Planning and marking for thoracoscopic anatomical segmentectomies
Introduction
While thoracoscopic lobectomy is now recommended in early non-small cell lung cancer (NSCLC) (1), the incidence of thoracoscopic sublobar resections (SLRs) is increasing (2), partly explained by several evolutions: (I) surgeons tend preserving pulmonary function and quality of life, which means sparing lung parenchyma whenever possible; (II) the profile of patients is changing: there are more elderly patients with co-morbidities for whom the invasiveness of the surgical procedure must be minimized (3); and (III) in contrast—we have also more younger patients with small tumors for whom parenchymal resection could be discussed in order to spare parenchyma if a second cancer should develop.
Lobectomy remains the gold standard but is responsible for a morbidity of more than 30%, with an average length of stay superior to 7 days (4). In the 119,000-patient series of the National Cancer Database, average mortality ranged from 2–4% depending on whether or not the resection was extended (5). It should also be noticed that we are seeing more and more young patients with multiple ground glass opacities (GGO) and synchronous or metachronous early stage NSCLC. In these patients who undergo surgery for lung cancer, the likelihood of a second operation in their lifetime is very high. Sparing pulmonary function in these patients is important, that means limiting the first resection to a sublobar one (6).
Among the SLRs, we can distinguish between anatomical segmentectomies which will be discussed in this article and wedge resections which will be deliberately excluded. The oncological results of these limited resections are debated. Several Japanese and a few North American authors have reported identical survival after lobectomy and segmentectomy in stage I NSCLC (7-9). But in a large retrospective cohort of 14,473 patients undergoing stage I NSCLC surgery, there was a significant survival gain in the lobectomy group, regardless of tumour size (10). Some authors stress the survival between lobectomy and segmentectomy is the same when a lymph node dissection is performed during segmentectomy (11). While—up to now—these resections have been performed in patients at high risk because of their age, co-morbidities or compromised respiratory function, the most recent studies seem demonstrating these patients have a reduced survival rate, not linked to the type of surgical resection (segment vs. lobe) but to a higher overall risk of mortality. In addition, patients intentionally treated with an anatomically segmentectomy combined with lymph node dissection even when they could tolerate a lobectomy would have equivalent survival (12).
Evolutions in the radiological features analysis of the nodule would probably help us determining lesion invasiveness and surgical requires (13,14).
Thus, while SLRs are dramatically increasing, the thoracoscopic approach poses specific problems:
- Anatomical variations: since most patients may have anatomical variations, surgeons need to be aware of standard anatomy to perform anatomical segmentectomy without compromising the viability of the remaining segments;
- Absence of manual manipulation of the parenchyma, replaced by instrumental palpation. This leads to difficult—or even impossible—detection of small size tumor, if not immediately subpleural. Similarly, the increasing number of patients undergoing surgery for pure GGO—whose detection is yet difficult when operated by thoracotomy—makes it even more challenging to manage them by thoracoscopy;
- Altered and or reduced spatial landmarks make some surgical tasks difficult, e.g., delimitation of the intersegmental plane.
It is therefore essential to plan the surgical procedure pre-operatively. By planning is meant: modeling (3D reconstruction, 3D printing, 3D virtual navigation) but also any tracking technique associated with pulmonary resection that will make the procedure more accurate, whatever the purpose, i.e., lesion finding and/or the severing the intersegmental plane [CT scan, ultrasounds, fluorescence, electromagnetic navigation bronchoscopy (ENB)..].
Preoperative modelling is also widely used in other surgical specialties (interventional neuroradiology, ENT surgery, orthopedics, etc.) and has shown being helpful by reducing the operating time, reducing the size of incisions, shortening hospitalization stays, minimizing surgical costs, increasing patient safety and participating in the training of junior surgeons (15,16).
Pre-operative modelling
As recently described by Shimizu in a review on thoracic reconstructions (17), “knowledge of anatomy based on 3D reconstruction is essential for a safe and accurate anatomical segmentectomy”. It makes it possible to (I) specify the exact lesion location within a pulmonary segment, (II) define the segmental vascularization and segmental bronchial tree divisions to identify possible anatomical abnormalities that could make the intervention complex or impossible, (III) integrate safety margins to increase the oncological validity of the planned resection, (IV) discuss alternative treatment in compromised patient for whom 3D model predicted a lobectomy for oncological reason.
