
The association between neutrophil-to-lymphocyte count ratio and mortality in septic patients: a retrospective analysis of the MIMIC-III database
Introduction
Sepsis, a serious and life-threatening syndrome designated as immune disorder and organ dysfunction subsequent to the host response to infection, is notoriously known for its favor to patients in the intensive care unit (ICU) (1). A great leap that had been made over these years in the field of critical care medicine though, it was only contributed to a small step in lowering the mortality rate of septic patients which is still hovering at around 30% nowadays (2). Therefore, it’s crucial for doctors to predict the prognosis of septic patients.
In 2001, Zahorec et al. were first to propose that neutrophil-to-lymphocyte count ratio (NLCR) could be used as an additional infection marker in clinical practice, and suggesting NLCR as an indicator of disease severity as well (3). NLCR boasts its simplicity, cost-efficiency and feasibility compared with many other previously proposed biomarkers, making which promising for clinicians to diagnose through (4).
Accumulating evidence shows that NLCR can be an independent predictor of poor survival in patients with tumor and cardiovascular disease (5,6). However, no consensus has been reached on the relationship between NLCR level and clinical prognosis of patients with sepsis up till now. One research found that higher NLCR measured at the time of admission to ICU was significantly related to higher 28-day mortality in either unselected critically ill patients or non-septic patients but not in patients with sepsis (7). Whereas several other studies linked NLCR to short- or long-term death of sepsis (8-12).
Accordingly, the clinical value of NLCR in patients with sepsis remain controversial. In this study, we obtained a large quantity of patient data from Medical Information Mart for Intensive Care III (MIMIC-III) database to analyze whether NLCR could be an independent predictor of all-cause mortality in adult septic patients.
Methods
Database
All data used in the study was extracted from the MIMIC-III version 1.4 (v1.4) which is a large, single-center, publicly available critical care database (13). It includes unidentified health-related data of more than 60,000 ICU stays at Beth Israel Deaconess Medical Center (BIDMC) from June 2001 to October 2012. MIMIC-III was built by researchers at the Massachusetts Institute of Technology (MIT) Laboratory for Computational Physiology and collaborating research groups. To get access to the database, we complete the course “Protecting Human Research Participants” at the website of National Institutes of Health and obtained the certification (Record ID: 28006489). The project was approved by the institutional review boards of the MIT and BIDMC and was granted a waiver of informed consent.
Patients population
Adult patients (≥18 years old) from medical ICU (MICU) and surgical ICU (SICU) with a diagnosis of sepsis defined by ICD-9-based Angus implementation (14) were selected from MIMIC-III v1.4 using computer code. Worth noticing, only first ICU admission of each patient with exact lymphocyte and neutrophils record within 24 hours after ICU admission were selected into final cohort. Patients unable to meet the inclusion criteria above were excluded.
Data extraction
Structured Query Language (SQL) and PostgreSQL tools (version 9.6) were used for data extraction. Data of following aspects were extracted directly or calculated with the data from the database: age, gender, ethnicity, first care unit, comorbidities, laboratory parameters, the time of patients’ getting into and out of hospital and ICU and date of dead of patients. The comorbidities included atrial fibrillation (AFIB), coronary artery disease (CAD), congestive heart failure (CHF), diabetes, malignancy, chronic renal disease, liver disease. The laboratory parameters included hemoglobin, platelet, sodium, potassium, bicarbonate, chloride, creatinine, white blood cell counts, lymphocyte counts and neutrophil counts. Furthermore, the sequential organ failure assessment (SOFA) score (15) and simplified acute physiology score II (SAPSII) (16) were calculated for each patient with the SQL code provided by Johnson et al. (17). NLCR was computed based on the first laboratory parameters if there were more than one test of laboratory parameters mentioned above within 24 hours as a ratio of neutrophil/lymphocyte value. Patients were categorized into four groups (from the first quartile to the fourth quartile) according to the quartile of the initial NLCR value within 24 hours after ICU admission. The outcomes of our study included 7- and 28-day all-cause mortality, mortality in the ICU, in-hospital mortality and length of ICU stay.
