Considerations for patients awaiting heart transplantation—Insights from the UK experience
Introduction
Heart transplantation has been a limited but gold standard surgical therapy for end-stage heart failure for over 20 years. During that time there have been major improvements in the overall treatment of heart failure with several medications and devices including implantable cardiodefibrillators and cardiac resynchronisation therapy each leading to independent improvements in survival. It is important to appreciate that the types of patients that will benefit from transplantation has changed over time as we learn to avoid transplantation in some and to overcome the perceived risks in others. There has been significant variation in the number of transplants being performed in different countries which has a marked effect on which patients are listed. Finally, over the last 10 years the use of ventricular assist devices (VADs) as a bridge to transplant has had an important impact on the type of patients that we are caring for on the transplant list.
Heart transplant numbers
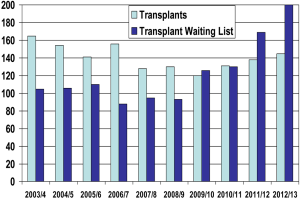
In the United Kingdom, the numbers of patients being transplanted over the last 10-15 years has undergone significant change (1). In the 10 years up to 2010 there was a near 50% fall in the number of adult transplants which compares to no change in the US and approximately a 5% fall in continental Europe over the same time period. The reasons why the UK performed so poorly over that time period are not entirely clear. Issues such as improved road traffic safety will have presumably affected all developed countries. Lack of resources with intensive care beds in the UK was one issue that was highlighted (2). This in addition to prioritisation of donation after cardiac death over donation after brain death is likely to be a significant factor with transplants other than cardiac increasing over the 10 years. Heart transplants at the present time cannot be used from donors after cardiac death, although we are attempting to address this particularly in paediatric patients. Whatever the reason, over the last 2 years there has been a subsequent recovery in numbers. There have been intensive efforts in the UK to identify and utilise all suitable donors that become available. As shown in Figure 1 there have also been significant increases in the number of patients listed for transplantation, which presumably will increase the chances of recipients finding a suitable donor.
Ventricular assist devices (VADs)
One issue that has increased the numbers on the heart transplant list is patients with VADs. In patients in whom it is felt that the wait time for a transplant will be too long (size, HLA antibody status, blood type, unstable heart failure) VADs implanted as a bridge to transplant can now reverse end-stage heart failure so that these patients are discharged home and live relatively normal lives. In some patients who are not suitable transplant candidates (elevated pulmonary vascular resistance, cachexia, renal insufficiency) a period of support on a VAD may improve their suitability for transplantation (bridge to candidacy). This has resulted in large numbers of patients with VADs, many of whom are at home and stable, being on the transplant list. This has increased transplant list numbers well beyond the numbers of performed transplants (3). However, life on a VAD is not without potential serious complications so that by 2 years, most patients have been re-admitted to hospital with a serious complication (4).
Treatment of heart failure and benefits from transplantation
Heart failure treatment has radically changed over the last 20 years and the days when patients were transplanted without any exposure to life prolonging drugs are now long past. How this impacts on our decisions to list patients for transplantation was very elegantly shown in 2007 by Lietz and Miller (5). In an analysis of US transplant listings from 1990 to 2005 they showed that the survival of urgently listed patients in the most recent era remains improved with transplantation compared to those not getting transplanted. However the survival of non-urgent candidates in the later era not transplanted versus those transplanted were very similar. These data illustrate that contemporary medical therapies for patients with advanced heart failure who are ambulatory are as good as transplantation and argues against listing such subjects. The authors did find some potential exceptions to this, such as patients who were not urgently listed but who did have a high risk of dying—including those with restrictive cardiomyopathies, secondary cardiomyopathies, and congenital heart disease.
