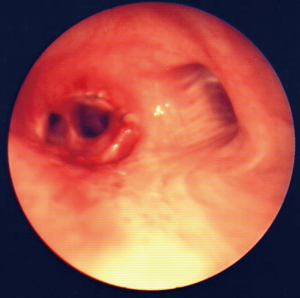
Case report 1: traumatic tracheobronchial injury of the intermediate bronchus (Figures 1-3)
Patient required immediate surgical intervention due to massive subcutaneous emphysema and chest tube air-leakage. Surgical options were bilobectomy or middle lobe sleeve resection without compromising the outcome of the patient. Preserving the middle lobe does have an influence on the lung function after 3 months. This has been documented in lung cancer patients (29). Exactly in such a situation even if seldom the surgical experience, in terms of interpretation of radiologic images, bronchoscopic findings and performing such a complex operation by the thoracic surgeon may be lifesaving.
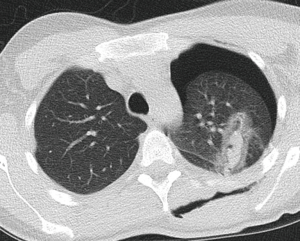
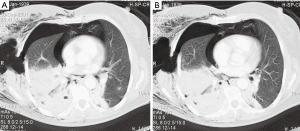
Case report 2: penetrating thoracic injury with retained foreign body (knife) and VATS treatment
A 31-year-old patient was admitted in the emergency room after being perforated in the chest dorsally through a sharp object (Figures 4,5). The patient was haemodynamically stable and CT-thorax was performed. A left-sided haemopneumothorax was diagnosed as well as an injury of the left upper lobe. Immediate surgery exploration was necessary, due to the stability of the patient the thoracic surgeon preferred to perform a VATS procedure. The foreign body was removed under thoracoscopic vision. The relatively small haemothorax was removed. A lesion of the intercostal artery was detected after removal of the foreign body. Ligation with endo-clips was sufficient. Postoperative course was uneventful.
Conclusions
Trauma care is complex. Blunt thoracic trauma is frequent but emergency surgical interventions are rare. Thoracic surgeons are usually not part of the trauma team in most trauma centres. The objective was to review specific details in thoracic trauma care, is there a place for minimal invasive surgery in thoracic trauma care and to demonstrate how the experience of the thoracic surgeon may be of advantage for severely injured patient.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
- Subcommittee A, American College of Surgeons' Committee on T, International Awg. Advanced trauma life support (ATLS(R)): the ninth edition. J Trauma Acute Care Surg 2013;74:1363-6.
- Kuhne CA, Ruchholtz S, Sauerland S, et al. Personnel and structural requirements for the shock trauma room management of multiple trauma. A systematic review of the literature. Unfallchirurg 2004;107:851-61. [PubMed]
- Kulshrestha P, Munshi I, Wait R. Profile of chest trauma in a level I trauma center. J Trauma 2004;57:576-81. [Crossref] [PubMed]
- Bouillon B, Probst C, Maegele M, et al. Emergency room management of multiple trauma: ATLS(R) and S3 guidelines. Chirurg 2013;84:745-52. [Crossref] [PubMed]
- Waydhas C, Sauerland S. Pre-hospital pleural decompression and chest tube placement after blunt trauma: A systematic review. Resuscitation 2007;72:11-25. [Crossref] [PubMed]
- Barton ED, Epperson M, Hoyt DB, et al. Prehospital needle aspiration and tube thoracostomy in trauma victims: a six-year experience with aeromedical crews. J Emerg Med 1995;13:155-63. [Crossref] [PubMed]
- McPherson JJ, Feigin DS, Bellamy RF. Prevalence of tension pneumothorax in fatally wounded combat casualties. J Trauma 2006;60:573-8. [Crossref] [PubMed]
- Bushby N, Fitzgerald M, Cameron P, et al. Prehospital intubation and chest decompression is associated with unexpected survival in major thoracic blunt trauma. Emerg Med Australas 2005;17:443-9. [Crossref] [PubMed]
