Predictors of long-term adherence to continuous positive airway pressure in patients with obstructive sleep apnoea and acute coronary syndrome
Introduction
Obstructive sleep apnoea (OSA) is a highly prevalent respiratory disorder that affects 6–13% of the general population or more, depending on gender and age group (1,2). OSA is characterized by recurrent episodes of upper airway collapse that result in sleep fragmentation, intermittent hypoxia and the disruption of sleep architecture. OSA is associated with daytime symptoms, a decrease in the quality of life and an increase in morbidity and mortality from metabolic, neural and cardiovascular alterations (3-5).
Continuous positive airway pressure (CPAP) is an effective treatment for OSA and is recommended for symptomatic patients with an apnoea-hypopnea index (AHI) ≥5 and for patients with an AHI ≥15 regardless of symptoms (6). The effectiveness of CPAP therapy in improving clinical symptoms, quality of life and potential consequences associated with OSA is strongly related to compliance (7-10); nevertheless, the predictors of adherence to this therapy are still not well known.
The most typical symptom of OSA is excessive daytime sleepiness. Most of the studies assessing compliance have been conducted on patients with hypersomnia, where excessive daytime sleepiness has been suggested as one of the most important determinants of adherence to CPAP. Although several studies have reported that excessive daytime sleepiness is absent in approximately 50% of all OSA patients (11), very few studies have focused on non-sleepy patients. The results of these studies are contradictory (10,12-14), with some authors reporting similar CPAP adherence regardless of daytime sleepiness and others finding differences.
It is estimated that in patients with acute coronary syndrome (ACS) the prevalence of sleep apnea hypopnea syndrome (SAHS) is higher than in the general population (15). Nevertheless, the evaluation of respiratory disorders during sleep is not routinely considered in guidelines for diagnosis and evaluation of patients with ACS (16). In addition, CPAP adherence has been rarely carried out among patients with ACS and it may be different from that of other OSA patients.
In this context, we designed this study to analyse long-term adherence and identify the predictors of non-compliance with CPAP treatment for patients with non-sleepy OSA who have suffered an ACS.
Methods
Patients
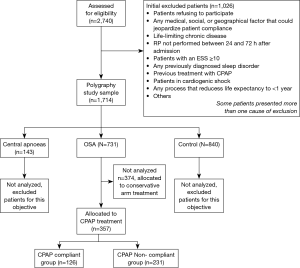
This is an ancillary study of the ISAACC study, which is a multicentre, prospective, open-label, parallel, randomized, controlled trial (NCT01335087) to evaluate the effect of CPAP treatment on the recurrence of cardiovascular events in non-sleepy patients with ACS and co-occurring OSA (17). Briefly, the study started in June 2011, and patients consecutively admitted for ACS to coronary care units or cardiology hospitalization rooms at 15 teaching hospitals in Spain were evaluated in regard to the inclusion and exclusion criteria. The ethics committee of each participating centre approved the study (CEIC-852).
ACS was defined as the acute presentation of coronary disease with or without ST-elevation infarction, unstable angina or type 1 myocardial infarction (18). The inclusion criteria were patients aged greater than 18 years, admitted to the hospital for ACS with or without ST-segment elevation, without excessive daytime sleepiness [Epworth Sleepiness Scale (ESS) score≤10], and having signed informed consent.
The exclusion criteria included previous treatment with CPAP, psychophysical inability to complete questionnaires, having 50% central apnoea or Cheyne-Stokes respiration, having a previously diagnosed sleep disorder or chronic disease (neoplasm, renal failure, severe chronic obstructive pulmonary, chronic depression, or other limiting chronic diseases), having a medical history that could interfere with the study objectives, having a life expectancy lower than 1 year or being in cardiogenic shock.
All patients included underwent cardiorespiratory polygraphy during the first 24–72 hours after admission, and patients presenting an (AHI) ≥15 events/hour were randomized to receive conservative care or CPAP treatment.
