
Effect of sequential nicorandil on myocardial microcirculation and short-term prognosis in acute myocardial infarction patients undergoing coronary intervention
Introduction
Timely and successful revascularization through primary percutaneous coronary intervention (PPCI) is the most effective treatment strategy to reduce the range of myocardial infarction (MI). After the recanalization of culprit vessels, approximately 25–37% of patients would develop myocardial reperfusion disorder and even no-reflow (1). The fundamental cause is the damage to the microcirculation of the myocardium. In recent years, research on the prevention and treatment of myocardial microcirculation injury has continuously progressed (2). Related studies have revealed that nicorandil has a protective effect on ischemic myocardium, and it can increase myocardial blood flow perfusion and improve clinical prognosis (3-6). However, there was no unified recommendation for the time and scheme of drug use (7-9). In the present study, patients with acute ST-segment elevation myocardial infarction (STEMI), who were receiving PPCI, were treated with the intracoronary administration and peripheral venous injection of nicorandil to evaluate its effect on myocardial microcirculation and short-term prognosis in STEMI patients.
Methods
Subjects
The present study is a single-center, prospective, randomized, single-blinded and blank-controlled study. Consecutive STEMI patients who received PPCI in the Heart Center of Tianjin Third Central Hospital from January 2016 to August 2017 were included into the present study. In order to ensure the comparability of experimental data, patients intervened by the same physician were selected. Finally, 140 eligible subjects were included.
Inclusion criteria: all patients who were definitely diagnosed with STEMI and received emergency coronary angiography after the onset of the disease were included. The angiography revealed that the thrombolysis in myocardial infarction (TIMI) grades of the culprit vessels were within grades 0–1. The STEMI diagnostic criteria were with reference to ESC 2012 (10). Exclusion criteria: patients who previously received coronary stent implantation; patients with a systolic blood pressure ≤90 mmHg or a diastolic blood pressure ≤60 mmHg; patients with liver dysfunction (serum ALT or total bilirubin >3 times of the normal upper limit) and renal insufficiency (serum creatinine >1.5 times of the normal upper limit); patients who were presently receiving nicorandil; patients who had undergone coronary artery bypass grafting (CABG); patients with prior MI; patients with cardiogenic shock.
The present study was conducted in accordance with the declaration of Helsinki and approved by the Ethics Committee of our hospital. All subjects and their families provided a signed informed consent.
Grouping
According to the random number table method, these patients were divided into three groups: sequential nicorandil group (n=48), intracoronary nicorandil group (n=47), and control group (n=45).
During the PPCI process, when the blood flow remained within TIMI 0–1 or obvious thrombus shadows could be observed after the guidewire passed through the culprit vessel, thrombus aspiration was first performed. The diagnosis of TIMI after PCI was assessed by an experienced technician without knowledge of the patient group allocation. Then, patients in the sequential nicorandil group received an intracoronary injection of 4 mg (8 mL) of nicorandil (Beijing SHKB Pharmaceutical Co., Ltd., National drug approval no.: H20120069, Specification: 12 mg), and nicorandil was continuously pumped through the peripheral vein at a rate of 4 mg (4 mL)/h for 24 h. Patients in the intracoronary nicorandil group received an intracoronary injection of 4 mg (8 mL) of nicorandil, and normal saline was continuously pumped through the peripheral vein at a rate of 4 mL/h for 24 h. Patients in the control group received an intracoronary injection of 8 mL of normal saline, and normal saline was continuously pumped through the peripheral vein at a rate of 4 mL/h for 24 h. During the PPCI process, if the blood flow reached TIMI 2–3 after the guidewire passed through the culprit vessel, thrombus aspiration was not necessary and patients were directly given the above treatments. Then, these three groups of patients received routine percutaneous coronary balloon dilatation (PTCA), and stent was implanted when necessary. All the stents were drug-eluting stent (DES). Before the operation, patients in the three groups orally took loading dose of antiplatelet drugs (300 mg of aspirin and 180 mg of ticagrelor) and were intravenously injected with normal heparin (50–70 U/kg). Whether tirofiban should be used intraoperatively and postoperatively was decided by surgeon according to the complexity of vascular lesions and thrombotic load. After the operation, patients in the three groups orally took 100 mg/d of aspirin and 90 mg/bid of ticagrelor, patients without contraindications were routinely given statins, β-receptor blockers and angiotensin-converting enzyme inhibitors.
