Clinical manifestations, diagnosis, and treatment of ischemic mitral regurgitation: a review
Clinical context
Ischemic mitral regurgitation (IMR) is a subtype of secondary MR, which is caused as a complication of ischemic heart disease. Valvular involvement can be primary (organic) or secondary (functional). Primary IMR happens after the rupture of the mitral subvalvular apparatus in the context of an acute myocardial infarction (AMI). Secondary IMR occurs when the valve leaflets and chordae are structurally normal and MR results from an imbalance between closing and tethering forces on the mitral valve (MV) secondary to alterations in the left ventricular (LV) geometry (1,2). In both cases, IMR is associated with a critical obstruction (>70%) of at least one main coronary vessel (3). It is most commonly seen in ischemic cardiomyopathies. The degree of annular dilatation is much less in IMR than in degenerative MR (4). The anatomical and physiopathological mechanisms that originate IMR have not been fully clarified. However, remodelling of the LV related to severe myocardial ischemia is the main factor that induces the lesion, together with the involvement of the papillary muscles (PM). Therefore, its cause is an affectation of the mitral valvular spatial disposition, due to the involvement of the LV or because of an intrinsic affectation of it by coronary ischemic heart disease. The PM displacement that creates greater tension of the leaflets is posterolateral-apical, which explains why a located inferior AMI can produce a significant IMR, and why its incidence is much lower in the anterior AMI (2). Identifying the etiology of MR in ischemic heart disease is not easy, since a significant percentage of patients have previously shown MR of degenerative origin (5).
Prevalence
Currently MR is the second most common valve disease in Europe and affects approximately 31% of the population (6). IMR is the second cause of MR (25% of cases), after degenerative (60%) and before rheumatic (12%). The prevalence of IMR has increased progressively in recent years. Between 17–40% of patients who have suffered an AMI show early clinical or echocardiographic evidence of IMR (7). In a large percentage of auscultations, early murmurs are heard after an AMI, often transient and disappearing at discharge (8). IMR appears in about 20% of AMIs and is much more common in lower-located AMIs (38%) than in anterior-location AMIs (10%) (9).
Mechanisms of the IMR
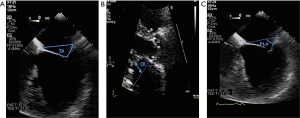
Several mechanisms have been proposed to explain the occurrence of IMR. Echocardiography determines global and local ventricular function and remodelling, degree of MV deformation, MV morphology, and severity of IMR (1). Chronic IMR, in most cases, is due to type IIIb of valvular dysfunction of the Carpentier classification. The restriction of leaflet motility occurs essentially in systole, being more frequent in patients with previous inferior AMI (asymmetric pattern) (Figure 1). Anterior and inferior AMI involves a systolic reduction of both leaflets, with incomplete coaptation (symmetrical pattern). Less frequently IMR is due to fibrosis and elongation of the PM (type II) (10). As we have already indicated, mitral tenting was directly determined by apical and posterior PM displacement. Although most patients present remote infarction and ventricular remodelling with leaflet tethering, acute ischemia with regional wall motion abnormalities can lead to similar findings of leaflet tethering in some patients (4). The saddle-shape of the mitral ring is important for maintaining the normal stress of the leaflets. Loss of this shape and crushing of the ring due to remodeling of the LV results in increased stress of the leaflets with secondary MR (11). Therefore, despite other contributing factors, the fundamental factor is the adverse ventricular remodelling that occurs after an AMI, which displaces PM producing “tension” of the cords, which restricts the movement of the leaflets, with reduced closing force of the MV due to systolic LV dysfunction and causes incomplete mitral closure. As these changes are dependent on load conditions and the phase of the cardiac cycle, secondary MR is dynamic in nature (1).
Clinic
The clinical expression of IMR assumes the sum of several cardiological factors and is usually asymptomatic (12). The acute appearance secondary to PM rupture due to AMI is presented as acute heart failure and it is associated with significant hemodynamic deterioration. However, progressive symptoms appear more frequently when the valvular lesion is severe and is manifested with the semiology of heart failure worsening when associated with left ventricular systolic dysfunction (LVSD) (13). In the physical examination, the pansystolic murmur in the mitral focus irradiated to the left axillary line stands out, although the presence of this in the IMR varies widely according to the series studied and may be absent in up to half of the patients (12). The intensity of the murmur has little relationship with the degree of regurgitation due to LVSD and left atrial (LA) compliance (14). Therefore, in the presence of ischemic cardiomyopathy, the absence of regurgitation murmur should not discard the possibility of an underlying MR.
Echocardiographic evaluation of the IMR
Echocardiography detects MV abnormalities (leaflets, subvalvular apparatus, mitral annulus and/or LV) that cause valvular regurgitation (1).
Ventricular remodelling and deformation of the mitral system
LV diameters, as well as 2D biplane volumes, or more accurately with 3D, as well as the sphericity index should be determined by echocardiography. In addition, echocardiography makes it possible to determine the extent and location of the segmental alterations and the parietal thinning of the LV, as well as the posterior and apical displacement of the posterior and anterior PM. Regarding the deformation of the mitral apparatus ring size, coaptation distance, leaflets angles and tenting area should be obtained in parasternal long axis in mesosystolic. Even 3D-echocardiography allows us to assess the volume of tenting, which seems to provide advantages over 2D.
Morphology of the MV
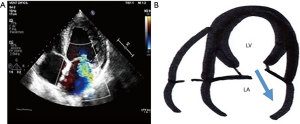
Asymmetric closure pattern of the IMR, shows a “hockey stick” or seagull sign on the echocardiogram. The anterior leaflet in systole is below the posterior leaflet, which is also stressed, altering the coaptation. An eccentric jet of insufficiency appears, ipsilateral to the posterior leaflet, which goes to the posterior region of the LA (Figure 2). This pattern is typical of inferior or inferolateral infarction.
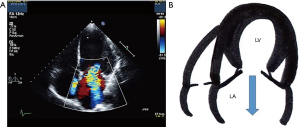
The symmetric closure is due to a global remodelling of the LV, with spheroidal shape and greater dysfunction. There is an apical displacement of both leaflets and the coaptation point, with greater dilation and flattening of the mitral annulus. The area and volume of tenting are greater than in the asymmetric pattern, being the origin and direction of the central regurgitation jet, by symmetrical affectation of both leaflets. It is more associated with anterior infarction or multiple infarcts (Figure 3).
Recurrence predictors of IMR after repair
IMR has recurrence rates after surgical repair of 30% at 3 years, rising to around 70% at 5 years (15). The presence of residual MR after surgery contributes to perpetuate the negative remodelling of the LV, associating more tethering of the mitral leaflets, generating a vicious circle that ends up increasing the severity of MR. The presence of significant postoperative IMR is associated with worse prognosis than its absence (16). Therefore, to prevent future complications it is necessary to avoid postoperative residual IMR (17). Echocardiography allows the study of the viability of valve repair in IMR. There are several independent predictors of MR recurrence, mainly divided into 2 groups (Table 1) (18):
- Global and local remodelling of the LV;
- Valve tethering.

Full table
Given that the underlying mechanism of MR recurrence is the persistence of leaflet tethering, the presence of aneurysm/dyskinesia is a potent recurrence predictor (19). On the other hand, a restrictive pattern of LV filling suggests very advanced myocardial dysfunction, giving greater negative remodelling after repair (20); severe dilatation of the LV is associated with a low probability of reverse remodelling after repair, leading to worse long-term results (21).
