
Aortic balloon occlusion technique in total arch replacement with frozen elephant trunk after thoracic endovascular aortic repair
Introduction
Thoracic endovascular aortic repair (TEVAR) had been increasingly applied in the treatment of thoracic aortic diseases (1). However, there were still some unavoidable early and late complications, including retrograde type A aortic dissection (RTAD), endoleak, paraplegia (2,3). Therefore, some patients had to undergo secondary open arch surgery due to these complications. And total arch replacement (TAR) with frozen elephant trunk (FET) was a preferred surgical strategy in this situation, especially in patients with RTAD (4) and type Ia endoleak (5). However, TAR with FET conventionally required the use of hypothermic circulatory arrest (HCA), which was associated with many adverse complications (6). For patients with prior TEVAR, the arch pathology and anatomy was more complicated, and it took more time to create a distal anastomosis plane. In some condition, the proximal part of the stent was anchored onto the aortic wall, and we had to remove the proximal part of the TEVAR stent by a wire scissor. Therefore, the HCA duration of patients undergoing TAR with FET after TEVAR was much longer than that of patients without prior TEVAR. And it resulted in a higher incidence of complications including paraplegia, renal failure and gastrointestinal dysfunction.
And the aortic balloon occlusion technique had been introduced in TAR with FET by our colleagues (7). The aortic balloon with the sheath was deployed into the descending aorta after FET implantation. Once the balloon was inflated and fixed, perfusion of the lower body was resumed through the femoral artery. Thus, the HCA duration was decreased to approximately 5 minutes with the aortic balloon occlusion technique, much shorter than the previous time of 20–25 minutes with the conventional technique. So, we had applied the aortic balloon occlusion technique in some patients when performing TAR with FET after prior TEVAR. In this study, we aimed to present our experiences in TAR with FET after prior TEVAR, and compare the clinical outcomes between the aortic balloon occlusion technique and the conventional technique.
Methods
Patients selection
Aortic surgery database of Beijing Fuwai Hospital was reviewed retrospectively. About 1,300 patients underwent TEVAR in our hospital between January 2016 and December 2019. And 76 patients received aortic reoperation after TEVAR during this period. Among them, 30 patients who received TAR with FET after TEVAR were enrolled in this study. The aortic balloon occlusion technique was applied in 9 patients, and the conventional technique in 21 patients. The study protocol was approved by the Institutional Ethics Committee of Fuwai Hospital, and written informed consent was obtained from all patients. The mean age of patients in the balloon occlusion group was older than that of patients in the conventional group (60.2±6.1 vs. 51.0±9.2 years, P=0.011). The characteristics of the patients are summarized in Table 1.
Full table
Among 30 patients, 10 patients received prior TEVAR in our hospital, while 20 patients received prior TEVAR in outside hospital. The indications for prior TEVAR included type B aortic dissection or hematoma, and penetrating aortic ulcer. The Left subclavian artery (LSA) was covered in 26 patients during TEVAR. And 10 patients also received additional procedures during TEVAR, including chimney graft implantation, fenestration technique, supra-aortic bypass and LSA coil-spring occlusion. And the median time interval from TEVAR to reoperation was 9 months (0–168 months). The details of prior TEVAR are shown in Table 2.
Full table
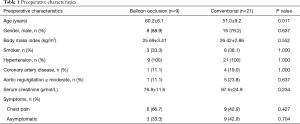
In the balloon occlusion group, the indications for TAR with FET included RTAD in 6 patients, arch false aneurysm in 1 patient and type Ia endoleak in 2 patients. Figure 1 shows the preoperative computed tomographic scan of patients with RTAD, type Ia endoleak and arch false aneurysm, respectively. Among 21 patients with the conventional technique, the indications for TAR with FET included RTAD in 6 patients, new ascending dissection in 3 patients, type Ia endoleak in 9 patients and type II endoleak in 3 patients. The details are exhibited in Table 3.

Full table
Surgical technique
The operative procedure had been previously described by our colleagues (7). The operation was performed under general anesthesia. A median sternotomy was performed in all patients. The right axillary artery, the femoral artery and the right atrium were cannulated for the cardiopulmonary bypass (CPB), and the right axillary artery was used for selective antegrade cerebral perfusion (SCP). During the cooling phase, the aortic root procedure or other surgery could be performed if necessary.