Observation indices
Basic information of patients
The information of patients included their general information (gender and age), the risk factors of coronary heart disease and complicated diseases (hypertension, diabetes, hyperlipidemia, smoking and drinking history, family history of coronary heart disease, atrial fibrillation and stroke), the characteristics of the disease, and the lesions in the culprit vessels (interval from the appearance of symptoms to revascularization, the location of the culprit vessel, the number of the sites of the lesions, the grade of TIMI blood flow in the culprit vessels before the recanalization, and the implantation of stents).
Calculations of corrected TIMI frame count (CTFC)
The CTFC was measured according to the method of Gibson et al. to determine blood flow at the time of coronary angiography. The cine frame counts derived from the injection of contrast medium to the peripheral landmark were measured on the coronary angiogram. The frame rate was measured at 30 frames/s. Coronary angiography was performed at a 30° right anterior oblique/caudal projection angle for the left anterior descending and left circumflex arteries and a 60° or 30° left anterior oblique/cranial projection angle for the right coronary artery. The CTFC was measured by a physician without knowledge of the patient group allocation.
Safety indexes
The heart rate and noninvasive blood pressure of the upper limb artery were measured before PPCI, and at 24 h after PPCI. Creatinine and alanine transaminase were measured before PPCI and at 48 h after PPCI. Changes in heart rate, blood pressure and liver and kidney functions before and after PPCI were observed in these three groups.
Outcome indicators
The main observation indexes included coronary blood flow and myocardial perfusion immediately after PPCI. The following indicators were observed in these three groups of patients: (I) The proportion of patients with TIMI 3 blood flow. The diagnosis of TIMI after PCI was assessed by an experienced technician without knowledge of the patient group allocation. (II) CTFC. (III) The ST segment resolution: The 12-lead electrocardiogram was recorded before the revascularization, immediately after PPCI and at 90 and 120 minutes after PPCI, respectively. STR was measured by a physician specialized without knowledge of the patient group allocation. Evaluation of anterior infarction was based on the total sum of the ST elevation in leads V1–6, I and aVL, and the total sum of the ST depression in leads II, III and aVF. Evaluation of non-anterior infarction was based on the total sum of the ST elevation in leads II, III and aVF (including I, aVL, V5, and V6, if present), and the total sum of the ST depression in V1–4. Compared with the preoperative electrocardiogram, STR at 2 h after revascularization by >70% was defined as complete resolution, otherwise it was defined as incomplete resolution. The elevation of the ST segment was defined as the elevation of the electrocardiogram signal from point J to a subsequent site after 0.08 s. ECG were evaluated by the ECG expert without knowledge of the patient group allocation. (IV) Peak time of creatinine kinase MB isoenzyme (CK-MB): The myocardial markers were measured at 12, 14 and 16 h after onset of the disease, and the time point when the myocardial enzyme reaches the peak is the peak time of myocardial enzyme. (V) CK-MB peak value.
Secondary observation indexes: (I) major adverse cardiovascular events (MACE) during the period of hospitalization, including all-cause mortality, chest pain (the symptom of persistent chest pain for more than 30 minutes), malignant arrhythmia (ventricular tachycardia/ventricular fibrillation/II–III degree atrioventricular block, repeat revascularization and deterioration of cardiac function (Killip classification increased by grade 1 or above). The MACE was reported to the researcher by the physician (not participating in the study). (II) The left ventricular ejection fraction (LVEF) was measured within 1 week after PPCI.
Statistical methods
All data were processed using SPSS 19.0 statistical software. Normally distributed measurement data were expressed as mean ± standard deviation. When the variance was homogeneous, the variables were compared among the three groups using univariate analysis of variance, and pairwise comparison among groups was performed using the Student-Newman-Keuls (SNK) and Dunnett tests. When the variance was not homogeneous, the variables were compared among the three groups using nonparametric test. The means of non-normally distributed measurement data were compared among the three groups using the Kruskal-Wallis H test. Comparisons in the same group between before and after treatment were conducted using matching t-test, and count data were expressed as percentage (%). The rates among the three groups were compared using the χ2-test. P<0.05 was considered statistically significant (P<0.017 was considered statistically significant in the pairwise comparison among the three groups).
