Veno-arterial extracorporeal membrane oxygenation without therapeutic anticoagulation for intra-operative cardiopulmonary support during lung transplantation
Introduction
Anticoagulation for intraoperative mechanical cardiopulmonary support during lung transplantation is associated with significantly increased bleeding complications especially when pleural adhesions are encountered or during re-transplantation (1). Cardiopulmonary bypass machine (CBM) has a reservoir containing a stagnant column of blood that necessitates therapeutic anticoagulation to avoid thrombosis. However, veno-arterial (VA) extracorporeal membranous oxygenation (ECMO) does not have such a blood reservoir. Furthermore, technological advancements including heparin-bonded tubing, heparin-coated polymethylpentene oxygenators, and centrifugal pumps have made the components in contemporary ECMO circuits significantly less thrombogenic (2,3). Hence, we postulated that it might be possible to use VA-ECMO without therapeutic anticoagulation. We recently reported the successful anticoagulation-free veno-venous ECMO support for respiratory failure and bridge to lung transplantation (4). Here, we used VA-ECMO without therapeutic anticoagulation for intraoperative support during lung transplantation.
Case presentation
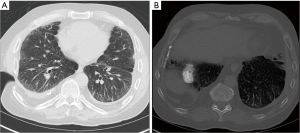
A 65-year-old male with end-stage lung disease and severe pulmonary hypertension from pneumoconiosis underwent left lung transplantation through posterolateral thoracotomy. A prior right-sided Clagett window (Figure 1A) for chronic empyema associated with significant pleural plaques (Figure 1B) precluded double lung transplantation. Intraoperatively, the patient was intolerant to right single lung ventilation and developed hypoxemia. Dense pleural adhesions and hilar scarring were encountered. The patient was, therefore, placed on VA-ECMO. A 6 mm Hemashield graft (vendor) was sewn onto the left femoral artery and used as arterial inflow. The femoral vein was directly cannulated using a 25 F Biomedicus multi-port cannula for venous drainage. The VA-ECMO was initiated and blood flows maintained at >4 liters/min. Oxygen saturations were monitored on the right hand to ensure adequate oxygenation of arch vessels. Left pneumonectomy was performed after extensive extrapleural mobilization, pleurolysis and tedious dissection of hilar structures. Subsequently, implantation of left single lung transplantation was performed with a total ischemic time of 210 minutes. The activated clotting time (ACT) was less than 160 seconds during the procedure. To de-air the allograft, pump flows were reduced to obtain right ventricular cardiac output of 1.5–2 L/min. Since we reduced the flows, we administered 5,000 U of unfractionated heparin. The allograft was ventilated after which the arterial and venous cannulas were removed, after clamping them for 5 minutes. The flow in the ECMO circuit was kept flowing using a bridge while we established hemodynamic and respiratory stability prior to removing the cannula. Circulation using an ECMO bridge was performed to facilitate going back on ECMO in case there was development of primary graft dysfunction. Total VA-ECMO time was 107 minutes. The patient had an uneventful post-operative course and was discharged breathing room air on day 10 without evidence of arterial and venous thromboembolism. The patient received routine prophylaxis using unfractionated heparin for deep vein thrombosis.
Discussion
Several centers including Toronto General Hospital, Vienna Medical Center, and Columbia University have published the feasibility of VA-ECMO during lung transplantation (5-7). The current guidelines of the Extracorporeal Life Support Organization (www.elso.org) recommend ACT levels of 180–220 for VA-ECMO. We postulate that VA-ECMO can be used safely without therapeutic anticoagulation particularly for short-term intra-operative support. To reduce thrombogenicity, we believe that the flows should be kept high and therefore adequate sized venous and arterial cannulas should be used. If there is reduction in circuit flow, for example due to peripheral vasodilation, extra fluid or vasopressors need to be administered intravenously since ECMO is a closed circuit. Alternatively, if flows over 3.5–4 L/min cannot be achieved consistently we recommend administering unfractionated heparin to reduce thrombogenicity. Our practice is to give 5,000 U of unfractionated heparin. The generally accepted guidelines for anticoagulation during VA-ECMO are based on limited or anecdotal experience (8). This case reflects equipoise in the conventional use of therapeutic anticoagulation for VA-ECMO. Prospective studies are required to evaluate use of VA-ECMO without heparin, especially in patients with high bleeding risk. Long-term use of VA-ECMO without systemic anticoagulation should also be further investigated. Avoiding or minimizing anticoagulation in selected patients may greatly improve the safety of ECMO. However, in patients with a pro-coagulant state, anticoagulation-free VA-ECMO is not advised.
Acknowledgements
We thank Ms. Elena Susan for administrative assistance in the submission of this manuscript.
Funding: This work was supported by National Institutes of Health [HL125940 to A Bharat]; and matching funds by the Thoracic Surgery Foundation [Agreement 2/3/16].
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Informed Consent: Written informed consent was obtained from the patient for publication of this manuscript and any accompanying images.
References
- Hoechter DJ, von Dossow V, Winter H, et al. The Munich Lung Transplant Group. Intraoperative Extracorporeal Circulation in Lung Transplantation. Thorac Cardiovasc Surg 2015;63:706-14. [Crossref] [PubMed]
- Bharat A, Pham DT, Prasad SM. Ambulatory Extracorporeal Membrane Oxygenation: A Surgical Innovation for Adult Respiratory Distress Syndrome. JAMA Surg 2016;151:478-9. [Crossref] [PubMed]
- Butt W, MacLaren G. Extracorporeal membrane oxygenation 2016: an update. F1000Res 2016.5. [PubMed]
- Tomasko J, Prasad SM, Dell DO, et al. Therapeutic anticoagulation-free extracorporeal membrane oxygenation as a bridge to lung transplantation. J Heart Lung Transplant 2016;35:947-8. [Crossref] [PubMed]
- Aigner C, Wisser W, Taghavi S, et al. Institutional experience with extracorporeal membrane oxygenation in lung transplantation. Eur J Cardiothorac Surg 2007;31:468-73; discussion 73-4. [Crossref] [PubMed]
- Machuca TN, Collaud S, Mercier O, et al. Outcomes of intraoperative extracorporeal membrane oxygenation versus cardiopulmonary bypass for lung transplantation. J Thorac Cardiovasc Surg 2015;149:1152-7. [Crossref] [PubMed]
- Biscotti M, Yang J, Sonett J, et al. Comparison of extracorporeal membrane oxygenation versus cardiopulmonary bypass for lung transplantation. J Thorac Cardiovasc Surg 2014;148:2410-5. [Crossref] [PubMed]
- Coughlin MA, Bartlett RH. Anticoagulation for Extracorporeal Life Support: Direct Thrombin Inhibitors and Heparin. ASAIO J 2015;61:652-5. [Crossref] [PubMed]


