The feasibility and efficacy of two micro-portal video-assisted thoracic surgery in pulmonary lobectomy for lung cancer
Introduction
Malignant tumors which lead to lung cancer, is the leading contributor to the high mortality and morbidity in China (1). Although chemotherapy, radiotherapy, specific immunotherapy and molecular target therapy have had great progress in the past decades, radical surgery is still the primary option for relatively early stage of lung cancer. With the advent of rapid technological progress and constant renewal of ideas, thoracic surgery is becoming more and more minimally invasive.
Conventional three-port video-assisted thoracic surgery (VATS) has thought as the main standard technique in treating lung cancer for many units around the world (2). Many studies have demonstrated that VATS lobectomy showed decreased hospitalization time, less loss of respiratory function, shorter chest tube drainage time and fewer postoperative complications (3). Besides, four-port (4) and two-port (5) VATS are also reported by several centers. Since 2010, uniportal VATS which is firstly performed by Gonzalez et al (6,7) through a small incision of about 4 cm has expanded an area of exploration in minimally invasive approach. A recent meta-analysis revealed better outcomes of uniportal VATS regarding to postoperative hospitalization, chest tube drainage time and pain score (8).
The aim of this study is to optimize two important aspects of VATS, that are cutting length and technique procedure. The most currently popular and matured option for incision selection of VATS include traditional three port and uniportal methods. The three-port procedure is not restricted by the operation angle, the resection is more convenient and the operation speed is faster. Single port procedure is operated through a single intercostal, which has beautiful incision, less pain (9). However, because of the limited internal angle, the operation is more complex, the operation speed is slow. Therefore, we designed a new procedure named two micro-portal VATS technique which shortened the length of viewing port and secondary operating port. So, the wound becomes smaller both outside and inside. In order to make the operation more reasonable and simpler, we designed different surgical procedures of hilar dissection for different lobectomy, which we called tangent line single-direct VATS lobectomy. There is no need to turn over the lobes too many times during the operation, which avoiding tumor cells entering the circulatory system as a result of turning and squeezing the lungs.
Methods
Patients
We retrospectively analyzed clinical outcomes of 208 patients who underwent VATS lobectomy by a single surgeon at our department. All patients were pathologically diagnosed as primary lung cancer postoperatively. One hundred and four patients achieved traditional three port VATS lobectomy between June 2016 and December 2016. From January 2017, we started the two micro-portal VATS lobectomy, and 104 patients were analyzed until December 2017. None patient received neoadjuvant therapy before surgery. Institutional review approval and written informed consent for each patient were obtained. Routine preoperative research including complete blood count, serum biochemistry tests, arterial blood gas analysis, pulmonary function test, transthoracic echocardiogram, abdominal ultrasonography, computed tomographic scan of the chest and brain, whole-body bone scanning and bronchoscopy were undergoing before every surgery.
The characteristic data studied in each patient include age, body mass index (BMI), gender, smoking status, tumor location, tumor maximum diameter, histologic type, lymph node staging, tumor staging, operative time (from the beginning of general anaesthetic to extubation), blood loss, number of lymph node retrieved, pain score on day 1, 2, 3, 7 after surgery, duration of chest tube in place, length of hospital stay and postoperative complications regarding to pulmonary infection, gas leak over 5 days and death. Patients who were older than 75 or younger than 18 were excluded. The tumor stage was determined according to the seventh edition of American Joint Committee on Cancer staging system.
Operative procedure and postoperative care
All patients were delivered general anesthesia with two lumen tube intubation and operated in a lateral decubitus position with the operating table flexed to increase the intercostal space.