Pre-operative vascular arterial planning
Already in 2008, some Japanese teams underlined the interest of 3D arterial reconstruction to detect anatomical variations of the pulmonary arteries and try to minimize the risk of bleeding during a thoracoscopic procedure (18). These authors performed 3D reconstructions based on injected CT scan and analyzed the adequacy of their reconstruction by comparing it with the intraoperative findings at surgery. In fact, there are such many pulmonary variations that it is almost impossible to describe a standard anatomy. The development of patient-specific 3D modeling tools therefore plays a key role in this strategy, not only in the preparation of the operation but also as support during the procedure by intraoperatively checking the anatomy on tools like tablets or laptops (19). As early as 2010, the same principles were extended to segmentectomies (20). It should be noted that prior to the introduction of 3D reconstruction, most surgeons limited themselves to simple segmentectomies: lingulectomy (S4-5), superior segment of the lower lobe (S6) or basilar segmentectomy (S7-10). But the realization of more complex segmentectomies such as one or two basilar segments was only made possible—according to the authors—by the precision of the anatomy helping the surgeon throughout the procedure (19,21-24). These reconstructions also minimize vascular risks during dissection by highlighting unusual and important anatomical variations (25).
Pre-operative venous vascular planning
Anatomical segmentectomy requires a thorough evaluation of the intersegmental veins and venous drainage of the segment to be resected. The numerous anatomical variations may necessitate dissection and division of proximal veins (from the hilum of by the fissure after cutting the arteries) and segmental bronchi. The surgeon can also check on the 3D model there is no venous drainage compromising of the adjacent segment after ligation of the segmental vein (26).
Should 3D reconstruction be done by the surgeon? Against this option are the important time devoted to image analysis, the insufficient competency in radiology and the need for absolute reliability of the 3D model compared to reality. However, with the progresses of softwares and more and more intuitive tools, these objections will vanish (27). For this option are: the surgeon’s independence from an external provider (whether it is a radiologist or an external company) and the possibility of carrying out this type of planning for any thoracic surgery.
Preoperative planning of the resection margin with confirmation of the segment(s) to be resected
Compared to lobectomies, SLRs have an increased risk of local recurrence, as resection margins tend to be closer to the tumor (28). Schuchert et al. clearly demonstrated that for patients with a Stage I NSCLC, the results of SLR were comparable to lobectomy, but recurrence was more frequent in patients with a margin/tumor ratio of less than 1 (8).
The distance between the lesion and the staple line of the intersegmental plane is therefore a crucial factor. Some authors have described the use of software to extrapolate a safety margin based on tumor diameter (29,30). On our 3D models, we have developed a virtual safety margin, which sometimes leads to extend the resection to the adjacent segment if the intersegmental parenchymal resection plan seems compromised. This does not preclude the need for an intraoperative frozen section analysis of the staple line in the event of a lesion that appears to be close (31).
Tracking
Preoperative marking of nodules
More and more patients are now being referred to the surgeon with a suspicion of pT1a NSCLC or with GGOs. One of the features of these newly encountered lesions is the difficult detection because of their size and their inconsistency at palpation, particularly for GGO. Different criteria have been established that should lead to use a preoperative localization method, especially lesion size and distance to the pleura (25,32). Several techniques have been described (33) and recently listed again (24,34) to assist in locating peripheral lung lesions: preoperative injection of dyes (methylene blue, lipiodol, colored collagen etc.), metallic markers placement within the lesion (coils, fiduciaries) or hooks, transthoracic or thoracoscopic ultrasound with all their advantages and limitations. Particularly noteworthy are the risks of pneumothorax for CT-guided procedures, the migration of hooks or coils, logistical difficulties related to the transfer of the patient from the radiology room to the operating theatre. These potential complications could be reduced by performing these techniques immediately before surgery in the operating room thanks to hybrid rooms equipped with C-arm CT (35,36) or ENB (37).
Marking of the intersegmental plane
With closed chest surgery, the plane determined by the intersegmental vein—the standard thoracotomy landmark—is much more difficult to identify. The delimitation of the plane according to other techniques is therefore necessary and has been described as follows:
- Realization of an inflation-deflation zone once the bronchus has been cut or by direct catheterization of the segmental bronchial stump is the most frequently used method. Tsubota published a slightly different method with inflation of the whole lung, then stapling or ligation of the segmental bronchus, thus trapping air within the segment to be resected (38). Okada et al. proposed the use of jet ventilation positioned under elective flexible fibroscopy in the segment to be resected (39), or segmented bronchial ligation with a slip knot method after inflation (40).
