
Short-term and mid-term effects of radiofrequency ablation in mitral valve surgery in patients with different left atrial sizes
Introduction
In both mitral valve regurgitation and mitral valve stenosis, an overload of left atrial pressure or volume leads to inflammation and fibrosis of the atrial muscle, which gradually results in anatomical remodeling of the atrial wall. Atrial that expand further can also cause electrophysiological activity disorder, which can result in persistence of the atrial fibrillation (AF) reentry loop (1).
Valvular AF has an incidence as high as 60% and is associated with high rates of disability and mortality. Not only does valvular AF significantly reduce cardiac output, but it also increases the risk of systemic arterial embolization. Cardiac valve surgery with improved bipolar atrial fibrillation radiofrequency ablation (AFRA) has been applied in the treatment of AF in clinical practice (2,3). Valve surgery combined with bipolar AFRA does not have an increased surgical risk compared with simple valve surgery; moreover, it has a higher sinus rhythm recovery rate, a lower incidence of cerebrovascular complications, and a better surgical effect (4). Left atrial diameter (LAD), preoperative cardiac function, and the duration of AF have been shown to be the major factors affecting postoperative sinus rhythm recovery and maintenance (5).
However, a conclusion on the range of the LAD for continuous AF has not been reached. Previous studies have shown that patients with simple AF with a LAD <45 mm can benefit from catheter ablation (6); however, valvular with long-term left atrial pressure overload can lead to significant enlargement of the left atrium. Little research has been carried out into the impact of valvular AF on surgical risk or the ablation effect.
This study aimed to compare the clinical and follow-up data of two groups of valvular AF patients with left atriums of different diameters (LAD >45 mm and LAD ≤45 mm) who underwent mitral valve surgery with improved bipolar AFRA, in order to provide a basis and reference for the future clinical treatment of valvular AF.
We present the following article in accordance with the STROBE reporting checklist (available at http://dx.doi.org/10.21037/jtd-20-2953).
Methods
General data collection
A total of 264 consecutive patients who underwent cardiac surgery with modified bipolar AFRA in the Department of Cardiovascular Surgery, PLA General Hospital, between May 2016 and January 2019, were enrolled. All patients were operated on by the same surgeon. The patients’ clinical data were collected and analysed. All patients had long-term persistent (duration >1 year) AF before the surgery. Of the patients, 190 underwent another procedure simultaneously with mitral valve surgery, with or without tricuspid valvuloplasty, including aortic or double valvuloplasty (52 patients), simultaneous secondary valve and simple tricuspid valve treatment (10 patients), surgical correction of congenital heart disease (8 patients), and coronary artery bypass graft (CABG; 4 patients). The patients who underwent simultaneous mitral valve surgery were divided into two groups according to the diameter of the left atrium. Group A comprised 75 patients with LAD >45 mm, and group B included 75 patients with LAD ≤45 mm. The preoperative general data of the two groups were matched by propensity score. The study was carried out in accordance with the Declaration of Helsinki (as revised in 2013). The study was approved by the Institutional Ethics Committee of the First Medical Center of PLA General Hospital. All personal data have been protected and secured according to current national and international laws. Individual consent for this retrospective analysis was waived.
Inclusion and exclusion criteria
The inclusion criteria were as follows: (I) patients with valvular heart disease and long-term persistent AF (>1 year) who underwent simple mitral valve surgery with or without tricuspid valvuloplasty and were diagnosed by cardiac color Doppler ultrasound and electrocardiogram before surgery; or patients with valvular heart disease and long-term persistent AF (>1 year) who underwent mitral valve surgery alone, with or without tricuspid valvuloplasty; (II) aged 40–70 years; and (III) left ventricular end diastolic period ≤70 mm; (IV) LAD ≤65 mm; (V) left ventricular ejection fraction (LVEF) ≥0.50. Patients who received a combination of mitral valve surgery with other surgical methods [e.g., CABG, aortic valve replacement (AVR), simple tricuspid valve formation (TVP)/tricuspid valve replacement (TVR), double valve replacement, or correction of congenital heart disease] were excluded.
