Bronchial involvement in chronic eosinophilic pneumonia: a case report
Introduction
Chronic eosinophilic pneumonia (CEP) is an idiopathic disease characterized by extensive accumulation of eosinophils and lymphocytes in the alveoli and interstitium with interstitial fibrosis (1,2). The typical radiographic finding is peripherally located, ill-defined dense opacities with non-segmental distribution. On CT, it is often characterized by peripheral and patchy consolidation with upper lobe dominance (1,3). We experienced a patient with bronchial involvement of CEP with progressed endobronchial obliteration on follow up CT mimicking other endobronchial pathology. This study was exempt from Institutional Review Board approval at Inha University Hospital because it only retrospectively reviewed data for single case report.
Case report
A 73-year-old man visited a private clinic because of cough one month ago. His past medical history was unremarkable and he denied any drug history. He underwent chest radiograph and CT. Pneumonic consolidation was observed in right lower lobe (RLL) and ill-defined ground glass opacity (GGO) in left lower lobe (LLL) with small right pleural effusion (Figure 1). Although a blood test did not indicate leukocytosis with left shift, C-reactive protein was elevated at 8.48 mg/dL. He was diagnosed as acute pneumonia and treated with antibiotics for one month. Pneumonic consolidation showed a gradual decrease in extent on serial chest radiographs and had nearly cleared on the last follow up.
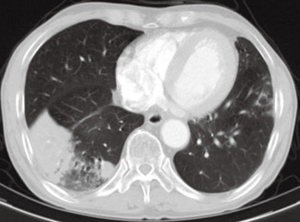
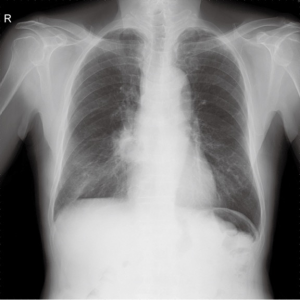
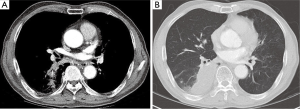
However, he was referred to our hospital due to a new lesion in the right infrahilar area of the last follow up chest radiograph (Figure 2). Follow up chest CT was performed. Pneumonic consolidation and GGO showed improvement in both lower lobes, however, superior segmental bronchus of RLL (B6) was obliterated (Figure 3A) with consolidation in the superior segment and posterior basal segment of RLL (Figure 3B). A B6 lesion was noted retrospectively on prior CT as an elongated soft tissue attenuated endobronchial lesion, which was enlarged and combined with consolidation on follow up CT.
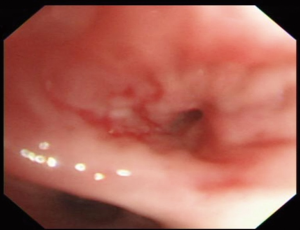
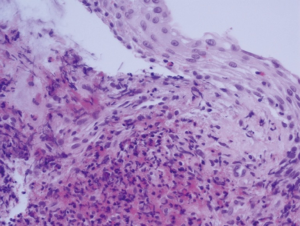
Bronchoscopy showed mucosal inflammation of superior segmental bronchus of RLL without any endobronchial nodule or mass (Figure 4).
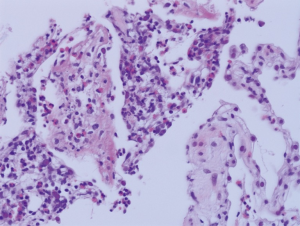
Histopathology of the mucosal biopsy revealed dense eosinophilic infiltration in the bronchial wall with squamous metaplasia (Figure 5). Percutaneous transthoracic biopsy at the consolidation in RLL demonstrated intraalveolar and interstitial eosinophilic infiltration with focal organization and fibrosis, histopathologically (Figure 6).
Laboratory findings showed peripheral blood eosinophilia (eosinophils 13.8%), elevated serum IG-E (856 IU/mL), eosinophilic cationic protein (over 200 µg/L), and negative for common parasitic antibody and ANCA. Retrospective review of the prior blood testing performed at a private clinic showed persistent and gradually elevated level of peripheral eosinophil count between 19.6% and 25.3%.
All serum parasite IgG antibodies for Clonorchis sinensis, Paragonimus westermani, Cysticercus and Sparuganum were negative. Whereas Aspergillus antibody was positive (>200 Unit/mL), Aspergillus antigen was negative. No fungus was isolated on bronchial washing. Clinical and laboratory findings of vasculitis were not evident, too. Serum C-ANCA, P-ANCA and antinuclear antibody was negative. Skin allergy test with 39 allergen including housedust, mite and certain food allergens, nothing provoked allergic response.
Asthma was excluded because he had no characteristic symptoms such as wheezing, shortness of breath or chest tightness. Even though he had coughing for a month, it started quite suddenly and it was persistent rather than triggered in response to exercise or cold air. While taking the bronchoscopy without using steroid and beta 2 agonist, he had no symptom such as wheezing.