Statistical analysis
Baseline characteristics of all patients were stratified by NLCR quartiles. Continuous variables were presented as median and interquartile range (IQR) and Wilcoxon rank-sum test was used for comparisons between groups. Categorical data were expressed as absolute values and percentages and were compared using chi-square test or Fisher’s exact test as appropriate. We compared the survival rates using log-rank tests and present the results as Kaplan-Meier curves. The outcomes data were compared with chi-square test or Wilcoxon rank-sum test as appropriate based on NLCR quartiles. Multivariable cox proportional hazards models were constructed to determine the independent effect of the quartiles of NLCR on 28-day mortality. Variables with P<0.05 in the univariate analysis were further incorporated into multivariate Cox proportional hazard models. Variables were selected by a method of Forward: LR. The probability for stepwise is 0.05 for entry and 0.10 for removal. The results were express as hazard ratios (HRs) with 95% confidence intervals (CIs). To evaluate the accuracy of the NLCR in predicting 28-day mortality of sepsis, receiver operating characteristic (ROC) curves were constructed and Youden’s J Indexes were calculated for the initial NLCR as well as SOFA score and SAPS II score. The optimal cut-off value of NLCR is determined by the highest Youden’s J Index.
Subgroup analyses were employed to evaluate the association between the NLCR and 28-day all-cause mortality, including age, gender, comorbidities, laboratory parameters, SOFA score and SAPSII score. The data were analyzed with SPSS software (v16.0; IBM, Armonk, NY). A two-tailed P<0.05 was considered statistically significant.
Results
Population characteristics
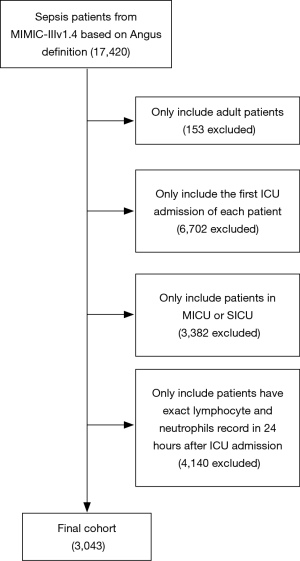
A total of 3,043 patients with complete data of neutrophil and lymphocyte available within 24 hours after ICU admission were included in this study. Complete designated endpoint data was available in all included patients. The procedure of data selection according to the criteria mention above is presented in Figure 1.
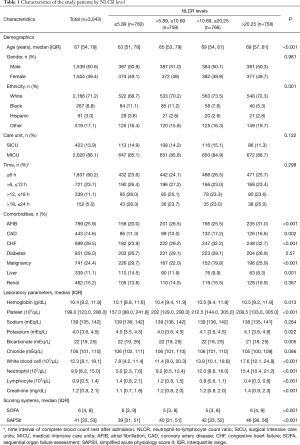
Baseline characteristics of the study population are shown in Table 1. The median age of the entire cohort was 67 years old (IQR, 54 to 79). Fifty-point-six percent of the patients were male. A vast majority of the patients were Caucasians (defined as “white” in the database). Eighty-six-point-one percent of patients were treated as medical inpatients, 13.9% as surgical admissions. The median of SOFA score was 6 (IQR, 4 to 8) and the median of SAPSII score was 41 (IQR, 33 to 53). Seven hundred and sixty, 759, 766 and 758 patients respectively belonged to the first quartile (≤5.89), the second quartile (>5.89, ≤10.69), the third quartile (>10.69, ≤20.25) and the fourth quartile (>20.25) of NLCR. Patients with NLCR >20.25 were more tend to be elderly with a history of AFIB and CHF while patients with NLCR <5.89 were more likely to have comorbidities of malignancy and hepatic diseases.