- Other methods using thermography or a combination of indocyanine green (ICG) and infrared thoracoscopy have been developed (17). After segmental artery ligation, intra-systemic injection of indocyanine creates a fluorescent boundary between viable (fluorescent) and devascularized segments (41,42).
- Endobronchial marking by dye injection either under bronchoscopy or assisted by ENB. Conversely, an injection of ICG into the segmental bronchus followed by ligation of the segmental vein creates a demarcation line by maintaining the targeted colored segment. The transbronchial injection of ICG allows the identification of the intersegmental plane not only at the surface but also in the pulmonary parenchyma (43).
- Virtual assisted pulmonary mapping (VAL-MAP), a technique developed by M. Sato et al allows simultaneous marking of the pulmonary lesion to be removed and resection lines on 3D images with an endobronchial dye injected via bronchoscopy (44).
3D printing and augmented reality
3D printing is also experiencing significant interest despite still high costs. In a meta-analysis of more than 600 articles, the benefit of simulation on the theoretical and practical learning of health professionals was clearly demonstrated (45). Three-D printing makes it possible to use a model either for teaching and training in thoracic surgery (46) or for the realization of patient-specific prototypes allowing simulating the intervention beforehand. Its applications in surgery have been highlighted in recent articles (16,27).
Finally, the interactive augmented reality (RAI) initially developed for liver surgery is slowly progressing (34,47). This appealing technique comes up against unresolved technical constraints in thoracic surgery to date, i.e., deflation of the lung. It should make it possible to superimpose the 3D reconstruction on the intraoperative view, which could help the surgeon in the detection of non-palpable nodule or GGO. Unfortunately, the deformities and displacements of the organs make it for the moment inaccurate in thoracic surgery (48).
In practice, what is the planning of an anatomical segmentectomy in our institution?
Our detailed technique was recently published (49).
3D-modelization
Currently, we use an online service that allows 3D modeling of CT-scans from medical DICOM images (Visible Patient™, Strasbourg, France). The images are anonymized and then uploaded by the surgeon to a secure and dedicated web portal. A 3D model based on these images is created according to the surgeon’s needs. The 3D model can be used via a dedicated software (Virtual Planning™) and manipulated in any direction whatever the support (laptop, tablet or smartphone). All surgical team members have direct access to the images on their own phone or tablet so that the strategy and technical issues can be discussed together. The software not only allows one studying the main anatomical landmarks in their entirety or separately, but also provides the following useful functions:
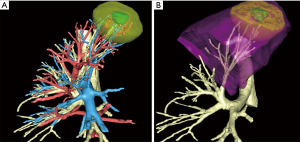
- Evaluating segmental arteries, veins and bronchi preoperatively (Figure 1A);
- Simulating virtual resections by clicking on a selected bronchus (Figure 1B);
- Calculating the volumetry of segments;
- Calculating and simulating a safety margin, twice as large as the diameter of the tumor, which is represented by a yellow halo around the tumor. This helps visualize whether the planned segmentectomy will match a safety margin, or whether a larger resection should be preferred;
- Confirming the exact location of the target lesion;
- Identifying the intersegmental plan.
Tracking techniques
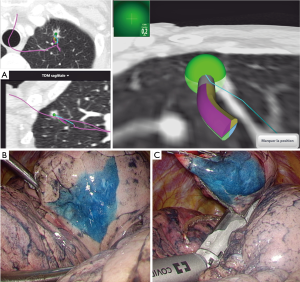
When dealing with a GGO or a deeply located nodule; we usually track the lesion with blue dye injection under ENB guidance before surgery once anesthesia has been inducted. According to our pre-operative ENB planning, we navigate to the lesion that is overtaken to reach the pleura. One mL of blue dye is injected after purging the navigation catheter. Thoracoscopy is then performed. This colored mark allows us to perform either a needle biopsy or a wedge resection. The specimen is then sent for frozen section examination (Figure 2).
Our initial method for demarcation of the intersegmental plan was the use of an inflation/deflation line after reventilation of the whole lung once the segmented bronchus had been severed.
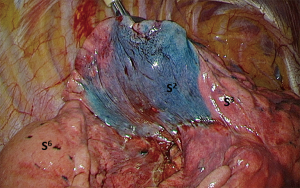
Since then, we added coloration of the entire segment to be resected under simple bronchoscopy or ENB guidance. In practice: all the segmental bronchi of the segment to be resected are catheterized under bronchoscopy and occluded with a balloon to avoid dye backflow. We then inject 1 mL of a blue dye followed by 20 cc of air.
