Using M-mode ultrasonography to assess diaphragm dysfunction and predict the success of mechanical ventilation weaning in elderly patients
Introduction
It has been estimated that 20–40% of patients who receive mechanical ventilation have difficulties when trying to weaning off breathing support (1-3). Animal experiments (4) suggest that control ventilation is more likely to cause diaphragmatic function decline in elderly rats. Whether this conclusion is applicable to elderly ventilated patients, and thereby affecting weaning outcomes, is poorly understood at present. Weaning from mechanical ventilation can be a long process, which account for about 40% of the total time receiving mechanical ventilation (5). One cause for weaning failure is diaphragmatic dysfunction (DD). As we know diaphragm is susceptible to infection, hypotension, hypoxia, hyperglycemia and all of which are particularly common among patients residing in intensive care units (ICU) (6,7). Furthermore, mechanical ventilation can itself lead to ventilator-induced diaphragmatic dysfunction (VIDD) (8,9). As a result, DD is a common occurrence in critically ill patients (10-15). However, data regarding the relationship between DD and mechanical ventilation weaning failure in patients are scarce (16). In our clinical experience, we observed that elderly patients who receive mechanical ventilation are particularly at risk of DD. However, the prevalence of DD and the association between DD and mechanical ventilation weaning failure in elderly patients is not yet clear.
Ultrasonography (US) is a non-invasive, easy to perform and bedside diagnosis tool for ICU clinician (17-19). The aim of this study was to evaluate diaphragmatic function measured by US and it’s predicting weaning from mechanical ventilation in elderly patients in the ICU. In this purpose, we enrolled patients more than 80 years old on the day of their first spontaneous breathing trial (SBT). Diaphragm function was assessed by M-mode US, diaphragmatic movements and diaphragmatic velocity of contraction were used to evaluate diaphragm function.
Methods
Subjects
This prospective, single-center, observational study was conducted at the medical ICU of Guangdong Geriatric Institute, a national large center of gerontology integrating clinical treatment, health care, and scientific research in South China. The study protocol was approved by the institutional ethics committee, and written informed consent was obtained from each patient’s family.
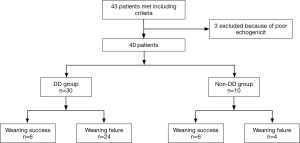
Patients were included when they met all of the following criteria: aged ≥80 years, received mechanical ventilation for >48 hours, suitable for an SBT, minimal doses of sedative and analgesic drugs were allowed but the patient must have been easily arousable (Ramsay score 3–4), muscle-paralyzing agents should have been discontinued ≥2 days before study entry, and aminoglycosides were not allowed. The exclusion criteria were as follows: patients with a history of neuromuscular diseases (myasthenia gravis, Guillain-Barre syndrome and amyotrophic lateral sclerosis), diaphragmatic paralysis, cervical injury, pneumothorax or mediastinal emphysema, and if the patient has a poor echogenicity or who were unable to tolerate US. The study flow was seen in Figure 1.
SBT
The readiness criteria for patients starting SBT include as follows: the patients’ respiratory rate ≥10 and ≤35 breaths/min, PaO2/FiO2 ratio was ≥150, with a positive end-expiratory pressure (PEEP) <5 cm H2O, FiO2 <50%, and pH value >7.25. Patients with chronic obstructive pulmonary disease (COPD) were required to have a pH >7.30, PaO2 >50 mmHg, and hemodynamic stability in the absence of vasopressors.
The SBT was conducted while the patients were connected to an independent source of oxygen (T-piece). In case of one of the following situations, the SBT was considered as a failure: SBRI >105, respiratory rate <10 and >35 breaths/min£¨heart rate >140 beats/min or changed <20% compared with the baseline or the onset of new arrhythm, tidal volume <4 mL/kg, SaO2 <90%.
Minimal ventilator settings [pressure support (PS) of 5–7 and 3–5 cmH2O PEEP] were allowed to overcome the increased work of breathing through the circuit. The initial SBT lasted a minimum of 30 minutes, up to a maximum of 120 minutes. If it is not clear that the patient had passed after 120 minutes, the SBT was considered a failure.