Results
Basic information (Table 1)
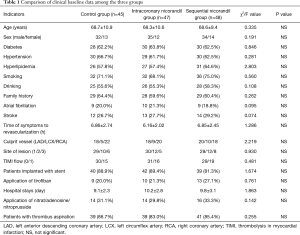
Full table
The mean age of patients enrolled in the present study was 68.5±10.3 years old (ranged, 40–80 years old). Among these patients, 101 patients (72.1%) were male and 39 patients (27.9%) were female. There’re 13.6% (n=19) of patients with no stent in the culprit artery. Ten of them had PTCA without stent, five of them had artery diameter less than 2.5 mm and the other 4 rejected to have stent implantation. For the risk factors of coronary heart disease, 88 patients (62.9%) had diabetes, 89 patients (63.6%) had hypertension, 84 patients (60%) had hyperlipidemia, 100 patients (71.4%) had a history of smoking, 79 patients (56.4%) had a history of drinking, and 86 patients (61.4%) had a family history of coronary heart disease. Furthermore, 28 (20%) patients had complicated diseases that included atrial fibrillation, and 39 patients had stroke (27.9%). For the characteristics of onset, the interval from the appearance of symptoms to revascularization was 6.62±2.42 h. Furthermore, the culprit vessel was the anterior descending branch or left main trunk-anterior descending branch in 56 patients (40%), the circumflex branch in 24 patients (17.1%), and the right coronary artery in 60 patients (42.9%). In addition, 87 patients (62.1%) had single-vessel disease, 34 patients (24.3%) had double-vessel disease, 19 patients (13.6%) had triple vessel disease. Furthermore, 90 patients (64.3%) had TIMI-0 flow, and 50 patients (35.7%) had TIMI-1 flow. Moreover, 121 patients (86.4%) were implanted with stents during PPCI. Differences in baseline data, risk factors of coronary heart disease, complicated diseases and the characteristics of onset among the three groups were not statistically significant.
Comparison of changes in perioperative blood pressure, heart rate and liver and kidney function among the three groups (Table 2)
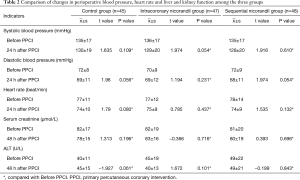
Full table
These three groups of patients were self-controlled. Blood pressure and heart rate before PPCI, and at 24 h after PPCI, and serum levels of creatinine and glutamic-pyruvic transaminase before and at 48 h after PPCI were compared. The differences were not statistically significant.
Comparison of indexes of its curative effect after PPCI among the three groups (Table 3)
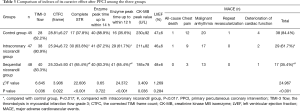
Full table
After PPCI, the difference in the proportion of patients with TIMI-3 flow among the three groups was statistically significant (P=0.036), where this proportion was higher in the sequential nicorandil group and intracoronary nicorandil group than in the control group (P=0.022 and P=0.047). The difference in CTFC among the three groups was statistically significant (P=0.022), where CTFC was lower in the sequential nicorandil group and intracoronary nicorandil group than in the control group (P=0.010 and P=0.031). The differences in the proportion of patients with complete STR and advancing of enzyme peak time to within 12 h between each two groups were statistically significant (P<0.001), where this proportion was the highest in the sequential nicorandil group. The difference in the CK-MB peak among the three groups was statistically significant (P=0.036), where the CK-MB peak was lower in the sequential nicorandil group than in the control group (P=0.012). The difference in the incidence of MACE between each two groups was statistically significant (P<0.001), where this incidence was the lowest in the sequential nicorandil group; The differences in the proportion of patients with advancing of enzyme peak time to within 14 h after revascularization and LVEF among the three groups were not statistically significant (P=0.722 and P=0.284).
Discussion
Timely myocardial reperfusion realized by means of PPCI is an effective treatment strategy to reduce the mortality of AMI. However, even if the culprit vessel is recanalized and normal blood flow is recovered some patients still feel chest pain, and the ST elevation is not complete resolution, even serious clinical manifestations such as malignant arrhythmia and heart failure occur. This is closely related to coronary microvascular dysfunction caused by reperfusion injury (11,12). This phenomenon reduces the benefit of PCI. In the past 10 or more years, a large number of clinical trials on glycoprotein IIb/IIIa receptor antagonists, nitrates, calcium antagonists and other drugs have been carried out to solve the above-mentioned problems. However, the effectiveness of these drugs has not been conclusive.