Full table
Association of neutrophil-to-lymphocyte ratio with outcomes
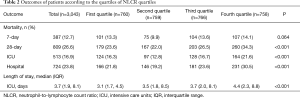
The overall 28-day all-cause mortality of the whole cohort was 26.6% and was different in four NLCR quartiles (P<0.001) while 7-day all-cause mortality was 12.7% but showed no difference (P=0.064). Mortality in ICU (16.3%, 12.8%, 16.7%, 21.6%; P<0.001), in-hospital mortality (21.8%, 19.2%, 23.6%, 30.5%; P<0.001) and length of ICU stay (median: 3.1, 3.5, 3.7, 4.4 days; P<0.001) were different in four NLCR quartiles. And length of ICU stays showed a tendency of increasing with the NLCR quartiles (Table 2).
Full table
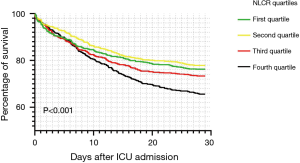
The association between NLCR quartiles and 28-day mortality was shown in the Kaplan-Meier curves in Figure 2. In univariate and multivariate analysis, NLCR was stratified by quartiles to determine whether NLCR was independent-associated with all-cause mortality (Table 3). In univariate model, the fourth quartile of NLCR increased the risk of 28-day mortality. In multivariate model, after adjusting for age, ethnicity, care unit, malignancy, liver disease, hemoglobin, potassium, creatinine, SOFA score and SAPSII score, the fourth quartile remained a statistically significant risk factor of 28-day all-cause mortality (HR 1.22, 95% CI: 1.01–1.49) (Table 3).
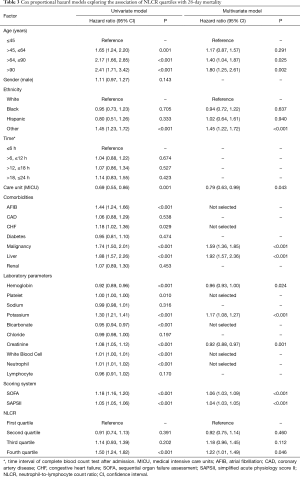
Full table
Discriminatory threshold of 28-day mortality
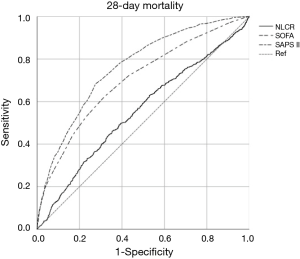
The discriminatory ability of SAPSII, SOFA and the NLCR to predict 28-day mortality is shown in Figure 3. The areas under the receiver operating characteristic curves (AUROCs) for NLCR was 0.553 (95% CI: 0.529–0.576) for 28-day mortality. The discriminatory NLCR thresholds were 14 (sensitivity =0.46, specificity =0.65) for 28-day mortality.
Subgroup analyses
Subgroup analyses were performed to assess the association between the NLCR and 28-day all-cause mortality (Table 4). In most strata, the results in each subgroup population were consistent with the main analysis. Patients with SOFA score <5 and patients with SAPSII score <40 presented an increased risk with high NLCR (>20.25) markedly (HR, 95% CI: 3.19, 1.97–5.15; 2.71, 1.70–4.34) (Table 4).