Between June 2011 and June 2014, the ISAACC study included 731 patients aged more than 18 years old who were diagnosed with moderate or severe OSA (AHI ≥15) without excessive daytime sleepiness (ESS ≤10). For the purpose of this study, only patients who were randomized to receive CPAP treatment (n=357) were considered for analysis of adherence to CPAP treatment. A flowchart for study participation in the study period is provided in Figure 1.
Procedures
All participants underwent overnight cardiorespiratory polygraphy with the same model of device (Embletta; ResMed, Australia). Nasal flow, thoracoabdominal effort and oximetry were recorded. All sleep studies were manually scored by trained personnel at each participating centre according to standard criteria (19). Apnoea was defined as an absence of airflow lasting ≥10 seconds. Hypopnea was defined as a reduction in airflow lasting ≥10 seconds associated with a decrease in arterial oxygen saturation (SaO2) ≥4%. The AHI was defined as the number of episodes of apnoea and hypopnea per hour of recording.
Baseline variables were systematically recorded using a standardized protocol. These variables included clinical and anthropometric data, subjective sleepiness/drowsiness based on the Spanish version of the ESS (20), quality of life (EuroQol test), smoking habit, and alcohol intake, and cardiovascular health was assessed through by collecting information regarding a history of diabetes, hypertension, dyslipidaemia, first episode of ACS, cardiomyopathy, stroke, neuropathy and neurological disease.
CPAP titration was performed by automatic CPAP following a validated protocol (21) on a second night. After titration, the patients were treated with fixed CPAP. The optimal pressure was determined visually on the raw data of the automatic CPAP device by analyzing the pressure that included 90% of the periods with a leak lower than 0.4 L/second (percentile 90). All these patients also received advice on conservative treatment based on dietary-hygienic measures.
Follow-up and endpoints
The follow-up period was extended until June 2015. Thus, CPAP treatment continued on an updated basis for at least 1 year for all the participants. Patients who interrupted CPAP treatment continued with their scheduled clinical visits. Patients were followed up and were evaluated at baseline, 1, 3, 6 and 12 months.
The goal of the visits at baseline and 1 month was to facilitate adaptation and adherence to CPAP treatment and to insist on dietary-hygienic advice. At each visit, CPAP use was reinforced and side effects or concerns related to treatment were addressed. All collaborating centres have extensive experience in the management of CPAP treatment. All the patients had a phone number that allowed them to contact the research team at any time. If any problems related to the adaptation to CPAP occurred, additional visits were scheduled in order to immediately resolve them. CPAP devices were provided free of charge. Compliance with CPAP treatment was objectively assessed by reading the internal counter of the device at each follow-up visit.
The endpoints of this study were CPAP treatment compliance and variables associated with adherence to treatment. For this purpose, lack of adherence was defined as mean CPAP use less than 4 hours per day at 12 months of CPAP treatment. Those patients with early known withdrawals from the trial or CPAP treatment were classified in the lack of adherence group. Patients who dropped out before completing 12 months of follow-up (and therefore who had missing compliance, including patients who were completely or partially lost to follow-up) were also classified in the lack of adherence group.
Data analysis
Non-normally distributed quantitative variables were summarized by their median (and interquartile intervals via the 25th and 75th percentiles). Qualitative variables were summarized by absolute and relative frequencies.
The association between CPAP lack of adherence at 12 months of follow-up and patient characteristics of clinical interest at baseline was tested using a Mann-Whitney U-test for continuous variables and/or a t-test for continuous non-normally or normally distributed variables. A Chi-square test was used to test qualitative variables.
Independent predictors of CPAP adherence were identified by performing multivariable logistic regression analysis starting with a model of all variables with a P value lower than 0.20 associated with their coefficients in the unadjusted analysis. The selection of variables included in the final model was based on the statistical significance of their coefficients. The final model was adjusted by hospital. Model calibration and discrimination were assessed, and the Hosmer-Lemeshow test and area under the curve (AUC) were estimated. Additionally, a mixed-effect logistic regression model was adjusted to test the random effects of the hospitals. A significance level of 0.05 was applied. R statistical software was used for data processing and analysis (22).