US
The patient’s rapid shallow breathing index (RSBI) was calculated at the bedside before SBT. RSBI = respiratory rate (breaths/min)/Tidal volume (L). During the first 30 minutes of SBT, each hemidiaphragm was evaluated using M-mode US performed by a well-trained expert using an EPIQ5 machine (Philips, Holland) connected to a 1–5 MHz curved array probe. Patients were in the semi-recumbent position. US was discontinued if the patient exhibited any signs of breathing fatigue.
Diaphragm movements were measured as previously described (20). Briefly, the two-dimensional mode was used to find the best approach and to select the exploration line of each hemi-diaphragm. During respiration and with the probe on the chest wall, the US beam was directed to the hemi-diaphragmatic domes at an angle of not <70° (9). Diaphragm contraction during inspiration was observed as an upward motion of the M-mode tracing. The diaphragmatic movement was measured on the vertical axis of the tracing, from the baseline to the point of maximum height of inspiration on the graph. The time of diaphragmatic contraction was defined as the difference between the beginning of inspiration and when the peak was reached during a quiet breath (21). Diaphragmatic velocity of contraction (mm/s) was calculated as diaphragmatic movement (mm) divided by the duration of diaphragmatic contraction (s). Six measurements were averaged for each side. All measurements were collected within 5 minutes and were performed during resting breathing, excluding smaller or deeper breaths.
Outcomes
DD was diagnosed if an excursion was <10 mm or negative, with negative indicating a paradoxical diaphragmatic movement (20,22-24). Patients were divided according to US findings into those experiencing DD and those without DD. All examinations were analyzed in a blinded manner.
Weaning was considered successful if the patient could maintain spontaneous breathing for ≥48 hours with no need for any level of ventilator support after extubation. Weaning after the SBT was achieved by the support of high-flow nasal cannulae (HFNC). Attending physicians were blind to the US results.
Weaning outcomes were as follows: weaning failure (y/n), weaning time (hours; time spent receiving partial support i.e., PS or continuous positive airway pressure, minus the full support period i.e., including volume-controlled or pressure-controlled support), and total mechanical ventilation time (hours). Factors which are known to affect weaning outcome were noted e.g., underlying diseases such as diabetes mellitus, COPD, acute respiratory distress syndrome, time to the SBT, and relevant blood biochemistry findings. The sensitivity, specificity, positive and negative likelihood ratios of DD in predicting the outcome of weaning was calculated and compared with that of RSBI.
Statistical analysis
Averaged data were expressed as mean ± standard deviation (SD) or median and interquartile range for continuous variables and as absolute or relative frequencies for categorical variables. Independent sample t-test or Mann-Whitney U tests were used to compare continuous variables and chi-square test or Fisher’s exact tests were used for categorical variables. Receiver operating characteristic (ROC) curve analysis was performed to determine the diagnostic efficacy of ultrasonographic DD and of RSBI for predicting weaning failure. All data was handled in SPSS 19.0 and P<0.05 was considered significant.
Results
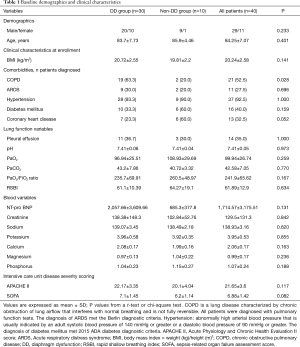
In total, 43 consecutive Chinese patients were enrolled but three were later excluded from the study due to poor US imaging quality between January 2015 and June 2015. Of the 40 remaining patients, the overall mean age was 84.25±7.07 years, and 29 (72.5%) were male (Table 1). US-defined DD was present in 30/40 patients (75.0%); as unilateral in 26 (right DD in nine, left DD in 17), and bilateral in four. Demographic factors, comorbidity, and laboratory findings did not differ significantly between the DD and non-DD groups, except for COPD, which was more prevalent in the DD group (P=0.028; Table 1).