When the nasopharyngeal temperature reached about 22–28 °C, SCP was initiated with the flow of 5–10 mL/kg/min via the right axillary artery cannula. The aortic arch was dissected and transected at the tip of bare spring. Then we inspected the arch and proximal descending aorta carefully. The aortic wall trauma caused by the proximal bare spring was identified and eliminated under direct inspection. The proximal bare spring of the stent was removed by a wire scissor. This procedure could protect the descending aorta from being injured by steel chips, and the residual TEVAR stent could also enhance the effect of FET when performing distal anastomosis since it could be in the middle of the sandwich structure. In the patients who had received chimney graft implantation during prior TEVAR, the chimney grafts were totally removed.
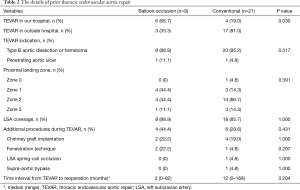
The FET (Cronus, MicroPort Endovascular Shanghai Co, Ltd, China) was inserted into the true lumen of the descending aorta under direct inspection. For patients with the aortic balloon occlusion technique, the aortic balloon (Coda Balloon Catheter, Cook Incorporated, Bloomington, IN, USA) with the sheath (W.L. Gore & Associatates, Inc., Flagstaff, AZ, USA) was deployed into the FET, and inflated with saline of about 40–45 mL (Figure 2). The inflated balloon was compressed by the sheath to avoid displacement. Then SCP ended and the lower body perfusion was resumed through the femoral artery. During the occlusion period, we performed the distal anastomosis meticulously. The distal end of four-branch vascular graft (Terumo, Vascutek Limited, Renfrewshire, UK) was anastomosed with the descending aorta, the residual TEVAR graft and the FET. Once distal arch anastomosis completed, the balloon with sheath was retracted from the FET. For patients with the conventional technique, the distal anastomosis was performed under HCA and SCP. Then the left carotid artery was anastomosed to one branch of the vascular graft, and rewarming began. Subsequently, the proximal end of the vascular graft was anastomosed to the ascending aorta, and the other two branches were anastomosed to the LSA and the innominate artery, respectively.
Statistical analysis
Continuous variables were expressed as median (range) or mean ± SD, and categorical variables as number (percentage). Continuous variables were compared with Student t test or Wilcoxon rank test. Categorical variables were compared with Fisher exact test. A P value of less than 0.05 was considered statistically significant. All statistical analyses were performed with SPSS 21.0 (SPSS Inc., Chicago, IL).
Results
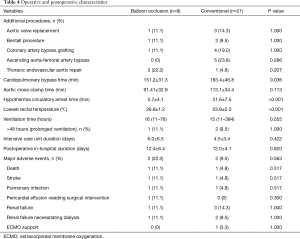
The additional procedures included aortic valve replacement, Bentall procedure, coronary artery bypass grafting, ascending aorta-femoral artery bypass and TEVAR. The patients with the balloon occlusion technique had shorter CPB time than patients with the conventional technique (151.2±31.3 vs. 183.4±46.8 min, P=0.036). The aortic-clamp time was also shorter in the balloon occlusion group, but without significant difference. The HCA duration was significantly decreased in the balloon occlusion group (5.7±4.1 vs. 21.6±7.5 min, P<0.001). The lowest rectal temperature was higher in the balloon occlusion group (26.9±1.2 vs. 23.8±2.0 °C, P<0.001).
The median ventilation time was 15.5 h (range, 11–394 h). And 3 patients had prolonged ventilation (>48 h). The mean durations in the intensive care unit and in the hospital postoperatively were 5.0±4.4 and 12.1±4.7 days, respectively. A total of 2 patients (6.7%) died in this study, 1 patient in the balloon occlusion group and 1 patient in the conventional group. The patient in the balloon occlusion group was a 62-year-old female. She was diagnosed with RTAD immediately after TEVAR according to the intraoperative aortic angiography, and underwent TAR with FET urgently. She suffered pulmonary infection and prolonged ventilation. She developed renal failure necessitating dialysis on the first postoperative day, a stroke on the 14th postoperative day and recovered from the stroke 4 days later. She was discharged and transferred to another hospital on the 22nd postoperative day and died 1 day later for an unknown cause. The other patient in the conventional group was a 56-year-old female. She underwent TAR with FET, and coronary artery bypass grafting. After surgery, she suffered pulmonary infection, renal failure necessitating dialysis, and myocardial dysfunction necessitating extracardiac membrane oxygenation. Severe stroke also occurred to her. And she died on the 17th postoperative day.