Parameters objectified by transthoracic echocardiography (TTE), predictors of failed MV repair are: coaptation distance ≥1 cm, tenting area >2.5 cm2 and angle of coaptation of the posterior leaflet ≥45º (high restriction of posterior leaflet motility), increase the probability of failed mitral repair (22). Some authors suggest that an angle between the tip of the anterior leaflet and the mitral ring >25° could be a predictor of recurrence (23). Other predictors are the presence of a jet of central regurgitation (severe mobile restriction of both leaflets) or complex jets of central and posteromedial origin (18).
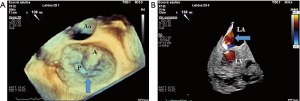
Using preoperative transesophageal echocardiography (TOE), a ring diameter ≥37 mm, with a tenting area ≥1.6 cm2 and a severe degree of MR implies a 50% probability of long-term surgical failure (24). TOE-3D may provide additional information in the surgical repair decision in IMR by providing a comprehensive visualization of the different components of the MV system (Figure 4).
Quantification of the severity of the IMR
TTE is an excellent method to assess the mechanism and severity of IMR, although it presents some limitations. The following echocardiographic methods are used to assess the severity of MR (1).
Colour flow imaging
It is the most used and simple method in the evaluation of IMR. It assumes that the greater the severity of MR, the greater the size and extension of the jet within LA. However, it is imprecise, because the relationship between extension of the jet and severity of regurgitation is not direct, depending on many technical and hemodynamic factors. Thus the interaction with a wall or valvular structure conditions its area, underestimating it (Coanda effect). While central flows drag flux in its path producing overestimation, a large eccentric flow that is distributed through the posterior wall of LA goes in favour of severe IMR. Conversely, small flows that appear just behind the mitral leaflets indicate slight insufficiencies.
Limitations
This method alone is not recommended to quantify the severity of the IMR, it should only be used to detect it. When more than just a small IMR central jet is observed, a more quantitative assessment is required.
Vena contracta (VC) width
The VC is the width of the insufficiency jet after traversing the regurgitant orifice, so it represents its area. Its measurement must be carried out in two perpendicular projections to the line of commissures (parasternal long axis or four-chamber apical axis). Measuring the lowest VC, immediately distal to the regurgitant orifice and perpendicular to the flow. A VC <3 mm indicates mild MR and a ≥7 mm defines it as severe. In the case of the IMR, the regurgitant orifice seems not to be circular, but rather more extended along the coaptation line. Furthermore, the Doppler image does not give an appropriate orientation of the 2D planes to obtain an accurate cross-sectional view of the VC. An average 8-mm VC diameter in 2D has been reported as severe MR of any etiology, including IMR. The VC seems to be less influenced by the load conditions of the LV and therefore, be more reproducible than the flow imaging colour methods.
Limitations
Intermediate VC values (3–7 mm) require confirmation by quantitative methods. Many times, VC is obtained in eccentric jets. In case of multiple jets, the respective values of VC are not additive. The assessment of VC by 3D echocardiography is still reserved for research.
Doppler volumetric method
Doppler volumetric method is a method used when the proximal isovelocity surface area (PISA) and the VC are not accurately applicable. The MR volume is obtained by calculating the difference between total stroke volume (product of the mitral ring area by the time-velocity integral (TVI) of the LV input tract flow) and the systemic stroke volume (product of the LV outflow tract area by the LV outflow tract TVI).
Limitations
It is time-consuming so it is not recommended as a first line method in the quantification of MR. Its calculation is inaccurate in the presence of significant aortic regurgitation.
The flow convergence method
It is the most recommended quantitative method. It is recommended: four-chamber apical view to determine PISA, then align the flow with the ultrasound beam, adjust the gain and lower the wall filter, decrease the depth and reduce the sector size to increase the spatio-temporal resolution. Decrease the aliasing speed immediately, creating a longer PISA, moving the baseline in the direction of the MR flow, up to a speed of 20–40 cm/s. Measure the PISA radius in mesosystolic, with the morphology of the flow as close to a hemisphere, in the first aliasing. Determine the TVI and peak flow velocity of MR with continuous Doppler. The effective regurgitant orifice area (EROA) is thus determined. This allows integrating the different severity indices, classifying MR in mild, moderate or severe (Table 2) (1). The primary MR is considered severe if EROA ≥40 mm2 and the regurgitant volume (R Vol) ≥60 mL. In secondary MR, the severity threshold is lower, 20 mm2 and 30 mL, respectively, indicating a subgroup of patients with an increased risk of cardiovascular events. EROA is the most robust parameter to determine the severity of the MR, being able to determine the PISA with both central and eccentric MR.

Full table
Limitations
In IMR there is a dynamic variation of the regurgitant orifice, with early and late systolic peaks and mesosystolic descent. Non-hemispheric PISA derived from eccentric jets, multiple or regurgitant or complex elliptical orifices may not be valid. The degree of MR could be underestimated, so a lower threshold is used in determining the severity of the functional MR.
Anterograde velocity of mitral inflow
In the absence of mitral stenosis a peak velocity E >1.5 m/s suggests severe MR, whereas a dominant A wave (atrial contraction) excludes it. This is applicable in patients over 50 years of age. The TVI ratio, between the Mitral inflow Doppler and the aortic flow at the level of the rings in four-chamber vision, is an additional strong parameter in the assessment of MR severity. A ratio of 1.4 suggests severity of MR.
Pulmonary venous flow
When MR severity increases there is a decrease in the S-wave velocity in the pulmonary vein flow determined by pulsed Doppler, being a sensitive but not very specific parameter of severity, since it can also appear in atrial fibrillation, ventricular dysfunction and increased LA pressure. In the most severe forms the S-wave can be reversed, being a specific parameter of severe MR.
Continuous wave Doppler of MR jet
Speed itself is not a parameter of severe MR. However, the intensity of the signal dense, dashed (“notched”), triangular and with an early peak speed (“blunt”) in its morphology indicates more severity. All of which indicates elevated LA pressure or a prominent regurgitant pressure wave in LA due to severe MR. In the eccentric MR it can be difficult to obtain a complete record, although the intensity of the signal is dense. The continuous jet Doppler MR is a qualitative parameter of MR severity.
Exercise stress echocardiography (ESE)
IMR is a dynamic valvulopathy. Many patients show episodes of heart failure due to an increase in R Vol and EROA (5). The European guidelines recommend the use of ESE to quantify the changes induced by exercise in MR, pulmonary arterial pressure and LV function. It is especially useful when there is a discrepancy between symptoms and severity of valve disease (6). ESE can identify symptoms and subclinical ischemic ventricular dysfunction in patients with asymptomatic IMR. In symptomatic patients it can help to objectively quantify the symptoms, confirm or rule out the ischemic etiology and evaluate the myocardial viability to decide the opportune treatment (25). It also has prognostic value, given that an increase in the EROA ≥13 mm2 with exercise is associated with greater morbidity and mortality and major cardiac events. An increase in the transtricuspid pressure gradient of exercise is associated with an increased risk of heart failure and death (5).
Gentry et al. established a series of high-risk criteria in relation to the findings in the ESE based on the MR classification (moderate or severe, primary or secondary) (Table 3) (26).