Surgical methods
Two groups of patients who underwent cardiopulmonary bypass (CPB) under general anaesthesia were treated with mitral valve plasty/replacement combined with modified bipolar radiofrequency ablation. Conventional thoracotomy was performed to establish CPB. The superior vena cava cannula was intubated at a right angle or directly via the right auricle of the heart, depending on the size of the right atrium. Before parallel circulation, the oblique sinus was opened to the upper and lower openings of the right pulmonary vein; the right pulmonary vein was dissociated, and vestibular ablation of the right pulmonary vein was carried out (AtriCure Inc., bipolar radiofrequency ablation forceps). The right atrium was cut longitudinally parallel to the atrioventricular sulcus after cardiopulmonary bypass. The ascending aorta was clamped, and the right and left coronary artery were perfused with cold histidine-tryptophan-ketoglutarate (HTK) myocardial protection solution. Part of the atrial septum was cut, and left heart drainage was performed. A lesion was placed from the proximal end of the right atriotomy into and beyond the orifice of the SVC. A similar lesion was then place from the proximal end of the right atriotomy into and beyond the orifice of the IVC. This completes the so-called “SVC-IVC lesion”. And then the “T” lesion was completed by placing an lesion from the distal end of the right atriotomy down to the tricuspid valve annulus. The heart was pulled to the upper right side to expose the left pulmonary vein. The electrotome was detached, the Marshall ligament was cut off, and the left pulmonary vein vestibule was ablated.
After perfusion of the myocardial protective fluid was completed and cardiac arrest was satisfactory, the pericardium was placed into ice-cold saline to protect the myocardium. The atrial septum and the left ventricular sulcus were incised, and the transverse sinus was opened. Ablation lines were performed from the right superior pulmonary vein through the left roof to the left superior pulmonary vein, from the right inferior pulmonary vein to the left inferior pulmonary vein, from the coronary sinus to the posterior mitral valve annulus, and from the left atrial appendage to the left inferior pulmonary vein. The left atrium was folded by applying a parallel mattress with continuous double sutures, and the left atrial appendage was sutured closed (Figure 1). After the completion of modified bipolar AFRA, mitral valve plasty or replacement was performed. Deleted the sentence CPB was discontinued after the recovery of body temperature and cardiac resuscitation; pacemaker wires were sewn, the incision was sewn layer by layer, and the patient was moved to the intensive care unit (ICU).
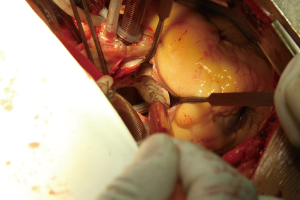
Observation and treatment of postoperative
The patients’ blood pressure, heart rate, oxygen saturation, and other vital signs were monitored closely. Intravenous amiodarone was continuously pumped to maintain the rhythm of the heart (a total of 1,200 mg was pumped within 24 hours after surgery and maintained for 48 hours). The patients were extubated On the first day after surgery, orally administered amiodarone tablets (0.2 mg, bid), and treated with the standard dose of warfarin anticoagulants. Once the patients were in a stable condition, they were transferred from the ICU to the general ward. Perioperative complications were treated promptly. The patients were discharged from hospital when the results of their chest radiography, electrocardiogram, and echocardiography were determined to be normal. After discharge, the patients continued taking anticoagulation and amiodarone treatment (0.2 mg, qd, 3 months after surgery).
Postoperative follow-up
All patients were re-examined before discharge. Outpatient follow-up was conducted at 3 months, 6 months, 1 year, and 2 years after the operation. The electrocardiogram and echocardiography results of the patients were reviewed. Auscultation was performed for at least 2 minutes; if arrhythmia was found or the patient reported palpitations, 24-hour holter electrocardiogram was carried out. Patients with no symptoms or arrhythmia after auscultation underwent routine 12-lead electrocardiogram. The heart morphology, cardiac function, and sinus rhythm recovery of the patients were recorded.
Observation indexes
Age, sex, height, body mass, smoking, drinking history, preoperative complications, heart size, heart function grading, degree of valvular regurgitation/narrowing, CPB time, aorta blocking time, intraoperative valve processing mode, duration of AF ablation, duration of postoperative care, postoperative mortality and complication rates, and the short- and medium-term sinus rhythm recovery rates were recorded for the two groups of patients.
Statistical analysis
The data were statistically analysed with the IBM SPSS 19.0 software package. The measurement data were expressed as (mean ± standard deviation). Mean values between the two groups were compared using the t-test, and count data were expressed as a rate. The χ2 test was used to compare rates. A P value <0.05 was considered to be statistically significant.
Results
Comparison of preoperative baseline data and related surgical indicators between groups A and B
Patients’ preoperative baseline data were compared between the two groups, with no statistically significant differences observed in age, sex, body mass, preoperative complications, preoperative cardiac function, or degree of valvular disease (P>0.05). The LAD, left ventricular diameter, and pulmonary artery pressure in group A were significantly higher than those in group B, and the difference was statistically significant (P<0.05). (Table 1). There were no significant differences between groups A and B in mitral valve stenosis or insufficiency or valve replacement, nor were any significant differences found in cardiopulmonary bypass time, aortic occlusion time, or bipolar radiofrequency time (P>0.05) (Table 2).