Full table
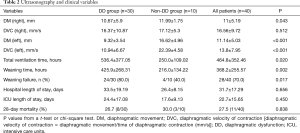
Mean diaphragmatic movements (both sides; mm) and left diaphragmatic velocity of contraction (mm/s) were significantly lower among patients of the DD group, as compared with the non-DD group. Mean diaphragmatic movements were 10.67±5.9 mm in the right and 9.32±3.54 mm in the left diaphragm of the DD group, compared with 11.99±1.75 and 16.62±4.96 mm in the non-DD group, respectively (Table 2). Mean left diaphragmatic velocity of contraction was 10.94±6.67 mm in the DD group compared with 22.39±4.58 in the non-DD group, with no significant differences in the right sides (Table 2). RSBI before the SBT did not differ significantly between the DD and non-DD groups (61.1±10.39 vs. 64.27±19.1, P=0.634; Table 1). Weaning time (mean: 536.4±377.05 vs.250±109.02 hours, respectively, P=0.02) and total mechanical ventilation time (mean: 425.9±268.31 vs. 216.0±134.22 hours, respectively, P=0.002) were significantly longer in the DD group than in the non-DD group (Table 2). Furthermore, the DD group showed higher incidence of weaning failure than in the non-DD group. (80.0% vs. 40.0% P=0.017; Table 2) There was no significantly difference in ICU and hospital lengths of stay, 28-day mortality between two groups. Among all patients, the overall incidence of weaning failure was 70.0% (28/40). For the 12 patients with weaning success, 7 patients had unilateral DD, and 2 patients exhibited high contralateral diaphragmatic activity (29.0 mm and 31.2 mm), and a fast diaphragmatic contraction rate.
Full table
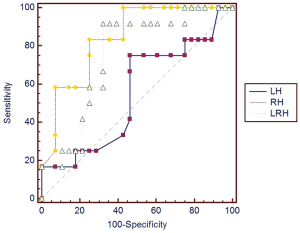
The best cut-off values for weaning success were calculated for diaphragmatic movement, diaphragmatic velocity of conduction and RSBI, and were significant by area under the curve. We performed a ROC curve analysis to assess the predictive value of these variables. The right diaphragmatic movement exhibited high diagnostic values (Table 3) with the the area under the receiver operating characteristic curve (AUROC) of 0.839 (95% CI, 0.689–0.936). The sensitivity, specificity for predicting weaning success were 83.33% and 75%, the positive and negative likelihood ratios were 3.33 and 0.22. The right diaphragmatic velocity of contraction also exhibited high diagnostic values (Table 3) with the AUROC of 0.833 (95% CI, 0.682–0.932). The sensitivity, specificity for predicting weaning success were 66.67% and 92.86%, the positive and negative likelihood ratios were 9.33 and 0.36. However, both the left diaphragmatic movement and diaphragmatic velocity of contraction showed poor diagnostic value.

Full table
Notably, the predictive values of these two parameters were higher than for RSBI (Table 3).
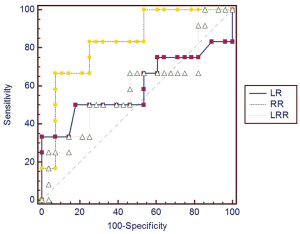
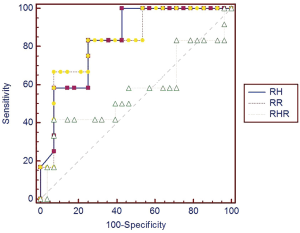
We further examined if a combination of several parameters could provide enhanced predictive power (Figures 2-5). However, predictive capacity was highest when variables were assessed individually.

Discussion
There is an increasing demand for mechanical ventilation to support the care of those who find themselves critically ill, including amongst elderly patients (25). It is therefore important to consider the factors that affect and predict the success of ventilator weaning. The diaphragm is vital for the control of breathing. Critically ill patients, in particular those receiving mechanical ventilation, often experience DD (10,13), which can make it harder for them to achieve successful ventilator weaning. As elderly patients can have high levels of comorbidity, and experience high incidence of DD, it is particularly important to investigate the relationship between DD and weaning in this patient group. In this study of elderly patients, we found that higher US measured right diaphragm movement and right diaphragm velocity of contraction were predictive of ventilator weaning success.
US is ideally suited for bedside diagnostic examinations and has become a vital tool in the management of critically ill patients (26-28). Diaphragm US is able to provide accurate and reliable measurements of diaphragmatic function in ICU patients (29-31), however, few studies have reported the use of M-mode US to diagnose DD and predict weaning outcomes in mechanically ventilated patients.