Full table
Strain echocardiography
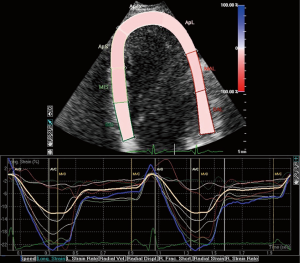
The role of the evaluation techniques of ventricular deformation, tissue Doppler and speckle tracking in the evaluation of valvular diseases is still under study. Longitudinal strain could be useful for the detection of subclinical LVSD (5) (Figure 5). Deterioration of the ventricular function in patients with severe chronic MR can go unnoticed by traditional 2D-echocardiography techniques, due to the loading conditions of the LV. The low afterload of these patients allows normal or even increased systolic function indices (15). The identification of early contractile dysfunction and its subsequent surgical correction could prevent the development of irreversible ventricular dysfunction in the postoperative period that would worsen the short and long-term prognosis (27). In addition, speckle tracking analysis was superior to tissue Doppler analysis and traditional methods of LV systolic function analysis (28). However, its use is limited by the heterogeneity of the algorithms used in different echocardiographic systems.
TOE: 2 and 3-dimensional
When TTE imaging is not optimal for characterization of valvular heart disease and establishment of surgical strategy, TOE is very useful and can replace it. TOE helps to exclude organic aetiology when evaluating the patient with MR of ischemic origin, and provides high image quality due to the proximity of the transducer to the valve, the subvalvular apparatus and the regurgitation jet, which is directed at it (29).
The quantification methods used in TOE have also been validated, and new parameters for estimating severity using TOE have also emerged (30,31). TOE allows a more accurate quantification of the mitral ring, tenting area, traction and decrease of the apposition surface compared to TTE (2) (Figure 6). As for 3D-TOE, it has been shown to be superior to 2D techniques for measuring the area of VC, the PISA area in those cases where it does not resemble a hemi-sphere and in obtaining the EROA by direct planimetry (25,32) (Figure 7).
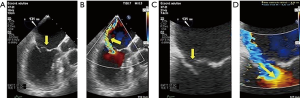
MR is sensitive to hemodynamic modifications; during the performance of a TOE, anaesthesia can modify R Vol, generating an underestimate of its severity, especially in functional MR (32,33). However, administration of vasocontracting drugs or intravascular volume infusion may restore basal haemodynamics allowing a true assessment of the magnitude of regurgitation (34).
Cardiovascular magnetic resonance in IMR
The study of cardiac pathology by means of magnetic resonance imaging (CMR) provides valuable information. However, the IMR evaluation is only relevant when echocardiography does not confirm the etiological diagnosis of MR. It presents lower special and temporal resolution, as well as worse morphoanatomical evaluation of the valvular and subvalvular apparatus (35). Using CMR, ventricular volumes, contractility, myocardial thickness and myocardial viability can be quantified by analysis of the late enhancement pattern with gadolinium (Figure 8). In addition, it allows to detect ischemia, to analyse the contractile reserve by dobutamine (36) and more complex techniques such as tagging or T1 mapping and its derivative study of extracellular volume (37,38). The application is greater in severe secondary chronic MR, which is difficult to evaluate hemodynamically, where the study of the underlying cardiomyopathy is the cornerstone of the therapeutic and prognostic approach.
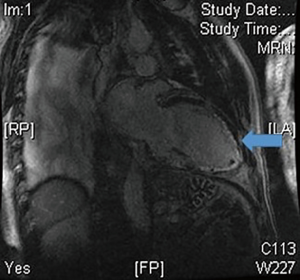
Echo gradient sequences are used to obtain cardiac functional studies. Their main characteristic is their high temporal resolution, which allows them to be analysed in MR-cinema mode, obtaining ventricular function and volumes (39). Subsequently, after gadolinium infusion using echo gradient sequences enhanced at modified T1 by the application of a previous 180° inversion pulse, we studied myocardial viability. In the case of IMR, we can analyse the viability of the segments related to the MV. The high spatial resolution of CMR differentiates necrotic myocardial segments from healthy ones, and also estimates the transmural extension of the infarction, increasing the diagnostic accuracy of the technique for myocardial viability study. In the classic study by Kim et al. for myocardial viability, they found an inverse relationship between the proportion of segments that improved after revascularization and transmural extension of late gadolinium enhancement (40). There was improvement in 78% of the segments without late gadolinium enhancement and only 2% in those with >75% deposition.
The severity of MR can be determined by calculating the flow volume in the mitral annulus and compare it to that of the aortic root. For this purpose, echo gradient sequences encode velocity by quantifying blood flow, so R Vol is the difference between the volume of flow in the valve ring and the volume of flow in the aorta (41). The Simpson method estimates the R Vol, comparing the systolic volume of the LV and the right ventricle, in the absence of other significant regurgitation or significant shunt.
Finally, CMR may play a significant role in the study of underlying cardiomyopathy responsible for IMR, rather than in the morphoanatomical evaluation of MR (42).
Prognosis
IMR has a great impact on the survival and development of post-AMI heart failure. Its existence is associated with a higher mortality compared to those patients who do not develop MR, being directly related to its severity (6). According to the severity, survival is estimated in 40% in moderate-severe IMR, 62% in mild and 84% in patients without IMR (3), and EROA ≥20 mm2, implied lower survival at 5 years than without MR (38%±5% vs. 61%±6%, P<0.001), regardless of ventricular function (8). This group also demonstrated that the existence of IMR independently increased 3.6 times the risk of suffering heart failure. Mortality rate 1 year after an AMI was 11% in patients without IMR, 22% in mild IMR and 52% with moderate-severe IMR (7). A more dramatic situation is acute primary IMR, with a mortality of 75% at 24 hours and 95% in the first 48 hours (9).
The presence of IMR in patients with non-ST elevation MI implies a higher frequency of major cardiovascular events, even in patients with mild IMR (43). In addition, there is worse prognosis in elderly patients, more than 1 diseased coronary vessel, previous AMI and LVSD. These results are consistent with records such as the Survival and Left Ventricular Enlargement Study. Conversely, shorter door-balloon times with the use of primary percutaneous coronary intervention in ST elevation MI resulted in lower MR rates and better long-term prognosis (44).
Treatment
Medical therapy
Acute IMR confers a significant haemodynamic impact and may require surgical correction. Although this is associated with higher mortality than scheduled surgery in chronic cases, if it is not treated intensively there is a high probability of mortality (45,46). On the other hand, vasodilators such as sodium nitroprusside, offer a reduction of the decisive afterload in the face of haemodynamic stabilization for surgical intervention (47,48). If the patient shows associated ventricular dysfunction, the possibility of adding dobutamine and intra-aortic balloon pump should be studied, although scientific evidence for this IMR options is scarce.
Conversely, in chronic cases, the presence of LVSD and the existence of coronary artery disease should be assessed. Ventricular dysfunction should be treated as recommended in the guidelines for patients with reduced ejection fraction (13,49). The goal of treatment is to control negative ventricular remodelling and decrease preload in patients with ventricular dysfunction (50). By means of angiotensin-converting enzyme inhibitors (ACEI) and aldosterone antagonists, both objectives are sought. There are few studies using ACEI on degenerative MR (51), although there are no studies that include IMR. There is evidence, although not significant, with captopril in the improvement of functional MR secondary to idiopathic dilated cardiomyopathy (52). The role of beta-blockers also seeks to avoid ventricular remodelling and improve ejection fraction through the reduction of myocardial oxygen consumption (13). A study with carvedilol for 4 months in patients with chronic heart failure, improved secondary MR in addition to the ejection fraction (53) and in another study it improved MR and increased diastolic reserve in patients with heart failure (54).