Results
The first 357 ACS patients who were diagnosed with OSA and randomized to CPAP treatment were analysed.
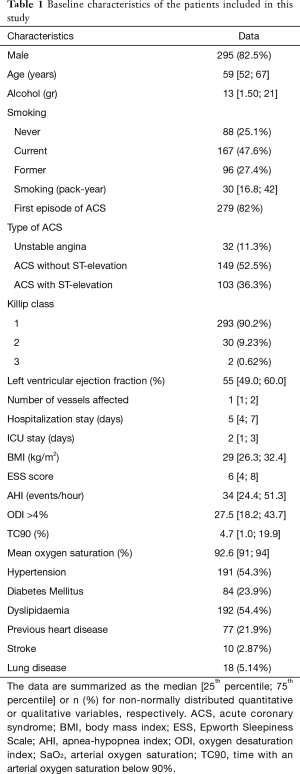
The patients’ baseline characteristics are described in Table 1. Briefly, 82.5% were men, the median age was 59 years (IQR, 52–67), and the median body mass index (BMI) was 29 kg/m2 (IQR, 26.3–32.4). For most patients, it was their first episode of ACS (82%). Concerning the respiratory variables, the patients had severe OSA (median AHI of 34 events/hour; IQR, 24.4–51.3), with a median time with SaO2 below 90% (TC90) of 4.7% (IQR, 1–19.9) and a median ESS score of 6 (IQR, 4–8).
Full table
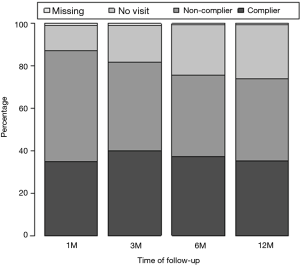
After 12 months, 4 patients had died and 88 were lost to follow-up (last bar in Figure 2). At the end of the study, 126 patients (35.3%) completed the 12-month follow-up period and reported good compliance, 54 (15.1%) reported good compliance after 1, 3 or 6 months but discontinued CPAP treatment before the last 12-month visit, and 177 (49.6%) showed non-compliance.
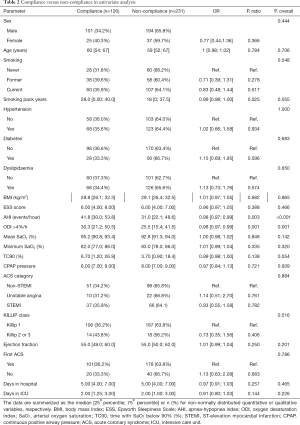
According to the unadjusted analysis (Table 2), higher AHI and oxygen desaturation index (ODI), both of which are indicators of OSA severity, were associated with a lower risk of becoming a non-complier of CPAP treatment after 12 months. No statistically significant differences were identified between the groups in their sociodemographic characteristics, medical history or ACS variables, such as ACS type, Killip class, left ventricle ejection fraction or number vessels affected.
Full table
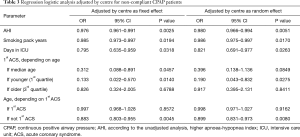
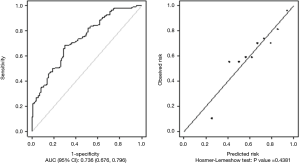
The results of multivariable analysis to identify the characteristics of patients associated with an increased risk of non-adherence to CPAP in a 12-month treatment period are shown in Table 3. The identified patient characteristics significantly associated with a lower risk of long-term non-compliance with CPAP treatment were high AHI values, high amounts of smoking pack years, and longer intensive care unit (ICU) stays. The same adjusted regression model identified a significant interaction between age and ACS personal history with a lower risk of non-compliance with CPAP treatment among patients without a personal history of ACS (for younger patient age) in comparison to patients with recurrent ACS. In addition, patients with recurrent ACS showed lower odds of CPAP non-compliance associated with ageing. The calibration and discrimination capability of this multivariable model to predict long-term non-compliance is shown in Figure 3, which indicates no significant lack of calibration (Hosmer-Lemeshow test, P=0.44) and an AUC of 0.736.