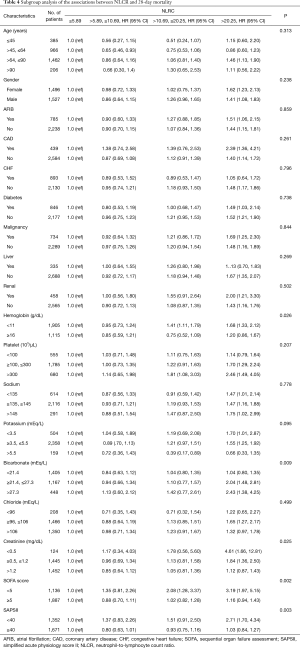
Full table
Discussion
We evaluated 3,043 patients to investigate the relationship between NLCR and all-cause mortality in patients with sepsis. We found that the high NLCR (>20.25) measured in 24 hours after admission to ICU was significantly associated with higher 28-day mortality in septic patients compared with the first quartile (NLCR ≤5.89). The findings remained robust after adjustment for multiple potential confounding variables, indicating that NLCR may be an independent risk factor of the outcome in septic patients. Our results were consistent with the previous study of NLCR tested in a population of oncology patients with sepsis (3) but contrary to that of Salciccioli et al.’s finding (7), which contradicts the relationship between NLCR and 28-day mortality in septic patients. The difference in cohort sizes may explain the divergent conclusion as ours is almost twice as much as Salciccioli’s. Besides, we found that the lowest NLCR group did not reflect the lowest mortality. Compared with the second quartile, the absolute neutrophil count of first quartile was significantly lower than the one of the second quartile. A study conducted by Bermejo-Martín et al. indicated that low circulating neutrophil count could negatively affect the outcome of septic shock patients due to whose inability to launch effective innate responses against the invading microbes (18). Increased neutrophil adhesion to vascular endothelium, resulting in endothelial damage and a decrease in circulating neutrophils (19) may aid in elucidating why the lowest NLCR group did not display the lowest mortality in our study.
NLCR reflects the relationship between the numbers of neutrophil and lymphocyte circulating in human body. The ratio can be used as an indicator of systemic inflammation as well (3). As a number of pathophysiologic processes in sepsis and septic shock are potentially associated with systemic inflammation, NLCR can also play a role in demonstrating sepsis and septic shock. Delayed apoptosis of neutrophil (20) and accelerated apoptosis of lymphocytes (21) were investigated during sepsis. Delayal of neutrophil apoptosis, as well as the upswing in immature neutrophil releasing, both contribute to the significant elevation of circulating neutrophils in septic patients, resulting in a boost of neutrophil-mediated killing, an innate immune response, and phenomenal tissue damage adversely (22). Moreover, it has been shown that neutrophils produce a massive amount of immunosuppressive cytokine IL-10 during sepsis (23), which may promote infection. Aside of neutrophils, lymphocytes are also vital for maintaining appropriate inflammatory responses. Growing apoptosis of lymphocytes in thymus and spleen lead to immunosuppression, multiple organ dysfunction and death (24), probably related to a prolonged, detrimental inflammatory state (25). Le Tulzo et al. (21) showed that higher mortality in septic patients are associated with failure to recover a normal non-apoptotic lymphocyte count. Another research focused on neonatal sepsis revealed that treatment reducing apoptosis of peripheral blood T lymphocytes could improve the prognosis of sepsis (26). And our hypothesis that NLCR is associated with outcomes is primarily based on the studies mentioned above. As our data showed, patients with higher NLCR had higher white blood cell count, higher neutrophil count, lower lymphocytes count and higher 28-day mortality. The mechanisms described above may provide a rationale for the result.
We also found that patients with SOFA score <5 and patients with SAPSII score <40 presented remarkably increased risk with high NLCR (>20.25), and high NLCR (>20.25) patients showing the highest 28-day mortality. It is a surprising result. Patients with SOFA score <5 and patients with SAPSII score <40 were not as serious as the patients with SOFA score ≥5 and patients with SAPSII score ≥40 respectively. We hypothesized that immune disorder might not play a major role in the group of more critical patients, while the existing organ dysfunction might affect more on the fatal outcomes. The insignificant ascending HR for 28-day mortality of the patients along with the increment of NLCR can be attributed to the hypothesis. On the contrary, patients with SOFA score <5 and patients with SAPSII score <40, who were less critical compared with patients with SOFA score ≥5 and patients with SAPSII score ≥40, immune factors maybe still play the leading role on the outcome and the patients’ HR for 28-day mortality increased significantly as the NLCR increased. This result may imply that NLCR might be more valuable for septic patients who were of less critical conditions. Relevant studies should be carried out to test and verify the hypothesis.