For traditional three port VATS lobectomy, two 10–20 mm incisions were made in the seventh intercostal space at the midaxillary line and the ninth intercostal space at the scapular line, as viewing port and secondary operating port separately. Then, a main operating port was made on the third or fourth intercostal space of the anterior axillary line, with a length of 20–25 mm incision that was attaching with a wound protector. After that, a 10 mm 30° oblique-viewing thoracoscope was used in the operation. The dissections were mainly done using endoscopic ultrasonic scalpel and occasionally using endoscopic hook electrocautery. The resection was performed using flexible curved-tip endoscopic staplers in the order of pulmonary vein, lobar bronchus, pulmonary artery and interlobar fissure. This procedure was learned from Prof. Liu, that he called single-direction thoracoscopic lobectomy (10). Subsequently, the resected lobe was removed using a protective specimen bag through the main operating hole. Afterwards, systemic mediastinal lymph node dissection was performed in a standard way, which included group 2, 3, 4, 7, 9 lymph nodes in the right side and group 5, 6, 7, 9 lymph nodes in the left side. Finally, a 10-mm rubber chest tube was placed through the secondary operating port for the middle and lower lobectomy. For upper lobectomy, another tube was placed through the viewing port to the cupula pleurae. At the end of operation, the tube was secured with a U-shaped suture to allow proper closure of the wound when removing the tube.
For two micro-portal VATS lobectomy, the viewing port and secondary operating port were replaced by a 5-mm trocar comparing with traditional three-port VATS lobectomy (Figure 1). Accordingly, the new procedure was performed using a 5-mm 30° oblique-viewing thoracoscope. The resection order was followed by the principle of “from back down to front up” way. In detail, for upper lobectomy (Figure 2), the resection was made in the order of pulmonary artery and its branches, lobar bronchus and pulmonary vein. For right middle lobectomy, we chose the main operating port in the third intercostal space of the anterior axillary line and followed the resectional order of vein-bronchus-artery. For lower lobectomy, the resection was performed in the order of vein-bronchus-artery or artery-bronchus-vein. In the end, the chest tube was changed to thinner and softer latex material.
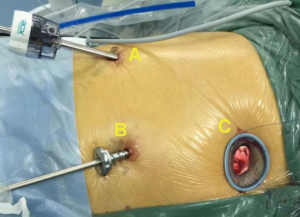
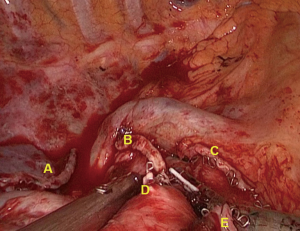
All patients were observed in our intense care unit for six hours at least. The same dose of disposable self-control PCA (sulfentanyl and dezocine) and prophylactic antibiotics were used routinely. The intensity of postoperative pain was scored with a 0–10 numerical analogue scale. Pain scores were assessed at 24, 48, and 72 h after surgery and at 1 week postoperatively. The chest drain was removed when the amount of chest draining was less than 100 mL per day, without air leakage, no pneumothorax or localized pleural effusion on chest X-rays. Patients were usually discharged from the hospital 1 day after the chest tube was removed.
Ethical approval was not required by the Tangdu Hospital. The procedure had standard indications and oncological principles. Meanwhile, all patients were signed written informed consent and not involved in a formal trial.
Statistical analysis
A database was obtained from online service of Linkdoc. Continuous variables were presented as means with standard deviations and categorical variables were presented as frequencies. Variables were compared using Student’s t-test, the χ2 test and variance analysis or Fisher’s exact test. Data were analyzed using SPSS 22.0 software (SPSS, Inc., Chicago, IL, USA), p-value less than 0.05 was considered statistically significant.
Results
There were 208 patients who underwent VATS lobectomy between June 2016 and December 2017 by Prof. Yan at our department. One hundred and four patients underwent traditional three port VATS and 104 patients underwent two micro-portal VATS lobectomy. All patients were confirmed to be primary lung cancer in postoperative pathological diagnoses and classified by pathologic stage. As demonstrated in Table 1, the two groups were well matched in regarding to age, body weight index (BMI), gender, smoking status, tumor site, TNM stage and histological type.
