Successful treatment of a ruptured left internal mammary artery aneurysm with a delayed diagnosis of type I neurofibromatosis
Introduction
Neurofibromatosis type I (NF-1), also known as von Recklinghausen's disease, is a hereditary autosomal dominant disorder that originates from neuroectodermal tumors in the peripheral nervous system. It is rare, having an incidence of 1 in 2,500 to 3,300 births (1,2). It is caused by an abnormality in the long arm of chromosome 17 (17q11.2) and is expressed in approximately 80% of those who have the gene (2). Clinical signs of NF-1 are usually associated with the following: multiple café au lait spots, variable forms of neurofibroma, Lisch nodules or iris hamartomas, freckling, abnormal bony lesions, pheochromocytoma, optic nerve glioma, and Schwann cell tumors. Vascular involvement is rare (up to 3.6% of patients), but may have life-threatening complications (3). Although there is no general consensus regarding a favorable effect, percutaneous embolization is expected to increase the chance of survival in patients with NF-1 who are hemodynamically unstable. We report the case of a patient with delayed diagnosis of spontaneous rupture of an aneurysm of a left internal mammary artery (IMA), presenting as a massive left hemothorax. This case was successfully treated with percutaneous embolization and intensive care.
Case presentation
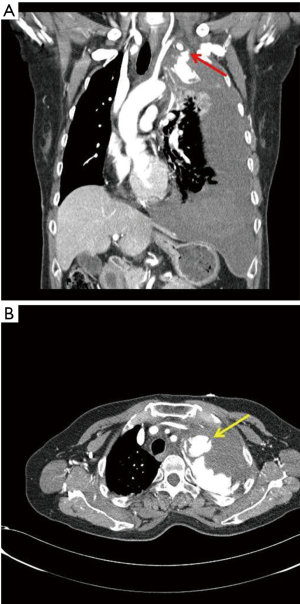
A 55-year-old female patient visited the emergency room of our hospital because of a sudden onset of pain on the left side of her chest, lasting 4 hours prior to presentation. No relevant medical history was identified. Cafe-au-lait spots, which were spread over her entire trunk, were observed during physical examination. The patient had been previously diagnosed with NF-1 at the age of 45, after presenting with skin lesions. She did not follow-up with the hospital after her diagnosis, because there were no specific symptoms to report. During this physical examination, she reported being unaware of the relationship between NF-1 and, her current chest pain. Her vital signs were stable: blood pressure, 110/70 mmHg; pulse, 100 beats/minute; respiratory rate, 22 breaths/minute; and oxygen saturation was stable at 100% in room air. Laboratory findings were also normal: hemoglobin (Hgb), 11.1 g/dL and hematocrit (Hct), 32.2%. Three hours after the start of her examination, she reported dyspnea and chest pain that did not respond to analgesics; subsequently, she went into shock. Her blood pressure decreased to 60/40 mmHg; pulse, 98 beats/minute; respiratory rate, 20 breaths/minute; and oxygen saturation, 90% under a face mask with 10 L/min of oxygen. Hgb and Hct decreased to 5.6 g/dL and 16.7%, respectively. A computed tomography (CT) scan of the chest was performed, which showed active bleeding in the proximal left IMA and massive hemothorax accompanied by mediastinal shifting (Figure 1). Two units each of packed red blood cells and fresh frozen plasma were transfused; surgery and endovascular treatment were considered as treatment methods. We had previously surgically treated one patient with NF-1, 10 years ago, but the patient died. Since that case, we developed strategies for patients with NF-1, including performing interventional treatment, if possible.