Full table
Discussion
This study shows that patients hospitalized due to an ACS who were newly diagnosed with OSA without sleepiness and who started CPAP treatment had low adherence to treatment, which was approximately 35.3% at 12 months. Predictors of compliance with CPAP treatment at 12 months were OSA severity, smoking pack years, ICU stay length, age and a personal history of ACS. High values of AHI, smoking pack years or ICU stay length were associated with a lower risk of non-compliance with CPAP treatment. The adjusted analysis also showed a significant interaction between age and a previous ACS personal history associated with CPAP compliance. Thus, younger patients showed the highest non-compliance rates when having a recurrent ACS compared to patients without any previous history of ACS.
A better understanding of the predictors of CPAP compliance is interesting because adherence to CPAP treatment is essential to achieving the beneficial effects of CPAP (23,24). Therefore, knowing the predictors of non-compliance may help in the development of clinical practice strategies directed towards improving adherence to treatment in groups with a high risk of poor compliance.
In most studies assessing this topic, sleepiness is suggested as the most relevant predictor of CPAP compliance. Nevertheless, approximately 40–50% of patients suffering from OSA do not report sleepiness (11,25,26); thus, it is also important to determine which variables can predict compliance in these patients. However, few studies have focused on non-sleepy patients, and the results of those studies have been conflicting.
In our study, only 35.3% of patients reported good compliance with CPAP treatment after 12 months. Although CPAP compliance is usually higher in patients with sleepiness than in non-sleepy ones (27,28), our results differ from adherences reported in other studies performed in non-sleepy patients. This fact could be related to the place where patients were recruited or the symptoms that they presented at baseline. Campos-Rodriguez et al. (12) and Gagnadoux et al. (13) reported adherences to CPAP treatment of approximately 64% and 67%, respectively, in non-sleepy patients. The difference between our results and these studies might be explained by the fact that both of the previous studies included patients who were recruited in sleep clinics. Thus, despite not having sleepiness, the patients probably exhibited other symptoms, such as snoring or witnessed apnoea, because they were suspicious of having OSA, and these other symptoms may motivate compliance. In contrast, our non-sleepy patients were evaluated during an ACS but were not being evaluated for suspected OSA. Nevertheless, there are also studies describing poor compliance in clinical trials performed in non-sleepy patients recruited in sleep units (10). The adherence shown in our study is similar to the values reported by Chai-Coetzer et al. (14) in the SAVE trial in which patients did not present to a sleep clinic and were mainly recruited in neurology or cardiology clinics, as in our study. The values of CPAP use and adherence reported in the SAVE trial did not significantly differ from those reported in our study (CPAP mean use: 3.3±2.4 hours per night and adherence of approximately 39%). Therefore, in line with the existing literature, our findings suggest that adherence to CPAP therapy is more difficult in non-sleepy patients than in more symptomatic ones, especially when treatment is initiated outside of a sleep unit and during hospitalization.
This study also shows that compliance rate remains fairly similar between the first follow-up visit at 1 month and at the end of the study at 12 months. These data are concordant with studies reporting that use obtained within the first months is an important determinant of long-term compliance (28,29). This fact suggests that patient’s early experience and initial comfort with CPAP, even during the titration night, may impact future compliance (30). Moreover, it suggests that there is probably a window of opportunity shortly after starting CPAP treatment to influence future compliance. Early identification of these patients could be useful to increase patient education and motivation (when possible) and improve compliance (31) or to allocate resources and propose alternative treatments in the other cases.
Moreover, our data also depict that after CPAP titration, there is a non-negligible percentage of patients with poor compliance or those who do not continue with follow-up visits. On one hand, these data could be related to the hospital admission for an ACS and not for suspected OSA. Thus, these patients probably have low awareness of the importance of OSA diagnosis and treatment. On the other hand, the patients included in our study are minimally symptomatic and probably could not perceive clear benefits from the treatment, which has been reported in previous studies as the main cause for discontinuing CPAP treatment (28).