In recent years, some scholars have applied nicorandil in the perioperative period of PCI. These results revealed that nicorandil played a positive role in improving coronary microcirculation (13,14) in patients with STEMI, lowering the incidence of no-reflow or slow flow, reducing the infarct area of patients with AMI, and protecting ischemic myocardium (15,16). Furthermore, it has the vasodilator effect of nitrates, and can dilate the coronary artery and peripheral veins. In addition, it can inhibit oxidative stress in vascular endothelial cells (17), reduce the proliferation of vascular smooth muscle cells, and improve vascular endothelial function (18,19). Myocardial reperfusion injury further causes coronary microvascular dysfunction and subsequently, which leads to a series of adverse events. Nicorandil could specifically dilate small coronary arteries, improve microcirculation, reduce reperfusion injury (20,21).
However, the administration timing, administration method and medication duration of nicorandil in PPCI for STEMI patients remain inconclusive. Some meta-analyses have revealed that it remains unclear whether the effect of the reperfusion therapy of nicorandil on AMI is correlated with different patterns of administration and durations of medication (22,23). In most clinical trials, nicorandil was used through intracoronary injection immediately after the recanalization of the culprit vessel in a single time. The results of some clinical trials have revealed that a single use of intracoronary injection of nicorandil could improve the clinical curative effect. But other clinical trials have revealed that the intracoronary injection of nicorandil did not achieve the desired clinical curative effect (16,18). However, a single intracoronary injection of nicorandil has a short action time, and the improvement of the microcirculation of the myocardium cannot be completed in a short time. Therefore, we studied on the sequential nicorandil treatment, the intracoronary and peripheral venous use of nicorandil, on the microcirculation of STEMI patients. We referred to Lee et al. (24) that intracoronary injection of 2 mg nicorandil were given before CAG and stent implantation, a total of 4 mg. Recently, a clinical trial has also attempted to adopt intracoronary administration of 6 mg of nicorandil bolus (7), which had no significant effect on blood pressure. Because in this study we attempted coronary artery combined with peripheral intravenous administration, from safety considerations, 4 mg was given through the coronary artery, the speed and dosage of continuous peripheral intravenous administration observed the recommendation of drug instructions.
The result of this study revealed that, the TIMI blood flow and CTFC were superior in the sequential nicorandil group and intracoronary nicorandil group to those in the control group. This suggests that, coronary artery administration of nicorandil improves myocardial blood flow. However, there was no significant difference between these two groups. The reason may be that TIMI blood flow and CTFC reflex immediate myocardial blood flow. Patients in these two groups received intracoronary administration of nicorandil bolus, the time and dose of administration were the same. Therefore, there was little difference in the effects on immediate myocardial blood flow between the two. With the progression of the disease, patients in the sequential nicorandil group continued to receive nicorandil intravenously, and the differences in the proportions of patients with complete STR and advancing of enzyme peak time to within 12 h among three groups were statistically significant, where the two were superior in the sequential nicorandil group than in the intracoronary nicorandil group, and were superior in these two groups than in the control group; the CK-MB peak was lower in the sequential group than in the control group, and the difference in the CK-MB peak between the intracoronary nicorandil group and the control group was not statistically significant. By prolonging the duration of medication, perhaps sequential nicorandil group exhibited its advantages over intracoronary nicorandil group.
The above results reveal that nicorandil can improve myocardial microcirculation in patients with STEMI and achieve myocardial perfusion, and the effects of the different administration methods on the improvement of myocardial microcirculation are different. Furthermore, the effect of a single dose of intracoronary injection combined with peripheral intravenous injection was superior to that of a single dose of intracoronary injection.
It was also observed that in the evaluation of the prognosis of these patients, during hospitalization, the incidence of MACE successively decreased in the sequential nicorandil group, and the differences between each two groups were statistically significant. This suggests that nicorandil is helpful for improving the short-term prognosis of patients with AMI, and this was better in the sequential nicorandil group than in the intracoronary nicorandil group. The reason may be that in the sequential nicorandil group, the myocardial microcirculation improved better. Furthermore, the differences in LVEF among the three groups were not statistically significant. The possible reason may be that the sample size was small, and the follow-up time was short. The sample size and follow-up time can be further increased to further investigate the effect of nicorandil on the long-term prognosis of AMI patients in the future.