Previous studies have reported that increased NLCR was associated with worse outcomes in patients with tumor of the brain (27), lung (28), breast (29), colon (30) and pancreas (31). Moreover, a prospective study found that higher NLCR was associated with higher death rate in patients with acute coronary syndrome (32). Some studies reported that NLCR was a predictor of bacteremia and can be used to detect the presence of sepsis (7,33). These studies implied that NLCR may be reflects the severity of many kinds of diseases. So far, research on the association between NLCR and clinical outcome in patients with sepsis and the results is less controversial compared to those focus on different groups of diseases. In contrast to other studies of NLCR in critical ill patients without specific sepsis, we focused on septic population and our investigation revealed a significantly higher median NLCR. This is consistent with the proposed mechanism of NLCR representing ascended systemic inflammation and severity of sepsis seen in the ICU.
Although NLCR is not magnificently highly specific according to our results, it is an easily accessible, cheap and simple laboratory parameter reflecting the shift of the balance of neutrophil and lymphocyte. And we did find that patients included in the group of the highest NLCR (>20.25) have significantly higher 28-day mortality compared to those of the other three groups. Clinicians should draw more attention to septic patients with high NLCR as they tend to be of higher mortality. Early recognition of septic patients in higher risk with higher NLCR allow medical workers to take relevant actions ahead of the other complex and specific biomarkers are shown. NLCR is a worth mentioning yet not highly specific marker in patients with sepsis.
The major strength of our study was that it was, to our knowledge, the first to investigate the association between NLCR and all-cause mortality of patients with sepsis based on a large, diverse population and publicly available database, MIMIC-III. By using this database, the population in our study is larger than those ever adopted in previous research. Moreover, all of the data were recorded by the medical staff in BIDMC or imported by medical record system directly and what we did was just to extract the data we needed, which ensure our results to be reliable. Compared with previous relevant studies, the confounding factors are more balanced among the four group of NLCR. As NLCR is an easily measurable and simple parameter, based on our findings, it could be served as a supporter of the other mortality markers to improve the accuracy of prognostic prediction in septic patients.
Several limitations inevitably exist. First of all, the data of MIMIC-III are from one medical center, which means that this is actually a retrospective single-center study and selection bias is unavoidable. Secondly, our research presented a phenomenon without fully explaining its pathophysiological mechanism. Thirdly, some important clinical indicators including C-reactive protein and lactate are seriously missing, which may affect the result analysis. Finally, only NLCR of septic patients within 24 hours after admission to the ICU were measured in this study, without assessing the dynamic change of NLCR during the ICU stay, which could be better representative than the one-time NLCR measurement. We should emphasize the relationship between the changes of NLCR and outcomes of the septic patients in the future.
The pathophysiological and immunological mechanism of the relationship between NLCR and clinical outcomes in septic patients requires further study. A perfect biomarker for sepsis has not yet been identified. More immunological experiments and multi-center study on this easy-obtain parameter will be of great significance for early predict the outcomes of sepsis, which must be beneficial for the treatment of sepsis.
Conclusions
Higher NLCR was independently associated with increased 28-day all-cause mortality in adult septic patients but of a limited sensibility and specificity. Further large multicenter prospective studies are needed to confirm the relationship and validate whose clinical significance.
Acknowledgments
Funding: This work was supported by grants from National Science and Technology Major Project (No. 2017ZX10204401), the Natural Science Foundation of China (No. 81870069 and 81970071) and the Natural Science Foundation of Guangdong Province, China (No. 2017A030313781 and 2017A030313712).