The major adverse events were defined as death, stroke, pulmonary infection, pericardial effusion needing surgical intervention, renal failure necessitating dialysis, extracorporeal membrane oxygenation support. A total of 4 patients (13.3%) suffered major adverse events. No significant differences in the incidence of major adverse events, and the mortality were noted between the two groups. Additionally, 1 patient in the balloon occlusion group also suffered left recurrent laryngeal nerve injury. The operative and postoperative details are shown in Table 4.
Full table
Follow-up was available in all 28 survivors. The mean follow-up time was 25.4±13.0 months (range, 2–48 months). No late death, aortic reoperation and complications occurred during follow-up.
Discussion
TEVAR was first reported in 1988 and had got widespread acceptance in the management of type B aortic dissection and other descending aortic diseases (1). However, TEVAR was also associated with some early and late complications, including RTAD, endoleak, paraplegia (2,3). Many patients with these complications or other new arch pathologies required secondary arch operation (8). And TAR with FET was the preferred strategy. However, reports regarding TAR with FET after prior TEVAR were especially limited. Since 2017, we had introduced the aortic balloon occlusion technique in our center when performing TAR with FET. We also applied this technique in some patients with prior TEVAR. The current report summarized our experiences in TAR with FET after prior TEVAR, and compared clinical outcomes between the aortic balloon occlusion technique with the conventional technique.
TAR with FET had been widely applied in the management of type A aortic dissection, arch aneurysm and some complicated type B aortic dissection (9). Although HCA with SCP had been confirmed to be a safe and effective method of cerebral protection (10), TAR with FET was still a high-risk procedure (9). TAR with FET in patients with prior TEVAR carried higher risks because of more complicated arch anatomy and pathology. The bare end of the stent was rigid and it might injure the aortic inner surface and adventitia. And the persistent contact of the stent graft with the aortic wall and the adventitia injury would also induce inflammatory reaction and enhance local adhesion. Therefore, it was harder to dissect the aortic arch during operation. And the HCA duration prolonged accordingly, which was associated with a higher incidence of complications including paraplegia, renal failure and gastrointestinal dysfunction.
In our center, we removed the proximal part of the TEVAR stent by a wire scissor and eliminate the pathological aortic wall totally to create a stable distal anastomosis plane. The removal of the proximal bare spring could also prevent postoperative anastomotic bleeding or leak, and aortic injury in the future. And we only remove the proximal part of the stent. The residual stent could enhance the effect of FET since it was in the middle of the sandwich structure. The steel wires of bare spring were interlaced, and the disruption of one steel wire can result in a drop of adjacent steel wire. It was a devastating condition if the steel wire fall into the aortic lumen. So, when cutting the steel wires of bare spring, we must hold the target steel wire with forceps tightly and inspect the bare spring carefully to prevent the drop of steel wires. The removal of the proximal bare spring would consume extra time and prolong the HCA duration further.
According to our previous experiences, the aortic balloon occlusion technique could shorten the HCA duration, and provide protective effects on the central nervous, hematological and visceral systems (7). Therefore, we applied the aortic balloon occlusion technique in TAR with FET after prior TEVAR to decrease the HCA duration. During the occlusion period, we can perform the distal arch anastomosis without HCA. In some circumstances, we would reduce the arterial flow rate if the retrograde flow from the distal end was tremendous to disturb the distal anastomosis.