We assessed 40 elderly (≥80 years of age) patients at the beginning of a SBT. DD was determined using US as a diaphragmatic excursion less than 10 mm from end-expiration to end-inspiration, or a negative value (paradoxical movement). We saw a high prevalence of DD (75%) among our patients, who had been mechanically ventilated for a mean duration of 464.8±352.46 hours. For the majority (75%) this was due to diaphragm muscle dysfunction (unilateral 26 and bilateral 4 patients). As expected, these results suggest that a longer duration of ventilation predisposes to DD. The total ventilation duration was significantly longer among patients with DD (536.4±377.05 hours for patients with DD compared with 250.0±109.02 hours for those without).
The prevalence of DD in the elderly ICU patients enrolled in our study was surprising, but may be even higher among the general elderly population (including the contribution of patients left out of this study due concomitant conditions). Our findings also confirm that DD is more common among patients with COPD, which is consistent with previous reports (32).
US measurements were performed during resting breathing, excluding small or deep breaths. The subsequent analysis was performed by blinded individuals. To reduce operator error, all US examinations were performed by two persons. Moreover, because diaphragmatic motion varies at different parts of the diaphragm, B-mode US was used to select the area of diaphragm with maximum motion for M-mode US. We observed a 70% (28/40 patients) incidence of weaning failure. Failure was much more common among patients with DD (80% vs. 40%; P=0.017). The reasons behind these weaning failures are likely to be complex. However, weaning and mechanical ventilation time was significantly longer in patients with diaphragm muscle dysfunction. We measured significantly smaller mean right and left diaphragmatic motion, and slower left velocity of contraction among patient with DD.
We saw that DD was significantly associated with longer total mechanical ventilation duration and weaning time. For the 12 patients with weaning success, 7 patients had unilateral DD, and 2 patients exhibited higher contralateral diaphragmatic activity (29.0 and 31.2 mm), and a faster diaphragmatic contraction rate. We speculate that for elderly patients with DD, weaning may succeed upon removal of the reversible causes of disease worsening, proper compensation from a relatively “normal” diaphragm, and restoration (or near restoration) of the patient’s pre-worsening pathophysiological state. In addition, we routinely used HFNC (33) for the weaning tests, which may have contributed to weaning success in the DD patients.
ROC curve analysis showed that a right diaphragmatic motion >10.7 mm, and a right diaphragm contraction rate >21.32 mm/s represent possible predictors for a successful weaning outcome in elderly patients. The AUROC curves for these variables were 0.839 and 0.833, respectively, and exhibited a higher predictive value than the RSBI (cut-off >68, AUC =0.695). RSBI is the conventional parameter for predicting weaning outcome (34); however, for the elderly patients in this study population, the weaning failure rate was high and the mean pre-weaning RSBI was 61.89±12.9. We speculate that this observation may be associated with the imbalance of respiratory load and ability, as well as inadequate respiratory endurance in elderly patients. Our results also suggested that a left diaphragmatic contraction rate ≤2.16 mm/s was predictive of weaning success; however, the predictive value was low (AUC =0.595). Moreover, several patients exhibited flat left diaphragm activity that directly contributed to the lower left diaphragmatic contraction rate, which lead to the erroneous conclusion that a low left diaphragmatic contraction rate is associated with a high weaning success rate. We speculate that this nonsensical result may be a result of the small sample size, which is a limitation for our study. Other limitations include a lack of epidemiological data on the normal diaphragmatic US measurements in elderly patients, that a definition of DD as diaphragm activity that may not have been ideal (<10 mm).
US evaluation of diaphragmatic function has broad array of applications (35). Indeed, many other studies (36,37) have shown that diaphragmatic thickness (atrophy) determines the length of ventilation or failure to wean. According to our past experience, it is difficult to measure the thickness of diaphragms. Of course, this study did not include the thickness of the diaphragm, which belongs to the design defect of this study. In follow-up studies, we will focus on the impact of diaphragmatic thickness on weaning outcomes in critically ill elderly patients. With respect to echocardiography and lung US in ICU patients, data from diaphragmatic ultrasonic imaging evaluations remain scarce, especially in elderly patients. Furthermore, governments and organizations have not developed uniform guidelines on the standards and methodology for US in the weaning of patients from mechanical ventilation. Our study shows that diaphragmatic US imaging of right diaphragm activity and the right diaphragm contraction rate could be used to guide the weaning of elderly patients off of mechanical ventilation.