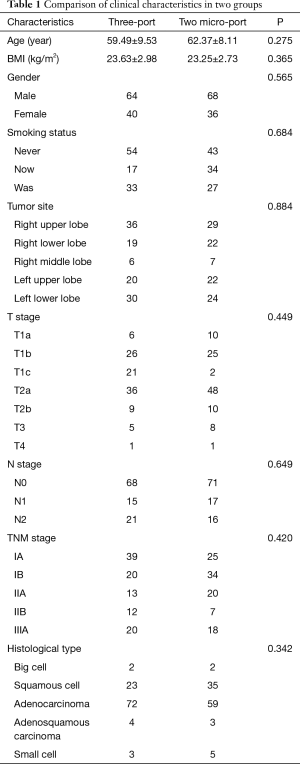
Full table
In terms of surgical outcomes, as shown in Table 2, there was no intraoperative or 30-day mortality in both groups. In the traditional three port group, there were 6 patients who needed conversion to thoracotomy, due to severe pleural adhesion (n=2), bleeding at pulmonary arterial branch (n=1), and anthracofibrotic lymph nodes around pulmonary artery (n=3). Similarly, there were 7 patients who converted to thoracotomy, due to severe pleural adhesion (n=1), bleeding at pulmonary arterial branch (n=2), and anthracofibrotic lymph nodes around pulmonary artery (n=3). The operative time and intraoperative bleeding in the traditional three port group and two micro-port group were similar (145.05±4.18 vs. 149.76±35.96 min, P=0.601; 202.60±224.30 vs. 275.38±444.96 mL, P=0.141). The number of lymph node retrieved in the traditional three port group and two micro-port group were 15.36±3.06 and 16.32±3.47, respectively (P=0.116). Postoperative air leak over 5 days was developed in 10 patients in the traditional three port group and 13 patients in the two micro-port group (P=0.659). The rate of pulmonary infection after lobectomy was also similar in either group (4/104 vs. 5/104, P=0.749). The chest tube duration and postoperative hospital stay in the two micro-port group were shorter than that in the traditional three port group (5.10±3.59 vs. 5.63±2.65 days, P=0.659; 7.63±4.72 vs. 8.32±3.43 days, P=0.989). However, the differences were not statistically significant.
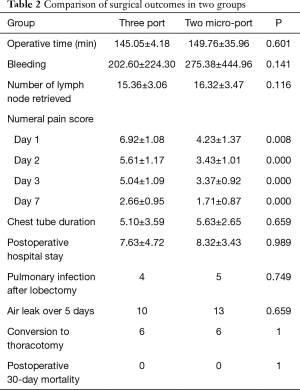
Full table
In the traditional three port VATS group, the mean pain scores at day 1, 2, 3 and 7 after surgery were 6.92±1.08, 5.61±1.17, 5.04±1.09 and 2.66±0.95. In the two micro-port VATS group, the pain scores were 4.23±1.37, 3.43±1.01, 3.37±0.92 and 1.71±0.87, which were significantly lower than the traditional three port VATS group. The result was confirmed an advantage for our two micro-port VATS regarding to reduced pain in the early postoperative period. We attributed this to reduced trauma and intercostal nerve injury.
Discussion
VATS lobectomy was widely practiced using 1 to 4 port incisions in China. The most representative procedure includes the single-direction thoracoscopic lobectomy of West China Hospital (10) and the single port thoracoscopic lobectomy of Shanghai Pulmonary Hospital (11). Several studies demonstrated that uniportal VATS minimized the amount of surgical trauma and had many advantages including decreased postoperative pain, shorter hospital stay, preservation of pulmonary function and so forth when compared with traditional three port VATS. Wang et al. (12) noted that uniportal VATS was geometrically favorable to multiportal techniques, which provided more anatomic and direct in-plane view of the target tissue, and made it easier for the surgeon to judge distances and improve the accuracy of the surgical maneuvers. However, due to its technical difficulty such as instrumentation impeding with each other and exclusive need of endoscopic instruments with two articulation, uniportal VATS was performed only in a limited number of hospitals. We considered that our method made best use of the advantages of traditional three port VATS and bypassed the disadvantages of uniportal VATS.
The result of this study revealed that there were no surgery related mortality and no significant differences in operative time, blood loss, number of lymph nodes dissected, chest tube duration, length of stay or postoperative complications between the two group. These results indicated that the safety and efficacy of two micro-port and three port VATS were similar. Moreover, our results showed that the numeral pain scores were significantly lower in the two micro-portal VATS group, which might be attributed to less incision trauma and reduced intercostal nerve injury. The total length of incision was 3–3.5 cm which was even shorter than the single port. Meanwhile, the use of 5 mm trocar and wound protector successfully avoided the injury to the chest wall from the instruments. A recent study by Nardini et al (13) also demonstrate that microlobectomy using 5 mm trocar and instrument is a valid alternative to traditional VATS techniques.