Adult congenital heart disease
Adults with congenital heart disease represent an important sub group of patients that might derive benefit from transplant which have up until now been underrepresented. There is an increasing population of children with congenital heart disease surviving to adulthood as a result of improved diagnosis, medical and surgical therapies, which has been estimated as increasing by 1,600 adult cases per year in the UK (6). The proportion of these patients that go on to develop refractory heart failure, without any further medical or surgical treatment options, is unlikely to be changed from previous eras and therefore the total number for whom transplantation is the only option is increasing. Indeed the mortality of these patients at a young age from causes amenable to transplant and the increasing burden of hospital admission with heart failure are well described (7,8). International Society of Heart and Lung Transplantation (ISHLT) data (9) suggests that despite worse early mortality compared to other diagnoses undergoing heart transplant, adult congenital heart disease patients have a superior unconditional survival to all groups at 10 years which continues thereafter. Despite this early mortality, we have shown that with increasing experience in a single centre 5 years survival can increase from 50% to 69% (10) and our current 5-year survival [2000-2014] for all diagnoses is 75% and 79% for the Fontan subgroup. The perceived risk of transplanting these patients potentially disadvantages them with regards to access to cardiac transplantation with one of the most important triggers in the UK for a unit’s program to be reviewed for poor outcomes being survival at 90 days. We have identified that morbidity and mortality of these patients may be improved with transplant relatively earlier in the disease process in particular for patients who are not suitable for VADs. However, early listing needs to become synonymous with earlier transplant to achieve this, which may not be possible with current listing criteria (see below) and the size of the list. The complexity of these patients means that these types of transplants are best concentrated in centres that can do sufficient numbers to achieve acceptable results (1).
Heart allocation: urgent vs. non urgent
In the UK delegates from all adult heart transplant centres have recently reviewed our heart allocation policies to ensure that we maximise the benefit from this scarce commodity but also to ensure that the organs are fairly distributed throughout the country to the sickest patients. The underlying principle of heart allocation that was adopted states ‘the principle aim of donor heart allocation is to improve survival in selected patients with advanced heart failure prioritising the sickest patients at greatest risk of dying, while ensuring risk of transplantation is within acceptable limits’. The background to the review was that in 2011/12 56% of heart transplants were performed from the urgent list compared with 28% in 2006/7. The UK working group were in favour of the increased use of the urgent system as it prioritised the sickest patients. Data showed that urgent patients transplanted had a one year survival of 84%, compared to non-urgent patients of 77%, suggesting that urgent transplant outcomes are at least as good as non-urgent transplants. Furthermore, deaths on the transplant waiting list have fallen over recent years (22 for 2006/7 compared with 13 and 16 in the most recent two financial years up to 2011/12). This, however, does not mean that fewer patients now require transplantation, as there has been a corresponding large increase in patients dying with VADs whom have never been on the list (implanted as a bridge to decision) or were on the list at some stage in their clinical course. Thus, the high risk patients, who are unable to wait for transplant need more immediately available advanced heart failure surgery are undergoing VAD insertion as opposed to a transplant.
To-date, there were two priority statuses on the transplant list—urgent and routine. To be on the urgent list essentially meant being in hospital on inotropes, an intra-aortic balloon pump or with a short term mechanical circulatory support device. All other patients were on the routine list. The urgent listed patient had access to suitable donors from throughout the UK as opposed to just the local retrieval zone in the area of the transplant centre for routine transplants. The new proposals were designed to simplify the criteria, and to expand on the urgent listing categories to produce a super urgent category. It was recognised that the number of urgent or super urgent transplants would increase, but as this prioritised the sickest patients, and produced at least as good results (or better) this was felt to be in the interests of our sickest patients. The following criteria were adopted for heart allocation (though have not yet been implemented):
- Super urgent listing: (i) patients on short term mechanical circulatory support which includes short term VADs (a device with which a patient cannot be discharged to home), extra-corporeal membrane oxygenation, or an intra-aortic balloon pump; (ii) a patient meeting criteria for urgent listing that is not suitable for a long term left VAD. Access for this latter type of patient will be approved by an arbitration group;
- Urgent listing: inpatient dependent on intravenous inotropes. Inotropes cannot be discontinued without grave risk of harm to patient;
- Urgent listing of long term VAD patients: (i) right ventricular failure dependent on intravenous inotropes; (ii) recurrent systemic infection related to the VAD (does not have to be inpatient); (iii) other VAD issues including recurrent or refractory VAD thrombosis (after appeal to the arbitration group, see above). This category does also not have to be inpatient;
- Exceptionally sick patients referred to the arbitration group. In exceptional circumstances, patients with risk of dying >50% at 30 days without a transplant, that do not meet either urgent or super urgent listing criteria above can be placed on one of these lists after approval by majority of delegates from other transplant centres.