In the evaluation of medication safety, the differences in changes in perioperative blood pressure, heart rate and liver and kidney function among the three groups were not statistically significant. This suggests that nicorandil has good safety in clinical applications.
Our study showed that TIMI grade 3 blood flow rate, complete STR rate and myocardial enzyme peak time after PCI were worse than most published data. We considered the following possible reasons: (I) Patient visit time is too long. (II) At that time, the hospital did not set up a chest pain center. After the patient went to the hospital, he routinely received the CCU ward, and then went to the catheter room for interventional treatment, which led to the extension of DtoB (door to balloon) time. The average time from onset to blood vessel opening was close to 7 h, it may cause increased thrombus composition, more patients with slow blood flow. (III) Because of the higher age of the selected patients, there is no routine use of glycoprotein IIb/IIIa antagonists to reduce the risk of bleeding, it may cause insufficient antithrombotic activity and increase the risk of no reflow. (IV) Our hospital is located in the old city center of Hedong District, Tianjin City. The residents around us are mainly old, so there are many serious complications. In addition, our hospital is a comprehensive hospital. Patients with single disease will choose a special heart hospital within the same distance, and patients with complex disease will first choose our hospital after the onset. This caused most of the patients admitted to our department of cardiology to be older, with many complications. This is an important reason why the above-mentioned therapeutic indicators after PCI in this study will be worse than most published data. At present, our hospital has set up a chest pain center, which greatly shortens the time of door to balloon (D-to-B). We have plans to count the above indicators in the next study to see if they have been effectively improved.
In summary, the results of the present study suggest that the intracoronary and peripheral venous use of nicorandil is helpful to realize the revascularization of the infarct area, reduce the incidence of slow flow/no-reflow, and limits the infarct area of STEMI patients after PPCI. Compared with intracoronary use alone, its subsequent use can better improve myocardial microcirculation, increase myocardial blood flow perfusion, and improve short-term prognosis. The present study provides reference basis for the administration timing and medication scheme of nicorandil. Due to the small sample size and short observation period, its long-term curative effect and prognosis need to be determined through further clinical trials.
Acknowledgements
Funding: This study was supported by Tianjin Science and Technology Project (16ZXMJSY00200).
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: The protocol was approved by the ethics committee of Third Central Hospital of Tianjin. Informed consent was obtained from each patient or from their nearest relatives.
References
- Liang T, Liu M, Wu C, et al. Risk Factors for No-Reflow Phenomenon after Percutaneous Coronary Intervention in Patients with Acute Coronary Syndrome. Rev Invest Clin 2017;69:139-45. [Crossref] [PubMed]
- Anderson RD, Pepine CJ. The Coronary Microcirculation in STEMI: The Next Frontier? Eur Heart J 2015;36:3178-81. [Crossref] [PubMed]
- Fukushima Y, Kumita S, Tokita Y, et al. Prognostic Value of Myocardial Perfusion SPECT After Intravenous Bolus Administration of Nicorandil in Patients with Acute Ischemic Heart Failure. J Nucl Med 2016;57:385-91. [Crossref] [PubMed]
- Chen C, Fu X, Li W, et al. Intracoronary administration of anisodamine and nicorandil in individuals undergoing primary percutaneous coronary intervention for acute inferior myocardial infarction: A randomized factorial trial. Exp Ther Med 2015;10:1059-65. [Crossref] [PubMed]
- Isono T, Kamihata H, Sutani Y, et al. Nicorandil suppressed myocardial injury after percutaneous coronary intervention. Int J Cardiol 2008;123:123-8. [Crossref] [PubMed]