Cardiac resynchronization therapy (CRT)
The second last step before heart transplantation in LVSD patients is to use CRT when medical treatment is optimized. Patients with secondary MR who have LVSD and who are candidates for treatment with electronic devices are indicated for CRT (13). The use of these therapies may help improve symptoms, quality of life, and the degree of MR (55,56). However, the presence of MR after CRT implantation implies worse long-term prognosis. A recent analysis of CRT in patients with LVSD and IMR secondary to inferior AMI showed a decrease of MR severity, and improved PM alignment and LV systolic function at 2 years follow-up (57). Patients with inferoposterior scar had low probably of improve severity of MR. Therefore, the use of CRT in IMR should be based on prior viability study in LV. Patients with myocardial viability should be candidates to CRT when they have implantation criteria (58).
Coronary revascularization and MV repair versus replacement for IMR
Indication
The surgical indications for IMR are not clearly stated. In general, according to the different clinical guidelines (2,4,13,59), it is accepted to perform a mitral intervention when MR is severe, patients are symptomatic and have been treated in an optimal medical manner. In the case of indications for coronary artery bypass grafting (CABG), surgery on the MV is a class I recommendation according to ESC/EACTS guidelines (2).
Moderate ischemic mitral insufficiency
The approach to moderate IMR in patients undergoing CABG remains controversial. Different recent meta-analyses (60-64) reflect a higher incidence of moderate or severe postoperative IMR in the isolated CABG group; however, this does not appear to result in differences in mortality, serious adverse events, functional ability, or ventricular remodelling. These studies have limitations and an important one is that they do not include patients with repair techniques on the subvalvular apparatus, without correcting the ventricular geometry and the traction forces on the mitral leaflets, which could lead to unsatisfactory repair. In the absence of solid evidence, the surgeon currently makes the surgical decision based on the patient›s symptoms and the additional risks of performing a procedure added to the CABG. Future studies will improve the evidence, such as the proposed algorithm that supports surgical decisions based on a mathematical model describing the biomechanics of the MV and all its components (65).
MV replacement versus repair
The most effective IMR surgical treatment is also a controversial one. Some authors found lower mortality with MV repair versus MV replacement (66), although others did not observe significant differences (67,68). Several studies describe an increase in the recurrence of IMR with MV repair (19,66,68-70), although this does not usually translate into a worsening of functional status, quality of life or the appearance of major adverse events (19,68). The American Association for Thoracic Surgery Guidelines are the only ones, so far, that offer us guidance based on the presence or absence of different predictors of IMR recurrence (basal aneurysm/dyskinesia, significant venous traction, ventricular dilation) (59). As with the moderate IMR approach, studies of MV repair versus MV replacement often do not include subvalve repair techniques, which may decrease IMR recurrence rates with MV repair and establish new surgical indications.
Myocardial revascularization
Correction of IMR with an isolated revascularization procedure is based on achieving a valve function restoration by re-establishing adequate myocardial perfusion to normalise ventricular geometry. However, this appears to be insufficient in a high percentage of patients and the action on the MV substantially improves the degree of postoperative IMR. However, there is a lack of evidence to support its superiority in terms of in-hospital mortality, IMR grade improvement ratio, New York Heart Association (NYHA) medium postoperative functional class, and short and long-term survival rate (71,72).
MV replacement
MV replacement with a mechanical or biological prosthesis was the treatment of choice until the development of reconstructive surgery. There is now a clear consensus on the need for replacement with complete preservation of both mitral leaflets which reduces left ventricular size and post-load, improving ventricular remodelling and systolic function (73). Furthermore, oversizing of the prosthesis should be avoided, as very large valves can impair ventricular dynamics and ventricular conduction in these already damaged ventricles (4). As for the type of prosthesis, there is evidence that mechanics are associated with improved haemodynamics and functional capacity in exercise (74).
Reconstructive surgery
MV repair in IMR began with the restrictive annuloplasty introduced by Bolling et al. using a ring one or two size smaller, according to the intertrigonal distance (75). Rigid and complete rings should be used, which appear to provide a more durable repair, as the anterior ring also expands. In addition, a coaptation of the leaflets of 8 mm or more must be achieved and a residual MR by TOE of more than 1/4 (76) must not be allowed. The results of this technique have not been very satisfactory, given that annuloplasty does not correct other mechanisms of insufficiency, such as the LV remodelling, the displacement of the PM and the traction on the leaflets. This has led to the emergence of new surgical alternatives that appear to have similar mortality, addressing traction better and with a better remodelling process and a lower recurrence of IMR (77).
PM sling
Hvass et al. proposed to approximate the two PMs by means of a band around them (78). Compared to isolated annuloplasty, this technique results in better reverse remodelling LV, less traction on mitral leaflets and a reduction in moderate or greater recurrent MR in the medium term (79).
Plication of the PM
A U-shaped suture is passed through both PM and tied together until they are repositioned in the midline (80). It provides long-lasting mitral repair, associated with reverse remodelling LV and a high probability of major cardiac event free survival (81).
Imbrication of the PM
The distance between both PM is reduced by a suture that is passed from the basal to the medial portion of each PM and tied together. It is a simple and reproducible technique that prevents or delays the recurrence of IMR (81).
Creation of a polytetrafluoroethylene (PTFE) interpapillary bridge
It is a new, safe and reproducible technique that saves time and eliminates MR in the post-operative phase with promising long-term results. Proposed by Alotti et al., it involves the creation of a bridge with two 4-0 double-needle PTFE sutures between the two PM, approximating them to a maximum of 2 cm, by means of a terry loop and a surgical knot made between both sutures (82). The two needles of each thread are passed from medial to lateral in each PM and are used as neochords. In addition, more neochords can be placed anchored to the interpapillary bridge.
Replacement of the PM
This technique is performed with a Gore-Tex suture between the head of each PM and the ipsilateral mitral ring, one or both of which can be repositioned. It is important to act on both heads of the posterior muscle (83) and the repositioning should be done in the direction of the anterior ring to preserve the diastolic opening of the MV (84).
Increase in the surface area of the leaflets
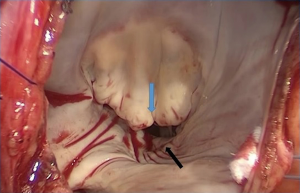
This technique, which can be performed on any of the mitral leaflets, is intended to increase the curvature of the leaflets and reduce traction (85,86). To do this, an incision is made near the base of the corresponding leaflet, parallel to the mitral annulus, generating an opening in which a pericardial patch is positioned. Good short- and medium-term results have been reported (Figure 9).

The Coapsys device
The Coapsys device consists of a left trans-ventricular cord that is placed under the MV and alters the morphology of the LV when stressed, decreasing the IMR and improving the functional class of the NYHA (87). The RESTOR-MV trial has shown an increase in survival and a decrease in major adverse effects with its use (88). On the other hand, it reduces stress on the myofibrils, which can contribute to its long-term effect on LV remodelling (89).
Ring-loop-string plastic
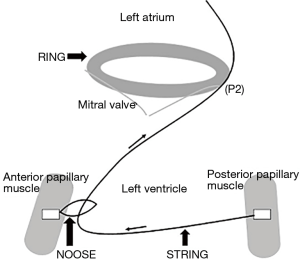
It is one of the most recent repair techniques and offers short-term acceptable results. Future studies are needed to demonstrate its medium and long term performance (90). Proposed by Bothe, it consists of suturing a PTFE loop at the base of the anterolateral papillary muscle, through which a PTFE suture (“cord”) is advanced with one of its ends fixed to the base of the posteromedial papillary muscle and with its other end located at the LA, crossing the MV at the P2 level (91). This brings the two MPs closer together. A restrictive mitral ring is then placed and a saline test is performed. Once the correct degree of coaptation has been achieved, the end of the PTFE “cord” is attached to the ring (Figure 10).