With exclusion of parasitic disease and any medical disease that can cause eosinophilia such as asthma or vasculitis, CEP was diagnosed and oral steroid therapy was started. One day after the start of steroid therapy, cough had subsided and peripheral eosinophil count became normalized as 0.3%. Chest radiograph showed a markedly decreased extent of consolidation in RLL. Nine days after the start of treatment, consolidation in RLL had nearly cleared on follow up chest radiograph.
He has been well followed up in the outpatient department without occurrence of any respiratory symptoms or abnormalities on chest radiographs during 17 months of follow up.
Discussion
CEP was first described by Carrington CB as a chronic variant of Loffler’s syndrome (4). Histologically it is characterized by extensive accumulation of eosinophils and lymphocytes in the alveoli and interstitium. Intraluminal fibroses are more prominent than in acute eosinophilic pneumonia (1,2). The clinical picture of the disease is progressive and severe illness characterized by high fever, weight loss, night sweats, and shortness of breath (5). The typical radiographic finding is dense opacities with ill-defined margins and without lobar or segmental distribution arranged peripherally, apposed pleura. The opacities are usually found in an apical location. Opacities surrounding the lung have the appearance of a photographic negative shadow usually seen in pulmonary edema (6). On CT, it is often characterized by peripheral and patchy consolidation with an upper lobe predominance. Less common findings include ground-glass opacities, nodules, and reticulation (1,3).
Bronchial involvement of CEP is rare. Only a few cases of bronchial involvement of CEP have been reported (7-9) (Table 1). In previous case reports reviewed, the main clinical symptoms were fever and cough. On plain radiograph or CT scan, they showed patchy parenchymal consolidation mimicking pneumonia. Also in our case, the patient had initially undergone treatment in accordance with pneumonia. In one case, bronchoscopy showed small nodules in bronchi (7) and in another case, small abscesses scattered along the trachea and main bronchi (8). Matsushima et al proposed the hypothesis that biopsies revealed severe allergic inflammatory reaction with subsequent necrotizing inflammation and many eosinophils.
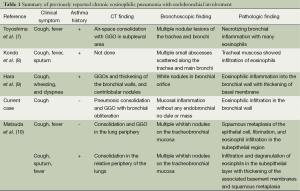
Full table
Previous reports have not demonstrated definite endobronchial obstructive lesions on their CT imaging, except bronchoscopic findings (7-10). In our case, bronchial mucosal inflammation was seen as an endobronchial lesion with luminal obliteration on CT scans. To the best of knowledge, presentation as a rapidly growing endobronchial lesion in CEP has not been reported in the English language literature. In addition, it made diagnosis more difficult that dry cough was the only symptom without fever or asthma history. Although he showed marked peripheral blood eosinophilia at the initial presentation, both radiologists and pulmonologists paid less attention because pneumonic consolidation showed improvement with antibiotic therapy. It showed rapid and dramatic response to corticosteroid therapy after correct diagnosis.
We reported on an unusual presentation of CEP without asthma. We proposed that in the case of considerable peripheral blood eosinophilia, CEP should be included in the differential diagnosis even when there is atypical finding such as endobronchial involvement.
Acknowledgements
Funding: This work was supported by INHA University research fund.
Disclosure: The authors declare no conflict of interest.
References
- Jeong YJ, Kim KI, Seo IJ, et al. Eosinophilic lung iseases: a clinical, radiologic, and pathologic overview. Radiographics 2007;27:617-37; discussion 637-9. [PubMed]
- Mochimaru H, Kawamoto M, Fukuda Y, et al. Clinicopathological differences between acute and chronic eosinophilic pneumonia. Respirology 2005;10:76-85. [PubMed]
- Ebara H, Ikezoe J, Johkoh T, et al. Chronic eosinophilic pneumonia: evolution of chest radiograms and CT features. J Comput Assist Tomogr 1994;18:737-44. [PubMed]
- Carrington CB, Addington WW, Goff AM, et al. Chronic eosinophilic pneumonia. N Engl J Med 1969;280:787-98. [PubMed]
- Sano S, Yamagami K, Yoshioka K. Chronic eosinophilic pneumonia: a case report and review of the literature. Cases J 2009;2:7735. [PubMed]
- Gaensler EA, Carrington CB. Peripheral opacities in chronic eosinophilic pneumonia: the photographic negative of pulmonary edema. AJR Am J Roentgenol 1977;128:1-13. [PubMed]
- Toyoshima M, Suda T, Chida K. Tracheobronchial involvement in chronic eosinophilic pneumonia. Am J Respir Crit Care Med 2012;186:1057. [PubMed]
- Kondo T, Suzuki H, Hirokawa Y, et al. Chronic eosinophilic pneumonia with small abscesses in the tracheo-bronchial mucosa and lung parenchyma. Intern Med 1992;31:391-3. [PubMed]
- Hara J, Nishi K, Demura Y, et al. Chronic eosinophilic pneumonia with endobronchial involvement. J Bronchology Interv Pulmonol 2011;18:285-7. [PubMed]
- Matsuda Y, Tachibana K, Sasaki Y, et al. Tracheobronchial lesions in eosinophilic pneumonia. Respir Investig 2014;52:21-7. [PubMed]


