Predictive value of pulmonary function measures for short-term outcomes following lung resection: analysis of a single high-volume institution
Introduction
The importance of preoperative risk-stratification is undisputed, particularly in the setting of major surgery. This information is critical not only for optimal patient selection and preoperative counseling, but also to evaluate outcomes of new minimally invasive alternatives. Yet, consensus on precise measures to identify high-risk patients for pulmonary resection remains elusive.
The predictive value of pulmonary function tests (PFTs) was established in a cohort of patients who underwent resection through a thoracotomy incision. The appeal of such an easy to interpret objective measure by which to risk stratify patients is obvious. However, prior investigation into the ability of pulmonary parameters to forecast poor clinical outcomes has yielded conflicting results. For lung resection there is no clear consensus on how to define “high risk” patients.
Recently, enrollment criteria for several national multicenter trials led by the American College of Surgery Oncology Group (ACOSOG) established forced expiratory volume in 1 second (FEV1) or diffusion capacity for carbon monoxide (DLCO) of ≤50% predicted as major high-risk criteria (1). However, this definition reflects expert opinion rather than empirical data. Due to the high visibility of these trials there is concern that this approach to risk assessment could be widely disseminated and deny patients potentially curative surgery. We sought to investigate the relationship between pulmonary function using the national study cooperative group definition of high risk and short-term postoperative outcomes in a cohort from a single high-volume institution.
Methods
Using our institutional Society of Thoracic Surgeons (STS) database, we identified 419 consecutive patients who underwent pulmonary resection for cancer between 2012 and 2016. We evaluated postoperative 30-day complications and discharge disposition based on patients with predicted FEV1 or DLCO ≤50% as compared to >50% using Pearson’s chi-squared or Fisher’s exact tests. We also performed multivariable logistic regression to assess characteristics associated with postoperative complications and discharge disposition. We chose covariates a priori based on previously identified risk factors (2,3) and performed analyses using SPSS 23 (SPSS, Chicago, IL) with significance level (P=0.05).
Results
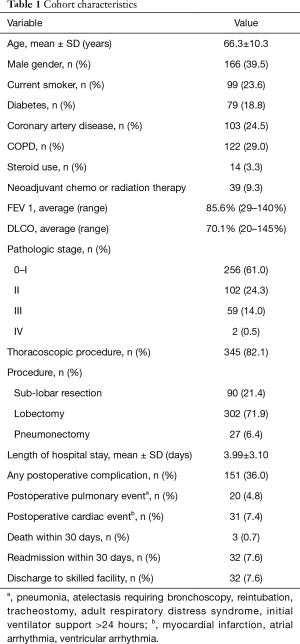
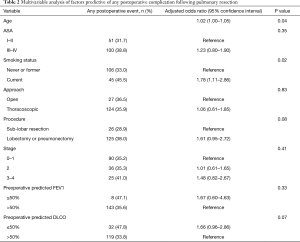
Table 1 describes the cohort characteristics. Overall, 36% of the cohort experienced any postoperative complication. Average FEV1 was 85.6% (range, 29–140%) and average DLCO was 70.1% (range, 20–145%). Average length of stay was 3.99 days. On univariate analysis, DLCO≤50% was associated with any postoperative complication (P=0.03), but not predictive of cardiac events, pulmonary complications, or 30-day readmission. There were no significant differences in any of these short-term outcomes for patients with FEV1 ≤50% as compared to >50% (P>0.05 for all). On multivariable analysis, neither FEV1 nor DLCO ≤50% were significantly associated with occurrence of postoperative complication (OR =1.67, 95% CI: 0.60–4.63; OR =1.66, 95% CI: 0.96–2.86, respectively) (Table 2).
Full table
Full table
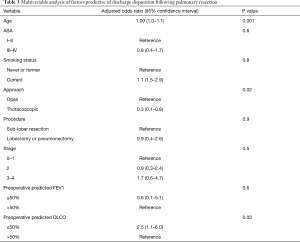
Notably, DLCO ≤50%—but not FEV1—was associated with discharge to a skilled facility on univariate (P=0.01) and multivariable analysis (OR =2.54, 95% CI: 1.08–5.99, P=0.03) (Table 3). Interestingly, when included in adjusted models as continuous variables, FEV1 (OR =0.99; 95% CI: 0.97–0.99, P=0.008) but not DLCO (OR =1.01, 95% CI: 0.99–1.02, P=0.4) was associated with postoperative complications. In contrast, the reverse held for discharge disposition in that there was an association with DLCO and discharge to a skilled facility but not FEV1 (FEV1 OR =1.01; 95% CI: 0.99–1.04, P=0.3; DLCO OR =0.96, 95% CI: 0.93–0.98, P=0.001).