Second-order tendon cord section
The second-order tendon cord section of the anterior leaflet can improve coaptation without affecting ventricular function (92).
Transcatheter MV therapies
LVSD presence is associated with high mortality in secondary MR and medical or surgical treatment, therefore less invasive techniques capable of solving this problem have been sought. For over a decade, trials have been initiated to evaluate the efficacy and safety of the edge-to-edge transcatheter technique. The first of them was the EVEREST I trial, which showed promising results, although it did not establish inclusion criteria according to the aetiology of MR (93).
Later, the EVEREST II trial demonstrated an effective and promising technique, with patients randomly assigned to transcatheter therapies and surgery (94). The LV size, functional NYHA and quality of life improved in the first year, with no difference in terms of mortality or the presence of significant residual IMR at 1 and 4 years of follow-up. Complications were significantly greater in the surgical group and the transcatheter treatments demonstrated a 4:1 need for surgical reoperation. However, recent Surgery Thoracic Score records show that the need for reoperation in these techniques is the same in both treatment groups (95). Recently, it has been agreed that patients with asymptomatic residual MR are not subsidiaries of percutaneous or surgical reoperation (13). These treatments have reduced the need for re-hospitalization versus medical treatment (96). An analysis of the secondary MR subgroup showed that there was no difference in mortality at 1 year follow-up compared to surgical treatment (94).
The MitraClip device is not approved in the United States even for secondary MR, but is recommended in the EU for patients with high surgical risk. As a result of 3 European registries with predominance of randomization of patients with IMR, results have shown that the edge-to-edge technique is a useful alternative in the profile for these patients (97-99). The ACCES-EU record showed a significant improvement in the severity of regurgitation and symptoms (99). Even the use of this therapy may be useful in patients with LVSD and optimal treatment with neurohormonal blockade and carriers of resynchronization therapies, improving symptomatology and negative remodelling (100). These are the reasons why European guidelines have recently recommended the use of edge-to-edge transcatheter therapies in patients with secondary MR when high surgical risk coexists and optimal medical therapies are obtained, as well as resynchronization therapies (2). This includes, on the one hand, those who have no indication of coronary revascularization with an ejection fraction >30% and, on the other hand, patients without revascularization options and with an ejection fraction of <30%. Both groups of patients have an indication IIb but with a level of evidence C. There are other ongoing trials that will evaluate this technique in patients with secondary MR such as the MITRA-FR (101) and the COAPT Trial (102). The MITRA-FR recently published has shown that in patients with secondary IMR death rate or hospitalization due to heart failure at 1 year does not differ between patients who underwent percutaneous mitral valve repair, in addition to medical treatment and those who only received medical treatment. However, in the COAPT trial, transcatheter mitral-valve repair showed in a lower rate of hospitalization for heart failure and lower all-cause mortality within 2 years of follow-up than medical therapy alone.
New transcatheter techniques such as the Mitral Contouring System of the Carillon, the Cardioband and the Mitraling technique are being evaluated. However, they do not yet have any indication of MR and further research is needed. Multiple devices have been developed in recent years for Transcatheter MV Therapies such as the Fortis valve, Medtronic valve, CardiAQ valve, NaviGate valve, Tiara valve, Tendyne valve among others (103).
Conclusions
IMR is a common complication of ischemic cardiomyopathy that has prognostic implications and its prevalence has increased in recent years. Ventricular remodelling plays a central role in its pathophysiology, and thus behaves as a dynamic and complex entity in IMR. Given the high probability of poor clinical expression, echocardiography plays an essential role in the diagnosis. It provides information on the mechanisms involved in its development and is an excellent method for assessing the severity of IMR. Currently, new echocardiographic techniques provide additional information about this condition, so echocardiographic study in these patients should not eliminate the diagnosis to identify the severity and clinical impact. These patients have a high probability of severe associated ventricular dysfunction, preventing surgical treatment as an option, reserving only medical treatment. In recent years, significant advances in edge-to-edge transcatheter techniques have opened up a new horizon and allow this patient profile to have effective long-term treatment options.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Informed Consent: Written informed consent was obtained from the patients for publication of any accompanying images.
References
- Lancellotti P, Tribouilloy C, Hagendorff A, et al. Recommendations for the echocardiographic assessment of native valvular regurgitation: An executive summary from the European Association of Cardiovascular Imaging. Eur Heart J Cardiovasc Imaging 2013;14:611-44. [Crossref] [PubMed]
- Baumgartner H, Falk V, Bax JJ, et al. 2017 ESC/EACTS Guidelines for the management of valvular heart disease. Eur Heart J 2017;38:2739-91. [Crossref] [PubMed]
- Bursi F, Enriquez-Sarano M, Nkomo VT, et al. Heart failure and death after myocardial infarction in the community: The emerging role of mitral regurgitation. Circulation 2005;111:295-301. [Crossref] [PubMed]
- Kron IL, Acker MA, Adams DH, et al. 2015 the American Association for Thoracic Surgery Consensus Guidelines: Ischemic mitral valve regurgitation. J Thorac Cardiovasc Surg 2016;151:940-56. [Crossref] [PubMed]
- Azpitarte J, Alonso AM, Gallego FG, et al. Guidelines of the Spanish Society of Cardiology on valve heart disease. Rev Esp Cardiol 2000;53:1209-78. [PubMed]
- Lancellotti P, Troisfontaines P, Toussaint AC, et al. Prognostic importance of exercise-induced changes in mitral regurgitation in patients with chronic ischemic left ventricular dysfunction. Circulation 2003;108:1713-7. [Crossref] [PubMed]
- Tcheng JE, Jackman JD Jr, Nelson CL, et al. Outcome of patients sustaining acute ischemic mitral regurgitation during myocardial infarction. Ann Intern Med 1992;117:18-24. [Crossref] [PubMed]
- Grigioni F, Detaint D, Avierinos JF, et al. Contribution of ischemic mitral regurgitation to congestive heart failure after myocardial infarction. J Am Coll Cardiol 2005;45:260-7. [Crossref] [PubMed]