The study also shows that high values of AHI, smoking pack years or ICU stay length were associated with a lower risk of non-compliance. Our results agree with previous studies reporting that the severity of OSA was a determinant of long-term adherence in non-sleepy (12,13) and sleepy patients (27). These results could be explained by more symptomatic diseases (aside from sleepiness) in patients with higher AHI. Moreover, smokers and patients with longer ICU stays could have greater comorbidities and worse overall health status, which may increase their awareness of the disease and motivate CPAP compliance.
The adjusted regression model also identified a significant interaction between age and ACS personal history, with a lower risk of non-compliance with CPAP treatment among patients without a personal history of ACS (in younger patients) in comparison to patients with recurrent ACS. These findings could be related to the impact of a new diagnosis of cardiovascular disease, and therefore, adherence to CPAP may have been encouraged by the patients’ recent awareness of increased cardiovascular risk. Furthermore, patients with recurrent ACS showed lower odds of CPAP non-compliance associated with ageing. These patients with recurrent ACS and advanced age may be the ones who have a good complier profile and they could also show better adherence to cardiovascular medications apart from CPAP.
In contrast to the results of Campos-Rodriguez et al. (12), we did not find that hypertension predicted compliance in our sample. This result could be related to the fact that all included patients have suffered an ACS and therefore were aware of their cardiovascular health status regardless of hypertension. In our study, instead of hypertension, ACS could be the main factor in increasing awareness of the importance of treatment.
Strengths and limitations
The main strength of this study is that it provides information on predictors of CPAP compliance in a group of patients in which poor adherence was recently described in the context of important clinical trials. Therefore, identifying predictors of compliance in patients with cardiovascular diseases could be useful in clinical practice and may also be helpful in additional research trials.
Another strength of our study is its multicentre design with a large number of patients. All participating centres performed the same methodology, and the sleep study was performed with the same model of polygraph; furthermore, the study includes the measurement of a broad range of sociodemographic, anthropometric, clinical, pharmacological and cardiovascular variables.
The following limitations should be acknowledged. First, the study was not originally designed to analyse compliance; therefore, some interesting variables, such as side effects, type of interface, humidification, medications and personal or social factors that could influence compliance were not considered. Second, sleepiness was evaluated in our study with ESS, a subjective scale. This scale could have limitations compared to objective methods and has not been validated in women or elders; however, we chose to use ESS because it is a widely used tool to evaluate sleepiness in clinical practice. Third, other OSA-related complaints and symptoms (snoring, fatigue, breathing pauses and choking) were not considered. Fourth, this study was conducted in a health system where CPAP treatment has no cost to the patient; thus, we cannot exclude that cost could be a determinant of compliance in systems where treatment is not free, even though the available data argue against a significant effect of cost on continued use (32).
Conclusions
In non-sleepy patients with ACS and OSA, the main predictors of low risk of non-CPAP compliance were high values of AHI, smoking pack years, and ICU stay length. A previous personal history of ACS was also associated with CPAP non-compliance, although it was dependent on patient age, such that younger patients with recurrent ACS displayed the worst CPAP compliance. This study sheds light on clinical profiles that could benefit from closer follow-up or more personalized visits in order to encourage and increase adherence to CPAP treatment.
Acknowledgements
Funding: This work was supported by ResMed, Ltd. (Australia); Fondo de Investigación Sanitaria (PI10/02763 and PI10/02745); Fondo Europeo de Desarrollo Regional (FEDER); Una manera de hacer Europa; the Spanish Respiratory Society (SEPAR); the Catalonian Cardiology Society, Esteve-Teijin (Spain); Oxigen Salud (Spain); and ALLER. This project has received funding from the European Union’s Seventh Framework Programme for research, technological development and demonstration under grant agreement No. [609396]. It was co-funded by the Ministerio de Economía y Competitividad (COFUND2014-51501). The funders had no role in the study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: The ethics committee of each participating centre approved the study (CEIC-852). Written informed consent was obtained from the patient for publication of this manuscript and any accompanying images.