Conclusions
Our results show that DD is more common in critically ill elderly patients, and may associated with an increased incidence of mechanical ventilation weaning failure. Right diaphragm movement and right diaphragm contraction velocity may exhibit slightly high predictive values for a successful weaning outcome in elderly patients on mechanical ventilation. Given the small sample size of this study, larger, randomized, controlled trials could be useful to validate these findings.
Acknowledgements
Funding: This work was supported by grant from Medical Scientific Research Foundation of Guangdong Province, Guangdong, People’s Republic of China. (Grant number: C2015046 and B2015076)
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: The study protocol was approved by the ethics committee of Guangdong General Hospital/Guangdong Academy of Medical Sciences [No. GDREC2015106H (R1)] and written informed consent was obtained from each patient’s family.
References
- Esteban A, Frutos F, Tobin MJ, et al. A comparison of four methods of weaning patients from mechanical ventilation. Spanish Lung Failure Collaborative Group. N Engl J Med 1995;332:345-50. [Crossref] [PubMed]
- Thille AW, Harrois A, Schortgen F, et al. Outcomes of extubation failure in medical intensive care unit patients. Crit Care Med 2011;39:2612-8. [Crossref] [PubMed]
- McConville JF, Kress JP. Weaning patients from the ventilator. N Engl J Med 2012;367:2233-9. [Crossref] [PubMed]
- Andrade PV, dos Santos JM, Silva HC, et al. Influence of hyperoxia and mechanical ventilation in lung inflammation and diaphragm function in aged versus adult rats. Inflammation 2014;37:486-94. [Crossref] [PubMed]
- Esteban A, Alía I, Ibañez J, et al. Modes of mechanical ventilation and weaning. A national survey of Spanish hospitals. The Spanish Lung Failure Collaborative Group. Chest 1994;106:1188-93. [Crossref] [PubMed]
- Khan J, Harrison TB, Rich MM. Mechanisms of neuromuscular dysfunction in critical illness. Crit Care Clin 2008;24:165-77. [Crossref] [PubMed]
- Herridge MS, Batt J, Hopkins RO. The pathophysiology of long-term neuromuscular and cognitive outcomes following critical illness. Crit Care Clin 2008;24:179-99. [Crossref] [PubMed]
- Jaber S, Jung B, Matecki S, et al. Clinical review: ventilator-induced diaphragmatic dysfunction--human studies confirm animal model findings! Critical Care 2011;15:206. [Crossref] [PubMed]
- Hermans G, Agten A, Testelmans D, et al. Increased duration of mechanical ventilation is associated with decreased diaphragmatic force: a prospective observational study. Crit Care 2010;14:R127. [Crossref] [PubMed]
- Powers SK, Wiggs MP, Sollanek KJ, et al. Ventilator-induced diaphragm dysfunction: cause and effect. Am J Physiol Regul Integr Comp Physiol 2013;305:R464-77. [Crossref] [PubMed]
- Haitsma JJ. Diaphragmatic dysfunction in mechanical ventilation. Curr Opin Anaesthesiol 2011;24:214-8. [Crossref] [PubMed]
- Powers SK, Kavazis AN, Levine S. Prolonged mechanical ventilation alters diaphragmatic structure and function. Crit Care Med 2009;37:S347-53. [Crossref] [PubMed]
- Lerolle N, Guérot E, Dimassi S, et al. Ultrasonographic diagnostic criterion for severe diaphragmatic dysfunction after cardiac surgery. Chest 2009;135:401-7. [Crossref] [PubMed]
- Azuelos I, Jung B, Picard M, et al. Relationship between Autophagy and Ventilator-induced Diaphragmatic Dysfunction. Anesthesiology 2015;122:1349-61. [Crossref] [PubMed]
- Mrozek S, Jung B, Petrof BJ, et al. Rapid onset of specific diaphragm weakness in a healthy murine model of ventilator-induced diaphragmatic dysfunction. Anesthesiology 2012;117:560-7. [Crossref] [PubMed]
- Kim WY, Suh HJ, Hong SB, et al. Diaphragm dysfunction assessed by ultrasonography: influence on weaning from mechanical ventilation. Crit Care Med 2011;39:2627-30. [Crossref] [PubMed]