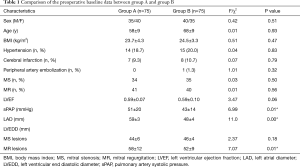
Full table
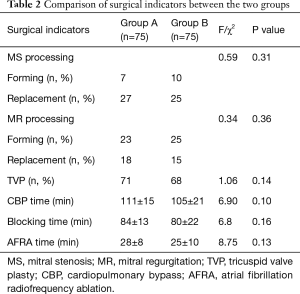
Full table
Comparison of postoperative indicators in groups A and B
Next, the postoperative data were compared between the two groups. The LAD was significantly reduced in both groups after the operation, and a significant difference was found in the LAD between the two groups (P<0.05). The duration of postoperative ventilator assistance, ICU stay, temporary pacemaker use and intravenous amiodarone use in group A were all longer than those in group B; however, the differences between the two groups were not statistically significant. One patient (1.3%) in group A died from septic shock after surgery, while no patients in group B died; the difference was not statistically significant (Table 3).
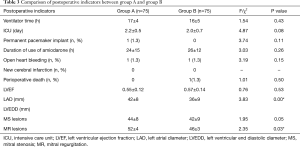
Full table
Comparison of sinus rhythm maintenance and atrial fibrillation recurrence in groups A and B in the 2 years after surgery
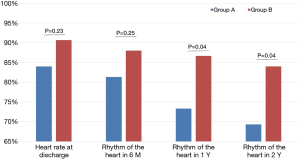
The rates of sinus rhythm recovery in group A (LAD >45 mm) at 1 week, 6 months, 1 year, and 2 years after surgery were 84.0%, 81.33%, 73.33%, and 69.33%, respectively, compared with 90.67.0%, 88.00%, 86.67%, and 84.00% at 1 week, 6 months, 1 year, and 2 years after surgery, respectively, in group B (LAD ≤45 mm). The difference between the two groups was statistically significant at the two points in time of 1 year, and 2 years (P<0.05) (Figure 2).
Comparison of the size of the left atrium and ventricle, and the LVEF in groups A and B in the 2 years after surgery
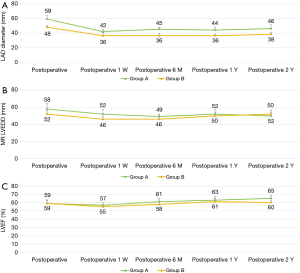
Postoperative left atrial size and cardiac function recovery were evaluated in both groups. The LADs of the patients of group A and group B were significantly reduced at 1 week after surgery compared with those before surgery (59±3 vs. 42±8 mm in group A and 48±4 vs. 36±9 mm in group B; P<0.01), and the difference was statistically significant. After 2 years of follow-up, no significant difference existed in the left atrial and ventricular diameters or cardiac function between the two groups (Figure 3A,B,C).
In 2 years of follow-up, no new cerebrovascular events were reported in either of the two groups. During follow-up, no major anticoagulant-associated adverse events, including death, heart failure, bleeding, and infarction, occurred.
Discussion
Most cases of paroxysmal valvular AF progress to persistent AF, leading to further enlargement of the left atrium over time, which causes irreversible electrical and structural remodelling. Therefore, early rhythm correction can help to improve recovery (7). The current success rate of ablation in AF patients with simple AF who have a LAD >45 mm is low, and long-term AF accompanied by definite morphology heart disease or left atrial thrombosis has been defined as a relative or absolute contraindication for catheter radiofrequency ablation (6). Surgical AF ablation has unique advantages in AF patients with a LAD of >45 mm; not only can it correct abnormal cardiac morphology and clear left atrial thrombosis, but it can also perform radiofrequency ablation at the same time to correct arrhythmia.