References
- Peppard PE, Young T, Barnet JH, et al. Increased prevalence of sleep-disordered breathing in adults. Am J Epidemiol 2013;177:1006-14. [Crossref] [PubMed]
- Heinzer R, Vat S, Marques-Vidal P, et al. Prevalence of sleep-disordered breathing in the general population: the HypnoLaus study. Lancet Respir Med 2015;3:310-8. [Crossref] [PubMed]
- Peppard PE, Young T, Palta M, et al. Prospective study of the association between sleep-disordered breathing and hypertension. N Engl J Med 2000;342:1378-84. [Crossref] [PubMed]
- Barbé F, Durán-Cantolla J, Sánchez-de-la-Torre M, et al. Effect of continuous positive airway pressure on the incidence of hypertension and cardiovascular events in nonsleepy patients with obstructive sleep apnea: a randomized controlled trial. JAMA 2012;307:2161-8. [Crossref] [PubMed]
- Sánchez-de-la-Torre M, Campos-Rodriguez F, Barbé F. Obstructive sleep apnoea and cardiovascular disease. Lancet Respir Med 2013;1:61-72. [Crossref] [PubMed]
- Epstein LJ, Kristo D, Strollo PJ Jr, et al. Clinical guideline for the evaluation, management and long-term care of obstructive sleep apnea in adults. J Clin Sleep Med 2009;5:263-76. [PubMed]
- Bratton DJ, Stradling JR, Barbé F, et al. Effect of CPAP on blood pressure in patients with minimally symptomatic obstructive sleep apnoea: a meta-analysis using individual patient data from four randomised controlled trials. Thorax 2014;69:1128-35. [Crossref] [PubMed]
- Martínez-García MA, Campos-Rodríguez F, Catalán-Serra P, et al. Cardiovascular mortality in obstructive sleep apnea in the elderly: role of long-term continuous positive airway pressure treatment: a prospective observational study. Am J Respir Crit Care Med 2012;186:909-16. [Crossref] [PubMed]
- Campos-Rodriguez F, Martinez-Garcia MA, Reyes-Nuñez N, et al. Role of sleep apnea and continuous positive airway pressure therapy in the incidence of stroke or coronary heart disease in women. Am J Respir Crit Care Med 2014;189:1544-50. [Crossref] [PubMed]
- Craig SE, Kohler M, Nicoll D, et al. Continuous positive airway pressure improves sleepiness but not calculated vascular risk in patients with minimally symptomatic obstructive sleep apnoea: the MOSAIC randomised controlled trial. Thorax 2012;67:1090-6. [Crossref] [PubMed]
- Kapur VK, Baldwin CM, Resnick HE, et al. Sleepiness in patients with moderate to severe sleep-disordered breathing. Sleep 2005;28:472-7. [Crossref] [PubMed]
- Campos-Rodriguez F, Martinez-Alonso M, Sanchez-de-la-Torre M, et al. Long-term adherence to continuous positive airway pressure therapy in non-sleepy sleep apnea patients. Sleep Med 2016;17:1-6. [Crossref] [PubMed]
- Gagnadoux F, Le Vaillant M, Paris A, et al. Adherence to positive airway pressure in non-sleepy patients with obstructive sleep apnoea. Eur Respir J 2013;42:863-6. [Crossref] [PubMed]
- Chai-Coetzer CL, Luo YM, Antic NA, et al. Predictors of long-term adherence to continuous positive airway pressure therapy in patients with obstructive sleep apnea and cardiovascular disease in the SAVE study. Sleep 2013;36:1929-37. [Crossref] [PubMed]
- Schiza SE, Simantirakis E, Bouloukaki I, et al. Sleep disordered breathing in patients with acute coronary syndromes. J Clin Sleep Med 2012;8:21-6. [PubMed]