Further changes in transplant listing criteria needed
Whereas the criteria listed above hopefully represent an improvement in allocation of donor hearts to benefit the sickest patients, there are still significant issues that need to be addressed. First and foremost are the increasing numbers of patients on the list, many of whom have no realistic chance of being transplanted. If that is accepted as a target for future change, then the next question is how to reduce the size of the waiting list. To derive a significant benefit from transplantation, ideally patients should be at high risk of dying without transplantation and following transplantation have a good chance of prolonged survival. The uncertainty arises from the fact that those factors that predict death on the waiting list, may also predict death after transplantation—such as age, non-compliance, and renal dysfunction. In addition scores which assess acute survival benefit potentially leave subgroups of patients whose risk from transplant is likely to increase significantly, at times to unacceptable levels, if their disease process is allowed to progress without transplant. In particular this applies to patients in whom a VAD cannot be easily deployed or is less effective such as in single ventricle adult congenital patients, those with restrictive cardiomyopathy whose pulmonary vascular resistance is increasing and patients with primarily right ventricular disease. Two recent studies perhaps help us understand how we could reduce the size of the list. Singh and colleagues (11) have looked at risk of dying on the transplant waiting list and the subsequent risk of transplantation. They created models to determine risk of death on the waiting list and patients were divided into deciles based on these. Models were also developed to determine risk of death after transplantation. The findings were that only the highest risk deciles showed a survival benefit with transplantation, findings which support those of Lietz and Miller (5) described above suggesting that less severe degrees of advanced heart failure may not benefit from transplantation.
The concept to balance waiting list mortality and post transplant outcomes has been used in the lung allocation score system (12). Smits and colleagues (13) from Eurotransplant with the aim to move towards a cardiac allocation score have assessed both heart failure survival models [heart failure survival score (HFSS) (14), the Seattle heart failure model (SHFM) (15), and the interagency registry for mechanically assisted circulatory support (4)], and the post-transplant index for mortality prediction after cardiac transplantation (IMPACT) (16) for predicting mortality. Their results show that in non-VAD patients the HFSS, SHFM and IMPACT provide accurate risk which could be considered as the basis for a new cardiac allocation score. This is a step in the right direction, but the absence of prediction in VAD patients still leaves a large number of patients with whom decisions about listing are unclear. In an accompanying editorial Dr. Stevenson (3) argued strongly that we needed to trim the waiting list. Her proposals to do this included not actively listing those patients who are non urgent, and to use benefit scores to decide which patients go on the list rather than listing by priority.
Proposal for transplant listing criteria
Incorporating the above evidence new transplant listing criteria can be constructed. However, there are two major limitations with any bold new move in that direction: (I) the absence of a benefit score for patients with VADs; and (II) the absence of any benefit score for patients with adult congenital heart disease. The limitations of current urgent listing criteria for patients not suitable for the therapies which allow urgent listing, particularly adult congenital patients is well recognised (17). Nevertheless a new system might incorporate the following (assuming the above limitations can be addressed): (I) only list patients that can derive significant long term survival benefit from transplantation—which includes adult acquired heart failure, VADs and adult congenital heart disease. For both VADs and adult congenital heart disease we need distinct benefit scoring systems. These must incorporate the risk of dying from heart failure, the risk of a serious VAD complication, and the risk that delaying listing for transplant can have a detrimental effect on transplant eligibility coupled with the risk of subsequent transplantation; (II) in patients with advanced heart failure though ambulatory and at home the majority would be ‘registered’ at a transplant centre, though not actively on the transplant list. This also will include some stable patients with VADs deemed at low risk for serious complications. The ultimate goal would be by restricting listing we can transplant those patients that need it in a reasonable time frame with acceptable risks and for those that do not urgently need transplantation to adopt a watchful waiting policy. This can have considerable knock on benefits. It is a more honest and transparent policy for our patients—if you are on the transplant list we intend to transplant you—rather than being on a list with a low expectation of ever getting transplanted. Bridge to transplant VADs may become less frequent, freeing up scarce resources for destination therapy in non-transplant candidates. Patients with adult congenital heart disease can at last be accommodated in a scheme that has been biased towards acquired heart failure since its inception. Transplantation is a scarce though wonderful resource. It is our responsibility to see that it is used in the fairest and most effective way.