- Mistry N, Bleetman A, Roberts KJ. Chest decompression during the resuscitation of patients in prehospital traumatic cardiac arrest. Emerg Med J 2009;26:738-40. [Crossref] [PubMed]
- Greenberg MD, Rosen CL. Evaluation of the patient with blunt chest trauma: an evidence based approach. Emerg Med Clin North Am 1999;17:41-62. viii. [Crossref] [PubMed]
- Okamoto K, Norio H, Kaneko N, et al. Use of early-phase dynamic spiral computed tomography for the primary screening of multiple trauma. Am J Emerg Med 2002;20:528-34. [Crossref] [PubMed]
- Hehir MD, Hollands MJ, Deane SA. The accuracy of the first chest X-ray in the trauma patient. Aust N Z J Surg 1990;60:529-32. [Crossref] [PubMed]
- Dulchavsky SA, Schwarz KL, Kirkpatrick AW, et al. Prospective evaluation of thoracic ultrasound in the detection of pneumothorax. J Trauma 2001;50:201-5. [Crossref] [PubMed]
- Kirkpatrick AW, Sirois M, Laupland KB, et al. Hand-held thoracic sonography for detecting post-traumatic pneumothoraces: the Extended Focused Assessment with Sonography for Trauma (EFAST). J Trauma 2004;57:288-95. [Crossref] [PubMed]
- Zhang M, Liu ZH, Yang JX, et al. Rapid detection of pneumothorax by ultrasonography in patients with multiple trauma. Crit Care 2006;10:R112. [Crossref] [PubMed]
- Rozycki GS, Feliciano DV, Ochsner MG, et al. The role of ultrasound in patients with possible penetrating cardiac wounds: a prospective multicenter study. J Trauma. 1999;46:543-51; discussion 551-2. [Crossref] [PubMed]
- Rozycki GS, Feliciano DV, Schmidt JA, et al. The role of surgeon-performed ultrasound in patients with possible cardiac wounds. Ann Surg 1996;223:737-44; discussion 744-6. [Crossref] [PubMed]
- Waydhas C. Thoracic trauma. Unfallchirurg 2000;103:871-89; quiz 90, 910.
- Gilbert TB, McGrath BJ, Soberman M. Chest tubes: indications, placement, management, and complications. J Intensive Care Med 1993;8:73-86. [Crossref] [PubMed]
- Inaba K, Lustenberger T, Recinos G, et al. Does size matter? A prospective analysis of 28-32 versus 36-40 French chest tube size in trauma. J Trauma Acute Care Surg 2012;72:422-7. [Crossref] [PubMed]
- Karmy-Jones R, Nathens A, Jurkovich GJ, et al. Urgent and emergent thoracotomy for penetrating chest trauma. J Trauma 2004;56:664-8; discussion 668-9. [Crossref] [PubMed]
- Casos SR, Richardson JD. Role of thoracoscopy in acute management of chest injury. Curr Opin Crit Care 2006;12:584-9. [Crossref] [PubMed]
- Schots JP, Vissers YL, Hulsewé KW, et al. Addition of Video-Assisted Thoracoscopic Surgery to the Treatment of Flail Chest. Ann Thorac Surg 2017;103:940-4. [Crossref] [PubMed]
- Chou YP, Lin HL, Wu TC. Video-assisted thoracoscopic surgery for retained hemothorax in blunt chest trauma. Curr Opin Pulm Med 2015;21:393-8. [Crossref] [PubMed]
- Qi Y. Clinical study on VATS combined mechanical ventilation treatment of ARDS secondary to severe chest trauma. Exp Ther Med 2016;12:1034-8. [PubMed]
- Yu PS, Chan HH, Lau RW, et al. Penetrating thoracic injury with retained foreign body: can video-assisted thoracic surgery take up the leading role in acute management? J Thorac Dis 2016;8:2247-51. [Crossref] [PubMed]
- Jin J, Song B, Lei YC, et al. Video-assisted thoracoscopic surgery for penetrating thoracic trauma. Chin J Traumatol 2015;18:39-40. [Crossref] [PubMed]
- Mizobuchi T, Iwai N, Kohno H, et al. Delayed diagnosis of traumatic diaphragmatic rupture. Gen Thorac Cardiovasc Surg 2009;57:430-2. [Crossref] [PubMed]
- Ludwig C, Morand P, Schnell J, et al. Preserving middle lobe to improve lung function in non-small-cell lung cancer. Asian Cardiovasc Thorac Ann 2009;17:153-6. [Crossref] [PubMed]