Footnote
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/jtd-20-1169). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The MIMIC III database has received ethical approval from the institutional review boards (IRBs) at BIDMC and MIT, and, because the database does not contain protected health information, a waiver of the requirement for informed consent was included in the IRB approval.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Shankar-Hari M, Phillips GS, Levy ML, et al. Developing a New Definition and Assessing New Clinical Criteria for Septic Shock: For the Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3). JAMA 2016;315:775-87. [Crossref] [PubMed]
- Cohen J, Vincent JL, Adhikari NK, et al. Sepsis: a roadmap for future research. Lancet Infect Dis 2015;15:581-614. [Crossref] [PubMed]
- Zahorec R. Ratio of neutrophil to lymphocyte counts--rapid and simple parameter of systemic inflammation and stress in critically ill. Bratisl Lek Listy 2001;102:5-14. [PubMed]
- Benites-Zapata VA, Hernandez AV, Nagarajan V, et al. Usefulness of neutrophil-to-lymphocyte ratio in risk stratification of patients with advanced heart failure. Am J Cardiol 2015;115:57-61. [Crossref] [PubMed]
- Park JJ, Jang HJ, Oh IY, et al. Prognostic value of neutrophil to lymphocyte ratio in patients presenting with ST-elevation myocardial infarction undergoing primary percutaneous coronary intervention. Am J Cardiol 2013;111:636-42. [Crossref] [PubMed]
- Yodying H, Matsuda A, Miyashita M, et al. Prognostic Significance of Neutrophil-to-Lymphocyte Ratio and Platelet-to-Lymphocyte Ratio in Oncologic Outcomes of Esophageal Cancer: A Systematic Review and Meta-analysis. Ann Surg Oncol 2016;23:646-54. [Crossref] [PubMed]
- Salciccioli JD, Marshall DC, Pimentel MA, et al. The association between the neutrophil-to-lymphocyte ratio and mortality in critical illness: an observational cohort study. Crit Care 2015;19:13. [Crossref] [PubMed]
- Riche F, Gayat E, Barthelemy R, et al. Reversal of neutrophil-to-lymphocyte count ratio in early versus late death from septic shock. Crit Care 2015;19:439. [Crossref] [PubMed]
- Liu X, Shen Y, Wang H, et al. Prognostic Significance of Neutrophil-to-Lymphocyte Ratio in Patients with Sepsis: A Prospective Observational Study. Mediators Inflamm 2016;2016:8191254.
- Djordjevic D, Rondovic G, Surbatovic M, et al. Neutrophil-to-Lymphocyte Ratio, Monocyte-to-Lymphocyte Ratio, Platelet-to-Lymphocyte Ratio, and Mean Platelet Volume-to-Platelet Count Ratio as Biomarkers in Critically Ill and Injured Patients: Which Ratio to Choose to Predict Outcome and Nature of Bacteremia? Mediators Inflamm 2018;2018:3758068.
- Hwang SY, Shin TG, Jo IJ, et al. Neutrophil-to-lymphocyte ratio as a prognostic marker in critically-ill septic patients. Am J Emerg Med 2017;35:234-9. [Crossref] [PubMed]
- Terradas R, Grau S, Blanch J, et al. Eosinophil count and neutrophil-lymphocyte count ratio as prognostic markers in patients with bacteremia: a retrospective cohort study. PLoS One 2012;7:e42860. [Crossref] [PubMed]
- Johnson AE, Pollard TJ, Shen L, et al. MIMIC-III, a freely accessible critical care database. Sci Data 2016;3:160035. [Crossref] [PubMed]
- Angus DC, Linde-Zwirble WT, Lidicker J, et al. Epidemiology of severe sepsis in the United States: analysis of incidence, outcome, and associated costs of care. Crit Care Med 2001;29:1303-10. [Crossref] [PubMed]
- Vincent JL, Moreno R, Takala J, et al. The SOFA (Sepsis-related Organ Failure Assessment) score to describe organ dysfunction/failure. On behalf of the Working Group on Sepsis-Related Problems of the European Society of Intensive Care Medicine. Intensive Care Med 1996;22:707-10. [Crossref] [PubMed]