- Ito N, Nanto S, Doi Y, et al. High index of microcirculatory resistance level after successful primary percutaneous coronary intervention can be improved by intracoronary administration of nicorandil. Circ J 2010;74:909-15. [Crossref] [PubMed]
- Wang ZQ, Chen MX, Liu DL, et al. The effect on myocardial perfusion and clinical outcome of intracoronary nicorandil injection prior to percutaneous coronary intervention in ST-segment elevation myocardial infarction. Zhonghua Xin Xue Guan Bing Za Zhi 2017;45:26-33. [PubMed]
- Soukoulis V, Boden WE, Smith SC Jr, et al. Nonantithrombotic medical options in acute coronary syndromes: old agents and new lines on the horizon. Circ Res 2014;114:1944-58. [Crossref] [PubMed]
- Takabatake W, Noma T, Iwado Y, et al. Intracoronary administration of nicorandil during primary percutaneous coronary intervention: Impact on restoration of regional myocardial perfusion in reperfused myocardium during the subacute phase of myocardial infarction. Int J Cardiol Heart Vasc 2015;8:81-6. [Crossref] [PubMed]
- Thygesen K, Alpert JS, Jaffe AS, et al. Third universal definition of myocardial infarction. J Am Coll Cardiol 2012;60:1581-98. [Crossref] [PubMed]
- Tritto I, Zuchi C, Vitale S, et al. Therapy against reperfusion-induced microvascular injury. Curr Pharm Des 2013;19:4586-96. [Crossref] [PubMed]
- Jang HJ, Koo BK, Lee HS, et al. Safety and efficacy of a novel hyperaemic agent, intracoronary nicorandil, for invasive physiological assessments in the cardiac catheterization laboratory. Eur Heart J 2013;34:2055-62. [Crossref] [PubMed]
- Ito N, Nanto S, Doi Y, et al. Beneficial effects of intracoronary nicorandil on microvascular dysfunction after primary percutaneous coronary intervention: demonstration of its superiority to nitroglycerin in a cross-over study. Cardiovasc Drugs Ther 2013;27:279-87. [Crossref] [PubMed]
- Suematsu Y, Murasato Y, Miura S, et al. Safety and feasibility of high-dose administration of nicorandil before reperfusion therapy in acute myocardial infarction. Cardiovasc Interv Ther 2013;28:352-61. [Crossref] [PubMed]
- Ota S, Nishikawa H, Takeuchi M, et al. Impact of nicorandil to prevent reperfusion injury in patients with acute myocardial infarction: Sigmart Multicenter Angioplasty Revascularization Trial (SMART). Circ J 2006;70:1099-104. [Crossref] [PubMed]
- Wu M, Huang Z, Xie H, et al. Nicorandil in patients with acute myocardial infarction undergoing primary percutaneous coronary intervention: a systematic review and meta-analysis. PLoS One 2013;8:e78231. [Crossref] [PubMed]
- Ekeløf S, Jensen SE, Rosenberg J, et al. Reduced oxidative stress in STEMI patients treated by primary percutaneous coronary intervention and with antioxidant therapy: a systematic review. Cardiovasc Drugs Ther 2014;28:173-81. [Crossref] [PubMed]
- Umaru B, Pyriochou A, Kotsikoris V, et al. ATP-sensitive potassium channel activation induces angiogenesis in vitro and in vivo. J Pharmacol Exp Ther 2015;354:79-87. [Crossref] [PubMed]
- Chen Z, Chen X, Li S, et al. Nicorandil improves myocardial function by regulating plasma nitric oxide and endothelin-1 in coronary slow flow. Coron Artery Dis 2015;26:114-20. [Crossref] [PubMed]
- Hausenloy DJ, Yellon DM. Myocardial ischemia-reperfusion injury: a neglected therapeutic target. J Clin Invest 2013;123:92-100. [Crossref] [PubMed]
- Wang A, Chen F, Xie Y, et al. Protective mechanism of nicorandil on rat myocardial ischemia-reperfusion. J Cardiovasc Med (Hagerstown) 2012;13:511-5. [Crossref] [PubMed]
- Chen QJ, Yang YN, Ma YT, et al. Effectiveness of Nicorandil for reperfusion of acute myocardial Infarction: A Meta-Analysis. Chinese Journal of Evidence-Based Medicine 2012;12:1330-8.
- Niu X, Zhang J, Bai M, et al. Effect of intracoronary agents on the no-reflow phenomenon during primary percutaneous coronary intervention in patients with ST-elevation myocardial infarction: a network meta-analysis. BMC Cardiovasc Disord 2018;18:3. [Crossref] [PubMed]
- Lee HC, An SG, Choi JH, et al. Effect of intra-coronary nicorandil administration prior to reperfusion in acute ST segment elevation myocardial infarction. Circ J 2008;72:1425-9. [Crossref] [PubMed]


