Quick spontaneous remodelling of thrombosed false lumen in acute type-A aortic dissection
Description
An 85-year-old woman was referred to intensive care unit complaining of a 10-hour evolution episode of sudden chest pain which irradiated to the back with concomitant sweating and nausea. The computed tomography (CT) diagnosis was a thrombosed acute Stanford type A aortic dissection (AAAD) with a small entry site in the aortic root and another in the abdominal aorta below both renal arteries (Figure 1). Her medical history consisted in hypothyroidism, chronic venous insufficiency and renal impairment (basal creatinine 1.8 mg/dL). She was under oral anticoagulation for paroxysmal atrial fibrillation, and losartan for blood pressure control. Ischemic heart disease was ruled out based on EKG, biochemical markers and transthoracic echocardiograms (TTE). Moderate aortic insufficiency and mild left ventricular dysfunction were previously described in the routine cardiology visit reports.
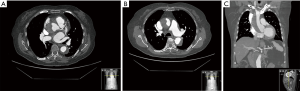
Logistic Euroscore predicted an operative death risk of 23.45%. Similar medical therapy outcomes have been reported by some authors (23.1% mortality) in the context of thrombosed false lumen AAAD, an ascending aorta smaller than 5 cm diameter and absence of significant pericardial effusion (<10 mm), as the current case described (1). Because of the patient’s age, imaging findings, haemodynamic stability, pain relief and absence of organ ischemia, after have been informed of both options, the patient agreed with a conservative management.
Arterial blood pressure (ABP) was 110/45 mmHg. Strict blood pressure control was carried out, anticoagulation reverted, and close follow-up with TTE was performed to rule out arising further complications. Echocardiographic findings consisted of a mild to moderate aortic regurgitation (4 mm vena contracta) in a tricuspid aortic valve, a 44 mm diameter ascending aorta without visible intimal flap and in absence of pericardial effusion. No changes could be addressed at 12- and 2-hour TTE control.
Hemoglobin levels remained stable. The patient didn’t experienced further pain episodes, nor dyspnoea, and was discharged from the intensive care unit after 72-hour of surveillance and beta-blockers administration for blood pressure management.
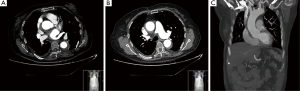
The control CT-scan, 6 days after the acute episode (Figure 2) showed an almost completely thrombosed and significant shrinkage of the false lumen with evident expansion of true lumen all along the aortic and supra-aortic trunks path. Entry site at the non-coronary sinus was considerably smaller than the reported in the previous study. At this point we restarted anticoagulation. The patient was discharged from the hospital after 13 days, and remains asymptomatic from a cardiovascular point of view after 15 months.
Discussion
Stanford type A acute aortic dissection (AAD) is a catastrophic syndrome with a reported mortality rate of 50% within 48 hours and 75% in 2-week, if it is left untreated. However, selected high risk patients for surgical intervention, have been successfully treated with a conservative approach (clotted false lumen, diameter of the aorta less than 50 mm, absence of significant pericardial effusion and hemodynamic stability) (1). Once the acute phase is overcome, a good prognosis can be achieved with medical treatment (up to 80% event free survival), as it is shown in some series (2). Surgical treatment for AAAD has been improving its outcomes and yield good prognosis data along the last years. A clotted false lumen in the ascending aorta has been described in 17% of AAD cases (3). Although both re-dissection and rupture cannot be predictable, emergent surgical treatment is commonly indicated in these patients because of these threats. Nevertheless, consistent results have been reported to conservative approach in patients with the aforementioned criteria (1). Conservative therapeutic strategies had also been applied on individual basis in elderly patients or in those with poor general condition, with a high expected surgical mortality (4). If cardiac tamponade or organ ischemia are detected, some cases should move into an emergent surgical approach (5).
False lumen clotting, that leads to fibrosis and scarring, is rare in patients under oral anticoagulant therapy (6). And according to the authors’ knowledge, there are no reports of a significant spontaneous narrowing in such a brief period of time, in this scenario.
Although there is a potential risk of sudden complication, conservative treatment may be considered in selected patients with predictable worse surgical outcomes.
Acknowledgements
None.
Footnote
Conflict of Interest: The authors have no conflicts of interest to declare.
References
- Kurimoto Y, Morishita K, Kawaharada N, et al. Initial management of acute type-A aortic dissection with a thrombosed false lumen: a retrospective cohort study. Surg Today 2004;34:652-7. [Crossref] [PubMed]
- Kozai Y, Watanabe S, Yonezawa M, et al. Long-term prognosis of acute aortic dissection with medical treatment: a survey of 263 unoperated patients. Jpn Circ J 2001;65:359-63. [Crossref] [PubMed]
- Erbel R, Oelert H, Meyer J, et al. Effect of medical and surgical therapy on aortic dissection evaluated by transesophageal echocardiography. Implications for prognosis and therapy. The European Cooperative Study Group on Echocardiography. Circulation 1993;87:1604-15. [Crossref] [PubMed]
- Rampoldi V, Trimarchi S, Eagle KA, et al. Simple risk models to predict surgical mortality in acute type A aortic dissection: the International Registry of Acute Aortic Dissection score. Ann Thorac Surg 2007;83:55-61. [Crossref] [PubMed]
- Ando T, Kobayashi T, Endo H, et al. Surgical treatment or conservative therapy for stanford type a acute aortic dissection with a thrombosed false lumen. Ann Vasc Dis 2012;5:428-34. [Crossref] [PubMed]
- Zeebregts CJ, Schepens MA, Vermeulen FE. Spontaneous resolution late after aortic dissection. Eur J Cardiothorac Surg 1997;12:513-5. [Crossref] [PubMed]