- Calafiore AM, Di Mauro M, Gallina S, et al. Mitral valve surgery for chronic ischemic mitral regurgitation. Ann Thorac Surg 2004;77:1989-97. [Crossref] [PubMed]
- Messika-Zeitoun D, Yiu SF, Grigioni F, et al. Determinants and prognosis of ischemic mitral regurgitation. Ann Cardiol Angeiol (Paris) 2003;52:86-90. [Crossref] [PubMed]
- Dal-Bianco JP, Levine RA. Anatomy of the Mitral Valve Apparatus. Role of 2D and 3D Echocardiography. Cardiol Clin 2013;31:151-64. [Crossref] [PubMed]
- Bursi F, Enriquez-Sarano M, Jacobsen SJ, et al. Mitral regurgitation after myocardial infarction: A review. Am J Med 2006;119:103-12. [Crossref] [PubMed]
- Nishimura RA, Otto CM, Bonow RO, et al. 2017 AHA/ACC Focused Update of the 2014 AHA/ACC Guideline for the Management of Patients With Valvular Heart Disease: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. J Am Coll Cardiol 2017;70:252-89. [Crossref] [PubMed]
- Desjardins VA, Enriquez-Sarano M, Tajik AJ, et al. Intensity of murmurs correlates with severity of valvular regurgitation. Am J Med 1996;100:149-56. [Crossref] [PubMed]
- Magne J, Pibarot P, Dumesnil JG, et al. Continued Global Left Ventricular Remodeling Is Not the Sole Mechanism Responsible for the Late Recurrence of Ischemic Mitral Regurgitation after Restrictive Annuloplasty. J Am Soc Echocardiogr 2009;22:1256-64. [Crossref] [PubMed]
- Hung J, Papakostas L, Tahta SA, et al. Mechanism of recurrent ischemic mitral regurgitation after annuloplasty: Continued LV remodeling as a moving target. Circulation 2004;110:II85-90. [Crossref] [PubMed]
- Agricola E, Oppizzi M, Pisani M, et al. Ischemic mitral regurgitation: Mechanisms and echocardiographic classification. Eur J Echocardiogr 2008;9:207-21. [PubMed]
- Lancellotti P, Moura L, Pierard LA, et al. European association of echocardiography recommendations for the assessment of valvular regurgitation. Part 2: Mitral and tricuspid regurgitation (native valve disease). Eur J Echocardiogr 2010;11:307-32. [Crossref] [PubMed]
- Kron IL, Hung J, Overbey JR, et al. Predicting recurrent mitral regurgitation after mitral valve repair for severe ischemic mitral regurgitation. J Thorac Cardiovasc Surg 2015;149:752-761.e1. [Crossref] [PubMed]
- Gelsomino S, Lorusso R, Billè G, et al. Left ventricular diastolic function after restrictive mitral ring annuloplasty in chronic ischemic mitral regurgitation and its predictive value on outcome and recurrence of regurgitation. Int J Cardiol 2009;132:419-28. [Crossref] [PubMed]
- Lancellotti P, Marwick T, Pierard LA. How to manage ischaemic mitral regurgitation. Heart 2008;94:1497-502. [Crossref] [PubMed]
- Magne J, Pibarot P, Dagenais F, et al. Preoperative posterior leaflet angle accurately predicts outcome after restrictive mitral valve annuloplasty for ischemic mitral regurgitation. Circulation 2007;115:782-91. [Crossref] [PubMed]
- Nappi F, Nenna A, Spadaccio C, et al. Predictive factors of long-term results following valve repair in ischemic mitral valve prolapse. Int J Cardiol 2016;204:218-28. [Crossref] [PubMed]
- Kongsaerepong V, Shiota M, Gillinov AM, et al. Echocardiographic Predictors of Successful Versus Unsuccessful Mitral Valve Repair in Ischemic Mitral Regurgitation. Am J Cardiol 2006;98:504-8. [Crossref] [PubMed]
- Zeng X, Levine RA, Hua L, et al. Diagnostic value of vena contracta area in the quantification of mitral regurgitation severity by color doppler 3D echocardiography. Circ Cardiovasc Imaging 2011;4:506-13. [Crossref] [PubMed]
- Gentry Iii JL, Phelan D, Desai MY, et al. The Role of Stress Echocardiography in Valvular Heart Disease: A Current Appraisal. Cardiology 2017;137:137-50. [Crossref] [PubMed]
- Starling MR, Kirsh MM, Montgomery DG, et al. Impaired left ventricular contractile function in patients with long-term mitral regurgitation and normal ejection fraction. J Am Coll Cardiol 1993;22:239-50. [Crossref] [PubMed]
- Matsumura T, Ohtaki E, Tanaka K, et al. Echocardiographic prediction of left ventricular dysfunction after mitral valve repair for mitral regurgitation as an indicator to decide the optimal timing of repair. J Am Coll Cardiol 2003;42:458-63. [Crossref] [PubMed]
- Dudzinski DM, Hung J. Echocardiographic assessment of ischemic mitral regurgitation. Cardiovasc Ultrasound 2014;12:46. [Crossref] [PubMed]
- Heinle SK, Hall SA, Brickner ME, et al. Comparison of Vena Contracta Width by Mitral Regurgitation. Am J Cardiol 1998;81:175-9. [Crossref] [PubMed]
- Yamanaka K, Mori M, Sato S, et al. Stacking-fault strengthening of biomedical Co-Cr-Mo alloy via multipass thermomechanical processing. Sci Rep 2017;7:10808. [Crossref] [PubMed]
- Lang RM, Tsang W, Weinert L, et al. Valvular heart disease: The value of 3-dimensional echocardiography. J Am Coll Cardiol 2011;58:1933-44. [Crossref] [PubMed]
- Bach DS, Deeb GM, Bolling SF. Accuracy of intraoperative transesophageal echocardiography for estimating the severity of functional mitral regurgitation. Am J Cardiol 1995;76:508-12. [Crossref] [PubMed]
- Savage RM, Konstadt S. CON: Proximal isovelocity surface area should not be measured routinely in all patients with mitral regurgitation. Anesth Analg 2007;105:944-6. [Crossref] [PubMed]
- Shanks M, Siebelink HM, Delgado V, et al. Quantitative Assessment of Mitral Regurgitation: Comparison Between Three-Dimensional Transesophageal Echocardiography and Magnetic Resonance Imaging. Circ Cardiovasc Imaging 2010;3:694-700. [Crossref] [PubMed]
- Baer FM, Voth E, Schneider CA, et al. Comparison of low-dose dobutamine-gradient-echo magnetic resonance imaging and positron emission tomography with [18F] fluorodeoxyglucose in patients with chronic coronary artery disease. A functional and morphological approach to the detection of residual myocardial viability. Circulation 1995;91:1006-15. [Crossref] [PubMed]
- Chinitz JS, Chen D, Goyal P, et al. Mitral apparatus assessment by delayed enhancement CMR: Relative impact of infarct distribution on mitral regurgitation. JACC Cardiovasc Imaging 2013;6:220-34. [Crossref] [PubMed]
- Calkoen EE, Roest AA, Kroft LJ, et al. Characterization and improved quantification of left ventricular inflow using streamline visualization with 4DFlow MRI in healthy controls and patients after atrioventricular septal defect correction. J Magn Reson Imaging 2015;41:1512-20. [Crossref] [PubMed]
- Bluemke DA, Atalar E, Mcveigh ER. Segmented K-space cine breath-hold cardiovascular MR imaging: Part 1. Principles and technique. AJR Am J Roentgenol 1997;169:395-400. [Crossref] [PubMed]
- Kim RJ, Wu E, Rafael A, et al. The use of contrast-enhanced magnetic resonance imaging to identify reversible myocardial dysfunction. The New England Journal of Medicine 2000;343:1445-1453. [Crossref] [PubMed]