These two endobronchial staining techniques are preferably carried out for “complex” segmentectomies, especially basilar segments and some upper lobes segments (Figure 3).
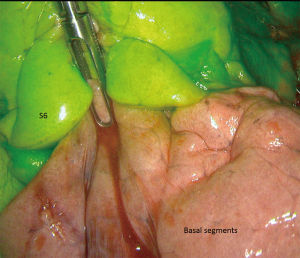
We are currently evaluating ICG systemic injection (at the dose of 0.2 mL/kg, possibly renewed once) after division of segmental bronchovascular elements. Given the ease of use of this method, we feel it could be extended to any segmentectomy including “simple” ones (Figure 4).
Conclusions
Performing a SLR by thoracoscopy means confirming the location of the lesion, identifying vascular and bronchial structures, preserving venous drainage of adjacent segments, separating the intersegmental plane and ensuring an oncological safety margin. Preoperative modelling and tracking not only ease these complex procedures but should now be considered as part of the operation. We are most likely only at the onset of these technical developments which, hopefully, will allow us to perform a more and more accurate surgery.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Informed Consent: The authors confirm that written informed consent was obtained from the patient for publication of this surgical technique and any accompanying images.
References
- Howington JA, Blum MG, Chang AC, et al. Treatment of stage I and II non-small cell lung cancer: Diagnosis and management of lung cancer, 3rd ed: American College of Chest Physicians evidence-based clinical practice guidelines. Chest 2013;143:e278S-e313S.
- Cao C, D'Amico T, Demmy T, et al. Less is more: a shift in the surgical approach to non-small-cell lung cancer. Lancet Respir Med 2016;4:e11-12. [Crossref] [PubMed]
- Traibi A, Grigoroiu M, Boulitrop C, et al. Predictive factors for complications of anatomical pulmonary segmentectomies. Interact Cardiovasc Thorac Surg 2013;17:838-44. [Crossref] [PubMed]
- Paul S, Altorki NK, Sheng S, et al. Thoracoscopic lobectomy is associated with lower morbidity than open lobectomy: a propensity-matched analysis from the STS database. J Thorac Cardiovasc Surg 2010;139:366-78. [Crossref] [PubMed]
- Rosen JE, Hancock JG, Kim AW, et al. Predictors of mortality after surgical management of lung cancer in the National Cancer Database. Ann Thorac Surg 2014;98:1953-60. [Crossref] [PubMed]
- Gu C, Wang R, Pan X, et al. Sublobar resection versus lobectomy in patients aged</=35 years with stage IA non-small cell lung cancer: a SEER database analysis. J Cancer Res Clin Oncol 2017;143:2375-82. [Crossref] [PubMed]
- Wisnivesky JP, Henschke CI, Swanson S, et al. Limited resection for the treatment of patients with stage IA lung cancer. Ann Surg 2010;251:550-4. [Crossref] [PubMed]
- Schuchert MJ, Abbas G, Awais O, et al. Anatomic segmentectomy for the solitary pulmonary nodule and early-stage lung cancer. Ann Thorac Surg 2012;93:1780-5; discussion 1786-7.
- Okada M, Tsutani Y, Ikeda T, et al. Radical hybrid video-assisted thoracic segmentectomy: long-term results of minimally invasive anatomical sublobar resection for treating lung cancer. Interact Cardiovasc Thorac Surg 2012;14:5-11. [Crossref] [PubMed]
- Whitson BA, Groth SS, Andrade RS, et al. Survival after lobectomy versus segmentectomy for stage I non-small cell lung cancer: a population-based analysis. Ann Thorac Surg 2011;92:1943-50. [Crossref] [PubMed]
- Wolf AS, Richards WG, Jaklitsch MT, et al. Lobectomy versus sublobar resection for small (2 cm or less) non-small cell lung cancers. Ann Thorac Surg 2011;92:1819-23; discussion 1824-5.