The aortic balloon occlusion technique had some advantages over the conventional technique when performing TAR with FET, especially in patients with prior TEAVR. First, it could shorten the HCA duration significantly and decrease the incidence of related complications accordingly. In this study, the HCA duration with the aortic balloon occlusion technique could be decreased from 21.6±7.5 to 5.7±4.1 min. But we did not notice a significant improvement in the mortality and the incidence of major adverse events with this technique. The incidence of renal failure did not show a difference between the two groups. Considering older age in the balloon occlusion group, we speculated that this technique might have some protective effects on renal function and overall morbidities. Second, because the HCA had ended during the occlusion period, we can perform the distal arch anastomosis carefully and meticulously with no hurry, which can make sure the anastomosis quality and reduce the risk of anastomotic bleeding. And we are confident that this technique is suitable for some junior surgeons or can be spread into some rural hospitals. Third, with the aortic balloon occlusion technique, the lowest rectal temperature during the operation could be set higher, because the HCA duration had been decreased significantly. In this study, the lowest rectal temperature with this technique had been increased from 23.8±2.0 to 26.9±1.2 °C. Then the cooling time and rewarming time of the CPB will also be shortened, and the CPB time will be decreased accordingly. The CPB time with this technique had been shortened from 183.4±46.8 to 151.2±31.3 min. Therefore, the coagulative complications will decrease considering the elevated temperature during the CPB and the shortened CPB time.
All patients had received successful operation, with the mortality of 6.7%. The follow-up outcome was optimal among all the survivors. We could conclude that TAR with FET was a safe, effective approach in patients with prior TEVAR.
Our study has some limitations. The study is restricted by its respective nature. Although we had compared the aortic balloon occlusion technique with the conventional technique in this study, the result was infected by the small sample size.
Conclusions
Although with some technical difficulty, TAR with FET was still a safe and effective procedure in patients with prior TEVAR, with satisfactory early and late outcomes. And the aortic balloon occlusion technique could be applied in patients with prior TEVAR when performing TAR with FET. This technique could decrease the HCA duration and the CPB time, and it may have protective effects on renal function and hematological system. Further study is needed to prove the advantage of this technique, especially in patients with prior TEVAR.
Acknowledgments
Funding: This work was supported by Beijing Municipal Science and Technology Commission Capital characteristic clinic project [Z181100001718197].
Footnote
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/jtd.2020.03.70). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The study protocol was approved by the Institutional Ethics Committee of Fuwai Hospital (No. 2018-1069) and all patients provided written informed consent prior to recruitment.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Nienaber CA, Clough RE. Management of acute aortic dissection. Lancet 2015;385:800-11. [Crossref] [PubMed]
- Eggebrecht H, Thompson M, Rousseau H, et al. Retrograde ascending aortic dissection during or after thoracic aortic stent graft placement: insight from the European registry on endovascular aortic repair complications. Circulation 2009;120:S276-81. [Crossref] [PubMed]
- Kpodonu J, Preventza O, Ramaiah VG, et al. Retrograde type A dissection after endovascular stenting of the descending thoracic aorta. Is the risk real? Eur J Cardiothorac Surg 2008;33:1014-8. [Crossref] [PubMed]
- Li B, Pan XD, Ma WG, et al. Stented elephant trunk technique for retrograde type A aortic dissection after endovascular stent graft repair. Ann Thorac Surg 2014;97:596-602. [Crossref] [PubMed]
- Qi RD, Zhu JM, Liu YM, et al. The repair of a type Ia endoleak following thoracic endovascular aortic repair using a stented elephant trunk procedure. J Thorac Cardiovasc Surg 2018;155:1391-6. [Crossref] [PubMed]
- Shrestha M, Martens A, Kaufeld T, et al. Single-centre experience with the frozen elephant trunk technique in 251 patients over 15 years. Eur J Cardiothorac Surg 2017;52:858-66. [Crossref] [PubMed]
- Sun X, Guo H, Liu Y, et al. The aortic balloon occlusion technique in total arch replacement with frozen elephant trunk. Eur J Cardiothorac Surg 2019;55:1219-21. [Crossref] [PubMed]
- Roselli EE, Abdel-Halim M, Johnston DR, et al. Open aortic repair after prior thoracic endovascular aortic repair. Ann Thorac Surg 2014;97:750-6. [Crossref] [PubMed]
- Martens A, Beckmann E, Kaufeld T, et al. Total aortic arch repair: risk factor analysis and follow-up in 199 patients. Eur J Cardiothorac Surg 2016;50:940-8. [Crossref] [PubMed]
- Cefarelli M, Murana G, Surace GG, et al. Elective Aortic Arch Repair: Factors Influencing Neurologic Outcome in 791 Patients. Ann Thorac Surg 2017;104:2016-23. [Crossref] [PubMed]