- Umbrello M, Formenti P, Longhi D, et al. Diaphragm ultrasound as indicator of respiratory effort in critically ill patients undergoing assisted mechanical ventilation: a pilot clinical study. Crit Care 2015;19:161. [Crossref] [PubMed]
- Goligher EC, Laghi F, Detsky ME, et al. Measuring diaphragm thickness with ultrasound in mechanically ventilated patients: feasibility, reproducibility and validity. Intensive Care Med 2015;41:734. [Crossref] [PubMed]
- He W, Xu Y. Ultrasound assessment of diaphragm mobility by three different approaches in ventilated patients. Zhonghua Wei Zhong Bing Ji Jiu Yi Xue 2014;26:914-6. [PubMed]
- Boussuges A, Gole Y, Blanc P. Diaphragmatic motion studied by m-mode ultrasonography: methods, reproducibility, and normal values. Chest 2009;135:391-400. [Crossref] [PubMed]
- Scott S, Fuld JP, Carter R, et al. Diaphragm ultrasonography as an alternative to whole-body plethysmography in pulmonary function testing. J Ultrasound Med 2006;25:225-32. [Crossref] [PubMed]
- Houston JG, Morris AD, Howie CA, et al. Technical report: quantitative assessment of diaphragmatic movement--a reproducible method using ultrasound. Clin Radiol 1992;46:405-7. [Crossref] [PubMed]
- Gerscovich EO, Cronan M, McGahan JP, et al. Ultrasonographic evaluation of diaphragmatic motion. J Ultrasound Med 2001;20:597-604. [Crossref] [PubMed]
- Ayoub J, Cohendy R, Dauzat M, et al. Non-invasive quantification of diaphragm kinetics using m-mode sonography. Can J Anaesth 1997;44:739-44. [Crossref] [PubMed]
- Orsini J, Butala A, Salomon S, et al. Prognostic factors associated with adverse outcome among critically ill elderly patients admitted to the intensive care unit. Geriatr Gerontol Int 2015;15:889-94. [Crossref] [PubMed]
- Rocco M, Carbone I, Morelli A, et al. Diagnostic accuracy of bedside ultrasonography in the ICU: feasibility of detecting pulmonary effusion and lung contusion in patients on respiratory support after severe blunt thoracic trauma. Acta Anaesthesiol Scand 2008;52:776-84. [Crossref] [PubMed]
- Beaulieu Y, Marik PE. Bedside ultrasonography in the ICU: part 2. Chest 2005;128:1766-81. [Crossref] [PubMed]
- Beaulieu Y, Marik PE. Bedside ultrasonography in the ICU: part 1. Chest 2005;128:881-95. [Crossref] [PubMed]
- Baria MR, Shahgholi L, Sorenson EJ, et al. B-mode ultrasound assessment of diaphragm structure and function in patients with COPD. Chest 2014;146:680-5. [Crossref] [PubMed]
- Gethin-Jones TL, Noble VE, Morse CR. Quantification of diaphragm function using ultrasound: evaluation of a novel technique. Ultrasound Med Biol 2010;36:1965-9. [Crossref] [PubMed]
- Matamis D, Soilemezi E, Tsagourias M, et al. Sonographic evaluation of the diaphragm in critically ill patients. Technique and clinical applications. Intensive Care Med 2013;39:801-10. [Crossref] [PubMed]
- Ottenheijm CA, Heunks LM, Dekhuijzen RP. Diaphragm adaptations in patients with COPD. Respir Res 2008;9:12. [Crossref] [PubMed]
- Frat JP, Thille AW, Mercat A, et al. High-flow oxygen through nasal cannula in acute hypoxemic respiratory failure. N Engl J Med 2015;372:2185-96. [Crossref] [PubMed]
- Yang KL, Tobin MJ. A prospective study of indexes predicting the outcome of trials of weaning from mechanical ventilation. N Engl J Med 1991;324:1445-50. [Crossref] [PubMed]
- Doorduin J, van Hees HW, van der Hoeven JG, et al. Monitoring of the respiratory muscles in the critically ill. Am J Respir Crit Care Med 2013;187:20-7. [Crossref] [PubMed]
- DiNino E, Gartman EJ, Sethi JM, et al. Diaphragm ultrasound as a predictor of successful extubation from mechanical ventilation. Thorax 2014;69:423-7. [Crossref] [PubMed]
- Schepens T, Verbrugghe W, Dams K, et al. The course of diaphragm atrophy in ventilated patients assessed with ultrasound: a longitudinal cohort study. Crit Care 2015;19:422. [Crossref] [PubMed]