- Antman EM, Anbe DT, Armstrong PW, et al. ACC/AHA guidelines for the management of patients with ST-elevation myocardial infarction--executive summary: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Writing Committee to Revise the 1999 Guidelines for the Management of Patients With Acute Myocardial Infarction). Circulation 2004;110:588-636. [Crossref] [PubMed]
- Esquinas C, Sánchez-de-la Torre M, Aldomá A, et al. Rationale and methodology of the impact of continuous positive airway pressure on patients with ACS and nonsleepy OSA: the ISAACC Trial. Clin Cardiol 2013;36:495-501. [Crossref] [PubMed]
- Deckers JW. Classification of myocardial infarction and unstable angina: a re-assessment. Int J Cardiol 2013;167:2387-90. [Crossref] [PubMed]
- Berry RB, Budhiraja R, Gottlieb DJ, et al. Rules for scoring respiratory events in sleep: update of the 2007 AASM Manual for the Scoring of Sleep and Associated Events. Deliberations of the Sleep Apnea Definitions Task Force of the American Academy of Sleep Medicine. J Clin Sleep Med 2012;8:597-619. [PubMed]
- Chiner E, Arriero JM, Signes-Costa J, et al. Validation of the Spanish version of the Epworth Sleepiness Scale in patients with a sleep apnea syndrome. Arch Bronconeumol 1999;35:422-7. [Crossref] [PubMed]
- Masa JF, Jiménez A, Durán J, et al. Alternative methods of titrating continuous positive airway pressure: a large multicenter study. Am J Respir Crit Care Med 2004;170:1218-24. [Crossref] [PubMed]
- RC Team. R: A language and environment for statistical computing. Vienna, Austria: R Foundation for Statistical Computing, 2016.
- Yang MC, Huang YC, Lan CC, et al. Beneficial Effects of Long-Term CPAP Treatment on Sleep Quality and Blood Pressure in Adherent Subjects With Obstructive Sleep Apnea. Respir Care 2015;60:1810-8. [Crossref] [PubMed]
- Crawford MR, Bartlett DJ, Coughlin SR, et al. The effect of continuous positive airway pressure usage on sleepiness in obstructive sleep apnoea: real effects or expectation of benefit? Thorax 2012;67:920-4. [Crossref] [PubMed]
- Durán J, Esnaola S, Rubio R, et al. Obstructive sleep apnea-hypopnea and related clinical features in a population-based sample of subjects aged 30 to 70 yr. Am J Respir Crit Care Med 2001;163:685-9. [Crossref] [PubMed]
- Roure N, Gomez S, Mediano O, et al. Daytime sleepiness and polysomnography in obstructive sleep apnea patients. Sleep Med 2008;9:727-31. [Crossref] [PubMed]
- Kohler M, Smith D, Tippett V, et al. Predictors of long-term compliance with continuous positive airway pressure. Thorax 2010;65:829-32. [Crossref] [PubMed]
- McArdle N, Devereux G, Heidarnejad H, et al. Long-term use of CPAP therapy for sleep apnea/hypopnea syndrome. Am J Respir Crit Care Med 1999;159:1108-14. [Crossref] [PubMed]
- Kribbs NB, Pack AI, Kline LR, et al. Objective measurement of patterns of nasal CPAP use by patients with obstructive sleep apnea. Am Rev Respir Dis 1993;147:887-95. [Crossref] [PubMed]
- Drake CL, Day R, Hudgel D, et al. Sleep during titration predicts continuous positive airway pressure compliance. Sleep 2003;26:308-11. [Crossref] [PubMed]
- Likar LL, Panciera TM, Erickson AD, et al. Group education sessions and compliance with nasal CPAP therapy. Chest 1997;111:1273-7. [Crossref] [PubMed]
- Rolfe I, Olson LG, Saunders NA. Long-term acceptance of continuous positive airway pressure in obstructive sleep apnea. Am Rev Respir Dis 1991;144:1130-3. [Crossref] [PubMed]