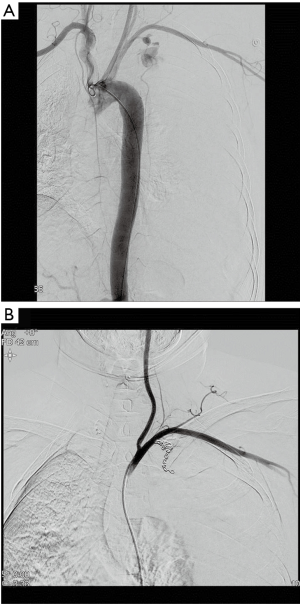
Taking into account the patient’s unstable state and our surgical history with NF-1 patients, percutaneous embolization was chosen. The patient was transferred to the intervention room for the procedure. After approaching via the right common femoral artery, an angiogram was performed. Extravasation was observed at the site of a 1.1 cm × 1.0 cm pseudoaneurysm in the same location as the bleeding. We then performed a pre-ballooning procedure using interventional coiling. After superselective catheterization of the left IMA using a microcatheter and microwire, an embolization was performed using an Interlock coil (5 each). In a follow-up angiogram, no further extravasation was observed and no further treatment was required (Figure 2). The patient’s clinical symptoms improved. She was transferred to the intensive care unit to be monitored for recurrent bleeding. A 24-Fr thoracic catheter was inserted and 1,300 mL of pleural fluid was drained, resulting in improvement of the dyspnea and hemothorax. The patient was transferred out of the intensive care unit 1 day after the treatment, and discharged 10 days after embolization, without any complications. Medication, consisting of acetaminophen 650 mg and tramadol Hcl 75 mg for pain control, was administered twice a day while she was hospitalized. There was no evidence of infection, therefore antibiotics were not used. Eighteen months later, the patient was healthy and had no findings upon examination by chest CT.
Discussion
NF-1 is a disease that involves many systems; when the arterial system is involved, it manifests as stenosis or aneurysm (4). Reubi et al. reported that intimal fibrosis, which forms due to intimal proliferation, blocks the vascular lumen and induces aneurysmal dilation of the vessel wall, but this theory does not explain all of the mechanisms (5). It has been reported that renal arteries are the most frequent locations for aneurysms in patients with NF-1, and in the thorax, aneurysms in the intercostal arteries and subclavian arteries are frequently observed. Involvement of the internal mammary arteries has rarely been reported (6). The IMA is a relatively large vessel, with an average 60 mL/min of blood flow. Although the incidence is low, even small aneurysms of the IMA are prone to bleeding, which can result in catastrophic complications (7). In total, two cases of IMA aneurysm rupture, related to NF-1, have been reported. In one case, surgery was selected because of the patient’s hemodynamic instability, and in the other case, an embolization procedure was performed; only the latter patient survived (8,9).
Patients with non-ruptured aneurysms and relatively low tissue fragility, who have undergone surgery and interventional treatment, have been previously reported in the literature (10,11). Due to the small study size, more effective treatment for patients with arterial aneurysms have not yet been determined (12). Moreover, as no multicenter randomized study has been performed to date, treatment methods for patients with ruptured aneurysms due to NF-1 have not been determined. Surgical intervention, percutaneous embolization, and a combination of the two have been considered. Some studies insist that surgery is the treatment of choice for hemodynamically compromised patients, but it is difficult to control bleeding via surgical repair of an aneurysm because of the fragility of the vessel tissue. There is a high probability of complications, and few cases are successful. Therefore, embolization has recently become the preferred method and has been shown to be an effective therapeutic option (10,13,14).We have previously performed surgical repair of aneurysms of the intercostal artery in a patient with NF-1, but the patient revisited the hospital several years later because of a ruptured aneurysm associated with an arteriovenous malformation (15,16). At that time, we chose to use an interventional procedure as the first line option of treatment. Compared with the case we previously managed, the hemodynamic instability was worse and laboratory findings also indicated more severe blood loss in this patient. Early diagnosis was delayed because the patient had stopped follow-up and was not able to listen to the initial history. Surgery is the conventional treatment for a patient in hypovolemic shock with tamponade physiology and accompanying mediastinal shifting, but we believed that embolization was the preferred treatment because of our previous experience managing patients with NF-1 surgery and the favorable results that have recently been observed (8).
To our knowledge, there are few cases in the literature comparing surgical to the interventional treatment in patients with NF- induced vasculopathy. Although surgery is considered standard treatment for aneurysmal ruptures, intervention has also been reported as a treatment option because of the challenges associated with surgery, including tissue fragility in patients with NF. In particular, patients with non-iatrogenic IMA aneurysms are very rare; therefore, the gold standard treatment for ruptured aneurysm due to NF-1 has not yet been determined.
In this case, we report that survival was achieved using a single stage of interventional treatment of the ruptured aneurysm.