- Kodama K, Higashiyama M, Okami J, et al. Oncologic outcomes of segmentectomy versus lobectomy for clinical T1a N0 M0 non-small cell lung cancer. Ann Thorac Surg 2016;101:504-11. [Crossref] [PubMed]
- Saji H, Matsubayashi J, Akata S, et al. Correlation between whole tumor size and solid component size on high-resolution computed tomography in the prediction of the degree of pathologic malignancy and the prognostic outcome in primary lung adenocarcinoma. Acta Radiol 2015;56:1187-95. [Crossref] [PubMed]
- Zhou J, Li Y, Zhang Y, et al. Solitary ground-glass opacity nodules of stage IA pulmonary adenocarcinoma: combination of 18F-FDG PET/CT and high-resolution computed tomography features to predict invasive adenocarcinoma. Oncotarget 2017;8:23312-21. [PubMed]
- Tetsworth K, Block S, Glatt V. Putting 3D modelling and 3D printing into practice: virtual surgery and preoperative planning to reconstruct complex post-traumatic skeletal deformities and defects. SICOT J 2017;3:16. [Crossref] [PubMed]
- Hoang D, Perrault D, Stevanovic M, et al. Surgical applications of three-dimensional printing: a review of the current literature & how to get started. Ann Transl Med 2016;4:456. [Crossref] [PubMed]
- Shimizu K, Nakazawa S, Nagashima T, et al. 3D-CT anatomy for VATS segmentectomy. J Vis Surg 2017;3:88. [Crossref] [PubMed]
- Fukuhara K, Akashi A, Nakane S, et al. Preoperative assessment of the pulmonary artery by three-dimensional computed tomography before video-assisted thoracic surgery lobectomy. Eur J Cardiothorac Surg 2008;34:875-77. [Crossref] [PubMed]
- Eguchi T, Takasuna K, Kitazawa A, et al. Three-dimensional imaging navigation during a lung segmentectomy using an iPad. Eur J Cardiothorac Surg 2012;41:893-7. [Crossref] [PubMed]
- Oizumi H, Endoh M, Takeda S, et al. Anatomical lung segmentectomy simulated by computed tomographic angiography. Ann Thorac Surg 2010;90:1382-3. [Crossref] [PubMed]
- Hagiwara M, Shimada Y, Kato Y, et al. High-quality 3-dimensional image simulation for pulmonary lobectomy and segmentectomy: results of preoperative assessment of pulmonary vessels and short-term surgical outcomes in consecutive patients undergoing video-assisted thoracic surgery. Eur J Cardiothorac Surg 2014;46:e120-126. [Crossref] [PubMed]
- Chen-Yoshikawa TF, Date H. Update on three-dimensional image reconstruction for preoperative simulation in thoracic surgery. J Thorac Dis 2016;8:S295-301. [PubMed]
- Nakamoto K, Omori K, Nezu K. Superselective segmentectomy for deep and small pulmonary nodules under the guidance of three-dimensional reconstructed computed tomographic angiography. Ann Thorac Surg 2010;89:877-83. [Crossref] [PubMed]
- Yang Q, Xie B, Hu M, et al. Thoracoscopic anatomic pulmonary segmentectomy: a 3-dimensional guided imaging system for lung operations. Interact Cardiovasc Thorac Surg 2016;23:183-9. [Crossref] [PubMed]
- Nakashima S, Watanabe A, Obama T, et al. Need for preoperative computed tomography-guided localization in video-assisted thoracoscopic surgery pulmonary resections of metastatic pulmonary nodules. Ann Thorac Surg 2010;89:212-8. [Crossref] [PubMed]
- Gossot D, Seguin-Givelet A. Anatomical variations and pitfalls to know during thoracoscopic segmentectomies. J Thorac Dis 2017. [Epub ahead of print].