- Le Gall JR, Lemeshow S, Saulnier F. A new Simplified Acute Physiology Score (SAPS II) based on a European/North American multicenter study. JAMA 1993;270:2957-63. [Crossref] [PubMed]
- Johnson AE, Stone DJ, Celi LA, et al. The MIMIC Code Repository: enabling reproducibility in critical care research. J Am Med Inform Assoc 2018;25:32-39. [Crossref] [PubMed]
- Bermejo-Martín JF, Tamayo E, Ruiz G, et al. Circulating neutrophil counts and mortality in septic shock. Crit Care 2014;18:407. [Crossref] [PubMed]
- Fox ED, Heffernan DS, Cioffi WG, et al. Neutrophils from critically ill septic patients mediate profound loss of endothelial barrier integrity. Crit Care 2013;17:R226. [Crossref] [PubMed]
- Tamayo E, Gómez E, Bustamante J, et al. Evolution of neutrophil apoptosis in septic shock survivors and nonsurvivors. J Crit Care 2012;27:415.e1-11. [Crossref] [PubMed]
- Le Tulzo Y, Pangault C, Gacouin A, et al. Early circulating lymphocyte apoptosis in human septic shock is associated with poor outcome. Shock 2002;18:487-94. [Crossref] [PubMed]
- Jimenez MF, Watson RW, Parodo J, et al. Dysregulated expression of neutrophil apoptosis in the systemic inflammatory response syndrome. Arch Surg 1997;132:1263-9; discussion 1269-70. [Crossref] [PubMed]
- Kasten KR, Muenzer JT, Caldwell CC. Neutrophils are significant producers of IL-10 during sepsis. Biochem Biophys Res Commun 2010;393:28-31. [Crossref] [PubMed]
- Wesche DE, Lomas-Neira JL, Perl M, et al. Leukocyte apoptosis and its significance in sepsis and shock. J Leukoc Biol 2005;78:325-37. [Crossref] [PubMed]
- Heffernan DS, Monaghan SF, Thakkar RK, et al. Failure to normalize lymphopenia following trauma is associated with increased mortality, independent of the leukocytosis pattern. Crit Care 2012;16:R12. [Crossref] [PubMed]
- Zheng G, Pan M, Li Z, et al. Effects of vitamin D on apoptosis of T-lymphocyte subsets in neonatal sepsis. Exp Ther Med 2018;16:629-34. [PubMed]
- Patel S, Wang S, Snuderl M, et al. Pre-treatment lymphopenia and indication of tumor-induced systemic immunosuppression in medulloblastoma. J Neurooncol 2018;136:541-4. [Crossref] [PubMed]
- Sarraf KM, Belcher E, Raevsky E, et al. Neutrophil/lymphocyte ratio and its association with survival after complete resection in non-small cell lung cancer. J Thorac Cardiovasc Surg 2009;137:425-8. [Crossref] [PubMed]
- Azab B, Bhatt VR, Phookan J, et al. Usefulness of the neutrophil-to-lymphocyte ratio in predicting short- and long-term mortality in breast cancer patients. Ann Surg Oncol 2012;19:217-24. [Crossref] [PubMed]
- Walsh SR, Cook EJ, Goulder F, et al. Neutrophil-lymphocyte ratio as a prognostic factor in colorectal cancer. J Surg Oncol 2005;91:181-4. [Crossref] [PubMed]
- Stotz M, Gerger A, Eisner F, et al. Increased neutrophil-lymphocyte ratio is a poor prognostic factor in patients with primary operable and inoperable pancreatic cancer Br J Cancer 2013;109:416-21. Erratum in: Br J Cancer. 2013 Oct 1;109(7):2026. Zoughbi, W [corrected to AlZoughbi, W]. [Crossref] [PubMed]
- Muhmmed Suliman MA, Bahnacy Juma AA, Ali Almadhani AA, et al. Predictive value of neutrophil to lymphocyte ratio in outcomes of patients with acute coronary syndrome. Arch Med Res 2010;41:618-22. [Crossref] [PubMed]
- Westerdijk K, Simons KS, Zegers M, et al. The value of the neutrophil-lymphocyte count ratio in the diagnosis of sepsis in patients admitted to the Intensive Care Unit: A retrospective cohort study. PLoS One 2019;14:e0212861. [Crossref] [PubMed]