There are two key points consider in this case. First, NF-induced vasculopathy can occur anywhere in the human vascular system. Therefore, when managing patients with NF, it should be taken into account that vascular involvement of sites not yet reported may also occur. We determined that the chance that our patient would survive surgery was low based on our previous experience (15). Furthermore, intervention may also be a good treatment option during an emergency. We believe that endo-repair is a plausible therapeutic option and may be helpful when choosing the appropriate treatment approach in patients with NF. We also suggest that the percutaneous embolization is preferred for the treatment of aneurysms, even if they rupture.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Informed Consent: Written informed consent was obtained from the patient for publication of this manuscript and any accompanying images.
References
- Riccardi VM. Neurofibromatosis: past, present, and future. N Engl J Med 1991;324:1283-5. [Crossref] [PubMed]
- Friedman JM, Arbiser J, Epstein JA, et al. Cardiovascular disease in neurofibromatosis 1: report of the NF1 Cardiovascular Task Force. Genet Med 2002;4:105-11. [Crossref] [PubMed]
- Dominguez J, Sancho C, Escalante E, et al. Percutaneous treatment of a ruptured intercostal aneurysm presenting as massive hemothorax in a patient with type I neurofibromatosis. J Thorac Cardiovasc Surg 2002;124:1230-2. [Crossref] [PubMed]
- Arai K, Sanada J, Kurozumi A, et al. Spontaneous hemothorax in neurofibromatosis treated with percutaneous embolization. Cardiovasc Intervent Radiol 2007;30:477-9. [Crossref] [PubMed]
- Reubi F. Neurofibromatose et lesions vasculaires (Neurofibromatosis and vascular lesions). Schweiz Med Wochenschr 1945;75:463-5.
- Oderich GS, Sullivan TM, Bower TC, et al. Vascular abnormalities in patients with neurofibromatosis syndrome type I: clinical spectrum, management, and results. J Vasc Surg 2007;46:475-84. [Crossref] [PubMed]
- Lindblom RP, Zemgulis V, Lilieqvist A, et al. Even small aneurysms can bleed: a ruptured small idiopathic aneurysm of the internal thoracic artery. Interact Cardiovasc Thorac Surg 2013;17:583-5. [Crossref] [PubMed]
- Urso S, Carballo J, Greco E, et al. Ruptured aneurysm of the mammary artery in a patient with neurofibromatosis type I. J Card Surg 2007;22:229-31. [Crossref] [PubMed]
- Kim SJ, Kim CW, Kim S, et al. Endovascular treatment of a ruptured internal thoracic artery pseudoaneurysm presenting as a massive hemothorax in a patient with type I neurofibromatosis. Cardiovasc Intervent Radiol 2005;28:818-21. [Crossref] [PubMed]
- Piffaretti G, Carrafiello G, Franchin M, et al. Stent-graft Repair of a True Internal Thoracic Artery Aneurysm. Ann Vasc Surg 2015;29:1452.e11-5. [Crossref] [PubMed]
- Okura Y, Kawasaki T, Hiura T, et al. Aneurysm of the internal mammary artery with cystic medial degeneration. Intern Med 2012;51:2355-9. [Crossref] [PubMed]
- Joshi D, James RL, Jones L. Endovascular versus open repair of asymptomatic popliteal artery aneurysm. Cochrane Database Syst Rev 2014.CD010149. [PubMed]
- Flood K, Nicholson AA. Inferior pancreaticoduodenal artery aneurysms associated with occlusive lesions of the celiac axis: diagnosis, treatment options, outcomes, and review of the literature. Cardiovasc Intervent Radiol 2013;36:578-87. [Crossref] [PubMed]
- Pezzetta E, Paroz A, Ris HB, et al. Spontaneous hemothorax associated with von Recklinghausen's disease. Eur J Cardiothorac Surg 2003;23:1062-4. [Crossref] [PubMed]
- Jang WC, Jeong IS, Lee KS, et al. Spontaneous Hemothorax in a Patient with Type I Neurofibromatosis. Korean J Thorac Cardiovasc Surg 2007;40:140-2.
- Kim HJ, Seon HJ, Choi S, et al. Ruptured aneurysm of intercostal arteriovenous malformation associated with neurofibromatosis type 1: a case report. Cardiovasc Intervent Radiol 2011;34 Suppl 2:S74-7. [Crossref] [PubMed]