- Szolar DH, Higgins CB. Cardiovascular Applications of Magnetic Rexonance Flow and Velocity Measurements. J Magn Reson Imaging 1996;6:78-89. [Crossref] [PubMed]
- Pflugfelder PW, Cheitlin D, Cassidy M, et al. Comparison of cine MR imaging with Doppler echocardiography for the evaluation of aortic regurgitation. AJR Am J Roentgenol 1989;152:729-35. [Crossref] [PubMed]
- Núñez Gil IJ, Pérez de Isla L, García-Rubira JC, et al. Ischemic mitral regurgitation and non-ST-segment elevation acute myocardial infarction: long-term prognosis. Rev Esp Cardiol 2009;62:1267-75. [PubMed]
- Mentias A, Raza MQ, Barakat AF, et al. Prognostic Significance of Ischemic Mitral Regurgitation on Outcomes in Acute ST-Elevation Myocardial Infarction Managed by Primary Percutaneous Coronary Intervention. Am J Cardiol 2017;119:20-6. [Crossref] [PubMed]
- Chatterjee S, Rankin JS, Gammie JS, et al. Isolated mitral valve surgery risk in 77,836 patients from the society of thoracic surgeons database. Ann Thorac Surg 2013;96:1587-94. [Crossref] [PubMed]
- Gammie JS, Sheng S, Griffith BP, et al. Trends in Mitral Valve Surgery in the United States: Results From The Society of Thoracic Surgeons Adult Cardiac Database. Ann Thorac Surg 2009;87:1431-7. [Crossref] [PubMed]
- Yoran C, Yellin EL, Becker RM, et al. Mechanism of reduction of mitral regurgitation with vasodilator therapy. Am J Cardiol 1979;43:773-7. [Crossref] [PubMed]
- Massie BM, Chatterjee K. Vasodilator therapy of pump failure complicating acute myocardial infarction. Med Clin North Am 1979;63:25-51. [Crossref] [PubMed]
- Ponikowski P, Voors AA, Anker SD, et al. 2016 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure: The Task Force for the diagnosis and treatment of acute and chronic heart failure of the European Society of Cardiology (ESC). Developed with the special contribution of the Heart Failure Association (HFA) of the ESC. Eur J Heart Fail 2016;18:891-975. [Crossref] [PubMed]
- Asgar AW, Mack MJ, Stone GW. Secondary mitral regurgitation in heart failure: Pathophysiology, prognosis, and therapeutic considerations. J Am Coll Cardiol 2015;65:1231-48. [Crossref] [PubMed]
- Tischler MD, Rowan M, LeWinter MM. Effect of Enalapril therapy on left ventricular mass and volumes in asymptomatic chronic, severe mitral regurgitation secondary to mitral valve prolapse. Am J Cardiol 1998;82:242-5. [Crossref] [PubMed]
- Evangelista-Masip A, Bruguera-Cortada J, Serrat-Serradell R, et al. Influence of mitral regurgitation on the response to captopril therapy for congestive heart failure caused by idiopathic dilated cardiomyopathy. Am J Cardiol 1992;69:373-6. [Crossref] [PubMed]
- Lowes BD, Gill EA, Abraham WT, et al. Effects of carvedilol on left ventricular mass, chamber geometry, and mitral regurgitation in chronic heart failure. Am J Cardiol 1999;83:1201-5. [Crossref] [PubMed]
- Capomolla S, Febo O, Gnemmi M, et al. Beta-blockade therapy in chronic heart failure: diastolic function and mitral regurgitation improvement by carvedilol. Am Heart J 2000;139:596-608. [Crossref] [PubMed]
- St John Sutton MG, Plappert T, Abraham WT, et al. Effect of cardiac resynchronization therapy on left ventricular size and function in chronic heart failure. Circulation 2003;107:1985-90. [Crossref] [PubMed]
- van Bommel RJ, Marsan NA, Delgado V, et al. Cardiac resynchronization therapy as a therapeutic option in patients with moderate-severe functional mitral regurgitation and high operative risk. Circulation 2011;124:912-9. [Crossref] [PubMed]
- Mihos CG, Yucel E, Capoulade R, et al. Impact of cardiac resynchronization therapy on mitral valve apparatus geometry and clinical outcomes in patients with secondary mitral regurgitation. Echocardiography 2017;34:1561-7. [Crossref] [PubMed]
- Sénéchal M, Lancellotti P, Magne J, et al. Impact of Mitral Regurgitation and Myocardial Viability on Left Ventricular Reverse Remodeling After Cardiac Resynchronization Therapy in Patients With Ischemic Cardiomyopathy. Am J Cardiol 2010;106:31-7. [Crossref] [PubMed]
- Kron IL, LaPar DJ, Acker MA, et al. 2016 update to The American Association for Thoracic Surgery consensus guidelines: Ischemic mitral valve regurgitation. J Thorac Cardiovasc Surg 2017;153:1076-9. [Crossref] [PubMed]
- Anantha Narayanan M, Aggarwal S, Reddy YNV, et al. Surgical Repair of Moderate Ischemic Mitral Regurgitation--A Systematic Review and Meta-analysis. Thorac Cardiovasc Surg 2017;65:447-56. [Crossref] [PubMed]
- Kopjar T, Gasparovic H, Mestres CA, et al. Meta-analysis of concomitant mitral valve repair and coronary artery bypass surgery versus isolated coronary artery bypass surgery in patients with moderate ischaemic mitral regurgitation. Eur J Cardiothorac Surg 2016;50:212-22. [Crossref] [PubMed]
- Michler RE, Smith PK, Parides MK, et al. Two-Year Outcomes of Surgical Treatment of Moderate Ischemic Mitral Regurgitation. N Engl J Med 2016;374:1932-41. [Crossref] [PubMed]
- Virk SA, Tian DH, Sriravindrarajah A, et al. Mitral valve surgery and coronary artery bypass grafting for moderate-to-severe ischemic mitral regurgitation: Meta-analysis of clinical and echocardiographic outcomes. J Thorac Cardiovasc Surg 2017;154:127-36. [Crossref] [PubMed]
- Zhang Y, Ma L, Zhao H. Efficacy of mitral valve repair as an adjunct procedure to coronary artery bypass grafting in moderate ischemic mitral regurgitation: A meta-analysis of randomized trials. J Card Surg 2015;30:623-30. [Crossref] [PubMed]
- Nappi F, Spadaccio C, Mihos CG, et al. Biomechanics raises solution to avoid geometric mitral valve configuration abnormalities in ischemic mitral regurgitation. J Thorac Dis 2017;9:S624-8. [Crossref] [PubMed]
- Yun-Dan D, Wen-Jing D, Xi-Jun X. Comparison of Outcomes following Mitral Valve Repair versus Replacement for Chronic Ischemic Mitral Regurgitation: A Meta-Analysis. Thorac Cardiovasc Surg 2017;65:432-41. [Crossref] [PubMed]
- Li B, Chen S, Sun H, et al. Mitral valve annuloplasty versus replacement for severe ischemic mitral regurgitation. Sci Rep 2018;8:1537. [Crossref] [PubMed]
- Acker MA, Parides MK, Perrault LP, et al. Mitral-valve repair versus replacement for severe ischemic mitral regurgitation. N Engl J Med 2014;370:23-32. [Crossref] [PubMed]
- Goldstein D, Moskowitz AJ, Gelijns AC, et al. Two-Year Outcomes of Surgical Treatment of Severe Ischemic Mitral Regurgitation. N Engl J Med 2016;374:344-53. [Crossref] [PubMed]
- Chan V, Levac-Martinho O, Sohmer B, et al. When Should the Mitral Valve Be be Repaired or Replaced in Patients With Ischemic Mitral Regurgitation? Ann Thorac Surg 2017;103:742-7. [Crossref] [PubMed]
- Yin L, Wang Z, Shen H, et al. Coronary artery bypass grafting versus combined coronary artery bypass grafting and mitral valve repair in treating ischaemic mitral regurgitation: A meta-analysis. Heart Lung Circ 2014;23:905-12. [Crossref] [PubMed]