- Sato T, Date H. 3D CT simulation in minimally invasive thoracic surgery. J Vis Surg 2017;3:26. [Crossref] [PubMed]
- Moon Y, Lee KY, Moon SW, et al. Sublobar Resection Margin Width Does Not Affect Recurrence of Clinical N0 Non-small Cell Lung Cancer Presenting as GGO-Predominant Nodule of 3 cm or Less. World J Surg 2017;41:472-9. [Crossref] [PubMed]
- Ueda K, Tanaka T, Hayashi M, et al. What proportion of lung cancers can be operated by segmentectomy? A computed-tomography-based simulation. Eur J Cardiothorac Surg 2012;41:341-5. [Crossref] [PubMed]
- Iwano S, Yokoi K, Taniguchi T, et al. Planning of segmentectomy using three-dimensional computed tomography angiography with a virtual safety margin: technique and initial experience. Lung Cancer 2013;81:410-5. [Crossref] [PubMed]
- Gossot D, Lutz JA, Grigoroiu M, et al. Unplanned Procedures During Thoracoscopic Segmentectomies. Ann Thorac Surg 2017;104:1710-7. [Crossref] [PubMed]
- Suzuki K, Shimohira M, Hashizume T, et al. Usefulness of CT-guided hookwire marking before video-assisted thoracoscopic surgery for small pulmonary lesions. J Med Imaging Radiat Oncol 2014;58:657-62. [Crossref] [PubMed]
- Zaman M, Bilal H, Woo CY, et al. In patients undergoing video-assisted thoracoscopic surgery excision, what is the best way to locate a subcentimetre solitary pulmonary nodule in order to achieve successful excision? Interact Cardiovasc Thorac Surg 2012;15:266-72. [Crossref] [PubMed]
- Heuts S, Sardari Nia P, Maessen JG. Preoperative planning of thoracic surgery with use of three-dimensional reconstruction, rapid prototyping, simulation and virtual navigation. J Vis Surg 2016;2:77. [Crossref] [PubMed]
- Rouzé S, de Latour B, Flécher E, et al. Small pulmonary nodule localization with cone beam computed tomography during video-assisted thoracic surgery: a feasibility study. Interact Cardiovasc Thorac Surg 2016;22:705-11. [Crossref] [PubMed]
- Gill RR, Zheng Y, Barlow JS, et al. Image-guided video assisted thoracoscopic surgery (iVATS) - phase I-II clinical trial. J Surg Oncol 2015;112:18-25. [Crossref] [PubMed]
- Muñoz-Largacha JA, Ebright MI, Litle VR, et al. Electromagnetic navigational bronchoscopy with dye marking for identification of small peripheral lung nodules during minimally invasive surgical resection. J Thorac Dis 2017;9:802-8. [Crossref] [PubMed]
- Tsubota N. An improved method for distinguishing the intersegmental plane of the lung. Surg Today 2000;30:963-4. [Crossref] [PubMed]
- Okada M, Mimura T, Ikegaki J, et al. A novel video-assisted anatomic segmentectomy technique: selective segmental inflation via bronchofiberoptic jet followed by cautery cutting. J Thorac Cardiovasc Surg 2007;133:753-8. [Crossref] [PubMed]
- Endoh M, Oizumi H, Kato H, et al. How to demarcate intersegmental plane with resected-segments inflation method using the slip knot technique in thoracoscopic anatomic segmentectomy. J Vis Surg 2017;3:100. [Crossref] [PubMed]
- Misaki N, Chang SS, Igai H, et al. New clinically applicable method for visualizing adjacent lung segments using an infrared thoracoscopy system. J Thorac Cardiovasc Surg 2010;140:752-6. [Crossref] [PubMed]
- Tarumi S, Misaki N, Kasai Y, et al. Clinical trial of video-assisted thoracoscopic segmentectomy using infrared thoracoscopy with indocyanine green. Eur J Cardiothorac Surg 2014;46:112-5. [Crossref] [PubMed]
- Sekine Y, Ko E, Oishi H, et al. A simple and effective technique for identification of intersegmental planes by infrared thoracoscopy after transbronchial injection of indocyanine green. J Thorac Cardiovasc Surg 2012;143:1330-5. [Crossref] [PubMed]
- Sato M, Kuwata T, Yamanashi K, et al. Safety and reproducibility of virtual-assisted lung mapping: a multicentre study in Japan. Eur J Cardiothorac Surg 2017;51:861-8. [PubMed]
- Cook DA, Hatala R, Brydges R, et al. Technology-enhanced simulation for health professions education: a systematic review and meta-analysis. JAMA 2011;306:978-88. [Crossref] [PubMed]
- Morikawa T, Yamashita M, Odaka M, et al. A step-by-step development of real-size chest model for simulation of thoracoscopic surgery. Interact Cardiovasc Thorac Surg 2017;25:173-6. [Crossref] [PubMed]
- Chen L, Tang W, John NW. Real-time geometry-aware augmented reality in minimally invasive surgery. Healthc Technol Lett 2017;4:163-7. [Crossref] [PubMed]
- Kanzaki M, Isaka T, Kikkawa T, et al. Binocular stereo-navigation for three-dimensional thoracoscopic lung resection. BMC Surg 2015;15:56. [Crossref] [PubMed]
- Gossot D, Lutz J, Grigoroiu M, et al. Thoracoscopic anatomic segmentectomies for lung cancer: technical aspects. J Vis Surg 2016;2:171. [Crossref] [PubMed]