Acknowledgements
Disclosure: The authors declare no conflict of interest.
References
- Macgowan GA, Parry G, Schueler S, et al. The decline in heart transplantation in the UK. BMJ 2011;342:d2483. [PubMed]
- Fabre J, Murphy P, Matesanz R. Presumed consent: a distraction in the quest for increasing rates of organ donation. BMJ 2010;341:c4973. [PubMed]
- Stevenson LW. The urgent priority for transplantation is to trim the waiting list. J Heart Lung Transplant 2013;32:861-7. [PubMed]
- Kirklin JK, Naftel DC, Kormos RL, et al. Fifth INTERMACS annual report: risk factor analysis from more than 6,000 mechanical circulatory support patients. J Heart Lung Transplant 2013;32:141-56. [PubMed]
- Lietz K, Miller LW. Improved survival of patients with end-stage heart failure listed for heart transplantation: analysis of organ procurement and transplantation network/U.S. United Network of Organ Sharing data, 1990 to 2005. J Am Coll Cardiol 2007;50:1282-90. [PubMed]
- Wren C, O'Sullivan JJ. Survival with congenital heart disease and need for follow up in adult life. Heart 2001;85:438-43. [PubMed]
- Verheugt CL, Uiterwaal CS, van der Velde ET, et al. Mortality in adult congenital heart disease. Eur Heart J 2010;31:1220-9. [PubMed]
- Opotowsky AR, Siddiqi OK, Webb GD. Trends in hospitalizations for adults with congenital heart disease in the U.S. J Am Coll Cardiol 2009;54:460-7. [PubMed]
- Lund LH, Edwards LB, Kucheryavaya AY, et al. The Registry of the International Society for Heart and Lung Transplantation: Thirtieth Official Adult Heart Transplant Report--2013; focus theme: age. J Heart Lung Transplant 2013;32:951-64. [PubMed]
- Irving C, Parry G, O'Sullivan J, et al. Cardiac transplantation in adults with congenital heart disease. Heart 2010;96:1217-22. [PubMed]
- Singh TP, Milliren CE, Almond CS, et al. Survival benefit from transplantation in patients listed for heart transplantation in the United States. J Am Coll Cardiol 2014;63:1169-78. [PubMed]
- Smits JM, Nossent GD, de Vries E, et al. Evaluation of the lung allocation score in highly urgent and urgent lung transplant candidates in Eurotransplant. J Heart Lung Transplant 2011;30:22-8. [PubMed]
- Smits JM, de Vries E, De Pauw M, et al. Is it time for a cardiac allocation score? First results from the Eurotransplant pilot study on a survival benefit-based heart allocation. J Heart Lung Transplant 2013;32:873-80. [PubMed]
- Aaronson KD, Schwartz JS, Chen TM, et al. Development and prospective validation of a clinical index to predict survival in ambulatory patients referred for cardiac transplant evaluation. Circulation 1997;95:2660-7. [PubMed]
- Levy WC, Mozaffarian D, Linker DT, et al. The Seattle Heart Failure Model: prediction of survival in heart failure. Circulation 2006;113:1424-33. [PubMed]
- Weiss ES, Allen JG, Arnaoutakis GJ, et al. Creation of a quantitative recipient risk index for mortality prediction after cardiac transplantation (IMPACT). Ann Thorac Surg 2011;92:914-21; discussion 921-2. [PubMed]
- Gelow JM, Song HK, Weiss JB, et al. Organ allocation in adults with congenital heart disease listed for heart transplant: impact of ventricular assist devices. J Heart Lung Transplant 2013;32:1059-64. [PubMed]