This study has several limitations. First of all, the retrospective nature of data lends themselves to inherent bias, even though the patients have received the standardized perioperative care. Secondly, we didn’t compare the intraoperative and postoperative outcomes of a specific lobectomy between the two groups due to limited sample size. In addition, the study had no long-term follow-up data on oncologic factors such as tumor recurrence rate and overall survival rate. This will be our further work to be on going.
Conclusions
In conclusion, our experience showed that the two micro-port VATS lobectomy had similar results in safety and efficacy comparing with the traditional three-port VATS lobectomy. Besides, patients who had two micro-port VATS lobectomy suffered from less pain, which might improve their satisfaction rate. This indicates that the two micro-port VATS remain an exciting new development in the field of minimally invasive thoracic surgery.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: Ethical approval was not required by the Tangdu Hospital. Meanwhile, all patients were signed written informed consent and not involved in a formal trial.
References
- Chen W, Zheng R, Baade PD, et al. Cancer statistics in China, 2015. CA Cancer J Clin 2016;66:115-32. [Crossref] [PubMed]
- Bendixen M, Jørgensen OD, Kronborg C, et al. Postoperative pain and quality of life after lobectomy via video-assisted thoracoscopic surgery or anterolateral thoracotomy for early stage lung cancer: a randomised controlled trial. Lancet Oncol 2016;17:836-44. [Crossref] [PubMed]
- D'Andrilli A, Rendina EA. Enhanced recovery after surgery (ERAS) and fast-track in video-assisted thoracic surgery (VATS) lobectomy: preoperative optimisation and care-plans. J Vis Surg 2018;4:4. [Crossref] [PubMed]
- Borro JM, Regueiro F, Pértega S, et al. Comparative Study of Survival following Videothoracoscopic Lobectomy Procedures for Lung Cancer: Single- versus Multiple-port Approaches. Arch Bronconeumol 2017;53:199-205. [PubMed]
- Wang GS, Wang Z, Wang J, et al. Biportal complete video-assisted thoracoscopic lobectomy and systematic lymphadenectomy. J Thorac Dis 2013;5:875-81. [PubMed]
- Gonzalez D, de la Torre M, Paradela M, et al. Video-assisted thoracic surgery lobectomy: 3-year initial experience with 200 cases. Eur J Cardiothorac Surg 2011;40:e21-8. [Crossref] [PubMed]
- Gonzalez-Rivas D. VATS lobectomy: surgical evolution from conventional VATS to uniportal approach. ScientificWorldJournal 2012;2012. [Crossref] [PubMed]
- Harris CG, James RS, Tian DH, et al. Systematic review and meta-analysis of uniportal versus multiportal video-assisted thoracoscopic lobectomy for lung cancer. Ann Cardiothorac Surg 2016;5:76-84. [Crossref] [PubMed]
- Fan J, Yao J, Wang Q, et al. Safety and feasibility of uniportal video-assisted thoracoscopic surgery for locally advanced non-small cell lung cancer. J Thorac Dis 2016;8:3543-50. [Crossref] [PubMed]
- Liu L, Che G, Pu Q, et al. A new concept of endoscopic lung cancer resection: Single-direction thoracoscopic lobectomy. Surg Oncol 2010;19:e71-7. [Crossref] [PubMed]
- Xie D, Wang H, Fei K, et al. Single-port video-assisted thoracic surgery in 1063 cases: a single-institution experience. Eur J Cardiothorac Surg 2016;49 Suppl 1:i31-6. [Crossref] [PubMed]
- Wang L, Liu D, Lu J, et al. The feasibility and advantage of uniportal video-assisted thoracoscopic surgery (VATS) in pulmonary lobectomy. BMC Cancer 2017;17:75. [Crossref] [PubMed]
- Nardini M, Bilancia R, Solli P, et al. Microlobectomy: completely portal pulmonary lobectomy. J Vis Surg 2018;4:153. [Crossref]