- Zhang H, Liu Y, Qiu S, et al. Short-Term and Long-Term Survival After Revascularization with or without Mitral Valve Surgery of Patients with Ischemic Mitral Valve Regurgitation: A Meta-Analysis. Med Sci Monit 2015;21:3784-91. [Crossref] [PubMed]
- Yun KL, Sintek CF, Miller DC, et al. Randomized trial comparing partial versus complete chordal-sparing mitral valve replacement: Effects on left ventricular volume and function. J Thorac Cardiovasc Surg 2002;123:707-14. [Crossref] [PubMed]
- Fino C, Iacovoni A, Pibarot P, et al. Exercise Hemodynamic and Functional Capacity After Mitral Valve Replacement in Patients With Ischemic Mitral Regurgitation. Circ Heart Fail 2018;11:e004056. [Crossref] [PubMed]
- Bolling SF, Deeb GM, Brunsting L, et al. Early outcome of mitral valve reconstruction in patients with end-stage cardiomyopathy. J Thorac Cardiovasc Surg 1995;109:676-682. [Crossref] [PubMed]
- Durán D, Lugo J, Montoto J, et al. El tratamiento quirúrgico de la insuficiencia mitral isquémica. Cirugía Cardiovasc 2011;18:309-17. [Crossref]
- Athanasopoulos LV, Casula RP, Punjabi PP, et al. A technical review of subvalvular techniques for repair of ischaemic mitral regurgitation and their associated echocardiographic and survival outcomes. Interact Cardiovasc Thorac Surg 2017;25:975-82. [Crossref] [PubMed]
- Hvass U, Tapia M, Baron F, et al. Papillary muscle sling: a new functional approach to mitral repair in patients with ischemic left ventricular dysfunction and functional mitral regurgitation. Ann Thorac Surg 2003;75:809-11. [Crossref] [PubMed]
- Mihos CG, Yucel E, Santana O. The role of papillary muscle approximation in mitral valve repair for the treatment of secondary mitral regurgitation. Eur J Cardiothorac Surg 2017;51:1023-30. [PubMed]
- Rama A, Praschker L, Barreda E, et al. Papillary Muscle Approximation for Functional Ischemic Mitral Regurgitation. Ann Thorac Surg 2007;84:2130-1. [Crossref] [PubMed]
- Furukawa K, Yano M, Nakamura E, et al. Comparison of mitral competence after mitral repair with papillary muscle approximation versus papillary muscle relocation for functional mitral regurgitation. Heart Vessels 2018;33:72-9. [Crossref] [PubMed]
- Alotti N. L Ume K, Sayour A. Early Results of a Novel Mitral Valve Repair Procedure: The Interpapillary Polytetrafluoroethylene Bridge Formation. Int J Cardiovasc Res 2016;5. [Crossref] [PubMed]
- Fattouch K, Lancellotti P, Castrovinci S, et al. Papillary muscle relocation in conjunction with valve annuloplasty improve repair results in severe ischemic mitral regurgitation. J Thorac Cardiovasc Surg 2012;143:1352-5. [Crossref] [PubMed]
- Watanabe T, Arai H, Nagaoka E, et al. Influence of procedural differences on mitral valve configuration after surgical repair for functional mitral regurgitation: in which direction should the papillary muscle be relocated? J Cardiothorac Surg 2014;9:185. [Crossref] [PubMed]
- Rabbah JPM, Siefert AW, Bolling SF, et al. Mitral valve annuloplasty and anterior leaflet augmentation for functional ischemic mitral regurgitation: Quantitative comparison of coaptation and subvalvular tethering. J Thorac Cardiovasc Surg 2014;148:1688-93. [Crossref] [PubMed]
- de Varennes B, Chaturvedi R, Sidhu S, et al. Initial results of posterior leaflet extension for severe type IIIb ischemic mitral regurgitation. Circulation 2009;119:2837-43. [Crossref] [PubMed]
- Mishra YK, Mittal S, Jaguri P, et al. Coapsys mitral annuloplasty for chronic functional ischemic mitral regurgitation: 1-Year results. Ann Thorac Surg 2006;81:42-6. [Crossref] [PubMed]
- Grossi EA, Patel N, Woo YJ, et al. Outcomes of the RESTOR-MV trial (Randomized Evaluation of a Surgical Treatment for Off-Pump Repair of the Mitral Valve). J Am Coll Cardiol 2010;56:1984-93. [Crossref] [PubMed]
- Carrick R, Ge L, Lee LC, et al. Patient-specific finite element-based analysis of ventricular myofiber stress after Coapsys: importance of residual stress. Ann Thorac Surg 2012;93:1964-71. [Crossref] [PubMed]
- Pingpoh C, Siepe M, Bothe W. Surgical treatment of secondary mitral regurgitation: is repair a reasonable option? J Vis Surg 2017;3:158. [Crossref] [PubMed]
- Bothe W, Doenst T. Ring-Noose-String Technique Allows Adjustable Papillary Muscle Repositioning during Minimally Invasive Mitral Valve Repair in Patients with Functional/Ischemic Mitral Regurgitation. Thorac Cardiovasc Surg 2016;64:447-9. [PubMed]
- Messas E. Efficacy of Chordal Cutting to Relieve Chronic Persistent Ischemic Mitral Regurgitation. Circulation 2003;108:II111-5. [Crossref] [PubMed]
- Feldman T, Kar S, Rinaldi M, et al. Percutaneous Mitral Repair With the MitraClip System. Safety and Midterm Durability in the Initial EVEREST (Endovascular Valve Edge-to-Edge REpair Study) Cohort. J Am Coll Cardiol 2009;54:686-94. [Crossref] [PubMed]
- Mauri L, Foster E, Glower DD, et al. 4-Year results of a randomized controlled trial of percutaneous repair versus surgery for mitral regurgitation. J Am Coll Cardiol 2013;62:317-28. [Crossref] [PubMed]
- Grover FL, Vemulapalli S, Carroll JD, et al. 2016 Annual Report of The Society of Thoracic Surgeons/American College of Cardiology Transcatheter Valve Therapy Registry. J Am Coll Cardiol 2017;69:1215-30. [Crossref] [PubMed]
- Giannini C, Fiorelli F, De Carlo M, et al. Comparison of percutaneous mitral valve repair versus conservative treatment in severe functional mitral regurgitation. Am J Cardiol 2016;117:271-7. [Crossref] [PubMed]
- Puls M, Lubos E, Boekstegers P, et al. One-year outcomes and predictors of mortality after MitraClip therapy in contemporary clinical practice: Results from the German transcatheter mitral valve interventions registry. Eur Heart J 2016;37:703-12. [Crossref] [PubMed]
- Alajaji WA, Akl EA, Farha A, et al. Surgical versus medical management of patients with acute ischemic mitral regurgitation: a systematic review. BMC Res Notes 2015;8:712. [Crossref] [PubMed]
- Maisano F, Franzen O, Baldus S, et al. Percutaneous mitral valve interventions in the real world: Early and 1-year results from the ACCESS-EU, A prospective, multicenter, nonrandomized post-approval study of the Mitraclip therapy in Europe. J Am Coll Cardiol 2013;62:1052-61. [Crossref] [PubMed]
- Auricchio A, Schillinger W, Meyer S, et al. Correction of mitral regurgitation in nonresponders to cardiac resynchronization therapy by MitraClip improves symptoms and promotes reverse remodeling. J Am Coll Cardiol 2011;58:2183-9. [Crossref] [PubMed]
- Obadia JF, Messika-Zeitoun D, Leurent G, et al. Percutaneous Repair or Medical Treatment for Secondary Mitral Regurgitation. N Engl J Med 2018;379:2297-306. [Crossref] [PubMed]
- Stone GW, Lindenfeld J, Abraham WT, et al. Transcatheter Mitral-Valve Repair in Patients with Heart Failure. N Engl J Med 2018. [Epub ahead of print]. [Crossref] [PubMed]
- Natarajan N, Patel P, Bartel T, et al. Peri-procedural imaging for transcatheter mitral valve replacement. Cardiovasc Diagn Ther 2016;6:144-59. [Crossref] [PubMed]