Full table
Discussion
For all-comers presenting to our institution for lung cancer resection, classification based on FEV1 or DLCO ≤50% may not reliably identify those at highest risk for short-term postoperative complications. While more complex risk models exist to guide preoperative decision making (2), the ease of using discrete objective selection criteria is undeniably appealing. Nonetheless, reliance on these values as stand-alone measures for risk stratification may deny patients potentially curative resection.
Previous studies have challenged the prognostic value of pulmonary parameters, postulating that improved pain management and chest wall mechanics following thoracoscopic surgery blunt the sequelae of poor preoperative lung function (3). In our analysis, the significance of FEV1 and DLCO as continuous measures suggests that preoperative pulmonary function may influence postoperative events, even when controlling for operative approach. However, a cutoff value of 50% may be too high to adequately capture this effect. Our findings build upon prior work reporting acceptable early outcomes for stage I patients based on this “high-risk” definition (4,5). Furthermore, our cohort offers a more diverse selection with respect to patient, disease, and operative characteristics.
Notably, we observed a significant association between discharge to a skilled facility and DLCO as either a binary or continuous measure. The need for postoperative skilled nursing care may be more heavily influenced by preoperative factors such as chronic conditions and underlying frailty. A recent competing risks analysis examining cause-specific mortality for patients with early stage non-small cell lung cancer describes an inverse relation between DLCO and 1-year mortality, non-cancer specific mortality and overall survival (6). This finding suggests that DLCO may serve as a valuable proxy for comorbid disease with potential to forecast longer-term outcomes. Further investigation into the relationship between DLCO and other markers of chronic cardiopulmonary conditions and frailty such as a 6-minute walk test is warranted.
Our single-institution study has both strengths and limitations. We deliberately chose to include all-comers during our study period which allowed us to assess the ACOSOG criteria in a more comprehensive group of patients than prior studies. Analysis of this cohort enables us to use our results to caution providers when applying this “high risk” definition broadly in preoperative assessment of patients with lung cancer. However, analyzing a heterogeneous cohort introduces a bias into our study that should be acknowledged when interpreting our findings. Furthermore, this study is underpowered to detect differences in specific outcomes including 30-day mortality and pulmonary complications as well as outcomes in subgroups of patients. Importantly, we operate at a high-volume center for lung volume reduction surgery, affording us extensive experience managing patients with poor pulmonary function that is not available at many facilities.
In conclusion, when applied to a diverse set of patients with lung cancer at a single institution, classification based on FEV1 or DLCO ≤50% may not accurately predict “high risk” patients for short term complications following pulmonary resection. This has significant implications for patient management, as surgical resection remains the gold standard of care and allows for nodal upstaging which is not possible with non-operative management. Our findings advocate for caution when interpreting “high-risk” criteria in isolation and offer an opportunity to better characterize postoperative risk following pulmonary resection.
Acknowledgements
Funding: Dr. Taylor is supported by a training award (T32CA090217) from the National Institutes of Health.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
- Fernando HC, Landreneau RJ, Mandrekar SJ, et al. Thirty- and ninety-day outcomes after sublobar resection with and without brachytherapy for non-small cell lung cancer: results from a multicenter phase III study. J Thorac Cardiovasc Surg 2011;142:1143-51. [Crossref] [PubMed]
- Fernandez FG, Kosinski AS, Burefind W, et al. The Society of Thoracic Surgeons lung cancer resection risk model: higher quality data and superior outcomes. Ann Thorac Surg 2016;102:370-7. [Crossref] [PubMed]
- Berry MF, Villamizar-Ortiz NR, Tong BC, et al. Pulmonary function tests do not predict pulmonary complications after thoracoscopic lobectomy. Ann Thorac Surg 2010;89:1044-51; discussion 1051-2. [Crossref] [PubMed]
- Sancheti MS, Melvan JN, Medbery RL, et al. Outcomes after surgery in high-risk patients with early stage lung cancer. Ann Thorac Surg 2016;101:1043-50. [Crossref] [PubMed]
- Puri V, Crabtree TD, Bell JM, et al. National cooperative group trials of “high-risk” patients with lung cancer: are they truly “high-risk”? Ann Thorac Surg 2014;97:1678-83; discussion 1683-5.
- Eguchi T, Bains S, Lee CM, et al. Impact of increasing age on cause-specific mortality and morbidity in patients with stage I non-small-cell lung cancer: a competing risks analysis. J Clin Oncol 2017;35:281-90. [Crossref] [PubMed]