As pointed out by Ad et al. (4), mitral valve surgery with AFRA does not increase surgical risk, has a high recovery rate, decreases the long-term AF burden, and significantly reduces cerebrovascular complications. In this study, the results for patients with a LAD >45 mm were good, and no increase was observed in surgical risk. The rate of early postoperative sinus rhythm recovery reached 84%. At 2 years after surgery, the rate of postoperative sinus rhythm remained high at 69.3%, and no cerebrovascular complications were reported. The results of recent medium-term studies show that even for AF patients with significant LAD enlargement (45≤ LAD ≤65 mm), satisfactory results can be achieved after mitral valve surgery with the improved Cox-maze IV procedure, which was developed by James Cox and introduced clinically in 1987. For patients with LAD enlargement, AF may also be corrected by performing surgery at the same time. For left atrial valvular AF <45 mm in diameter, the operative effect is better. The potential reasons are as follows. Firstly, pathophysiological mechanisms of valvular AF is not consistent with other nonvalvular AF. Valvular AF has increased volume or pressure load, which causes electrical remodeling with changes in electrophysiology and ion channel characteristics in the early stage, and causes fibrosis of atrial muscle and extracellular matrix, amyloidosis and even amyloidosis in the late stage. These leads to further enlarged anatomical remodeling of the left atrium. Surgery can be used to correct the mitral valve lesion, reduce the load on the left atrium, fold the left atrium, and restore the anatomical structure of the left atrium. The improved Maze IV surgery not only corrects the pathophysiological mechanism but also blocks the electrophysiological mechanism of AF, and AF ablation has a significant effect. Secondly, the surgical bipolar radiofrequency ablation system has unique advantages over the internal ablation system. Bipolar radiofrequency ablation forceps (AtriCure) were used for all patients in our study; these can concentrate energy between the bipolar poles, shortening the operation and making it more effective. At the same time, the bipolar radio frequency device has different impedance inductions, which can accurately determine the wall permeability of ablation according to the impedance change of the tissue. Thirdly, all patients in our cohort were treated by the same surgeon, who has vast experience in performing valve surgery; this ensured that the implementation method of each path was correct and had less influence on the experimental results. The experience of the surgeon and complete Cox-Maze IV ablation are also important factors in improving successful conversion. As noted by Ad et al. (4), simultaneous surgical ablation has significant efficacy; however, the results depend on the experience and rigorous training of the surgeon.
Jurin et al. reported that for every 5-mm increase in LAD, the risk of AF increases 1.39 fold, whereas a LAD <45 mm is a crucial factor in maintaining long-term sinus rhythm (5). In our study, patients in both group A and group B achieved good ablation effects at an early stage. However, over time in both groups, some patients became unable to maintain sinus rhythm, and reversion to atrial flutter or AF occurred. The sinus rhythm maintenance rate of patients with left atrial enlargement was significantly lower than that of patients in the other group, and the difference was statistically significant (P<0.05). Differences in left atrial size have a vital importance in radiofrequency ablation in mitral valve surgery. The reasons for this are as follows. First, according to the theory of multiple recursions and focal agitation in the maintenance mechanism of AF, the smaller the left atrium is, the shorter the duration of mitral valve disease, and the shorter the formation and duration of AF. Thus, the formation of new wavelets is reduced, which decreases the continuous collision and fusion between the wavelet to lower the recurrence of AF (8). Second, in patients with chronic long-term AF or permanent AF, the left atrium is clearly enlarged, and the left atrial wall exhibits obvious fibrosis, with focal and even calcified plaques. Consequently, when the bipolar video ablation clamp is applied to the atrial wall, the enlarged left atrial ablation line cannot be clamped completely, which means it is unable to form continuity with other ablation lines effectively. Also, fibrosis or calcification of the left atrium wall leads to the inability to complete transmural ablation or to uneven ablation energy distribution during ablation, resulting in local agitation (9). In this study, statistically significant differences were observed in the sinus rhythm maintenance rates between the two groups in each period (1 week, 6 months, 1 year, 2 years) in the 2 years after surgery (P<0.05), which further indicated that preoperative differences in LAD are closely related to the maintenance of postoperative sinus rhythm.
For patients with valvular AF, changes in heart rhythm are frequently ignored because mitral valve lesions are aggravated gradually, leading to left atrial thrombosis. According to the Framingham study, the incidence of stroke caused by rheumatic valvular disease complicated with AF was 17.6 times higher than that of the control group (10), and the mortality and disability rates were significantly increased. However, despite anticoagulant therapy, the annual incidence of severe systemic embolism after mechanical valve replacement still ranges from 0.8–3% (11). The risk of thromboembolism is increased by risk factors such as advanced age, history of stroke, and the presence of chronic AF, which is considered to be the most important and only correctable factor (12). Therefore, simultaneous radiofrequency ablation is of great significance to reducing the risk of serious complications for valvular AF patients after valve replacement/plasty (13). In this study, no new cerebrovascular events occurred in either group during the 2 years of follow-up. It is possible that radiofrequency ablation during the same period reduced the risk of left atrial thrombosis caused by AF. What’s more, all of the patients in this study underwent left atrial volume reduction during surgery, in which the left atrial appendage was closed and the left atrial anatomy was restored; this significantly improved cardiac function, prevented postoperative arterial embolization-related complications, and helped to maintain long-term sinus rhythm after surgery.
Mitral valve surgery with modified Maze IV bipolar radiofrequency ablation is an effective treatment for chronic long-term persistent valvular AF and has a high sinus rhythm recovery rate. The larger the LAD, the less likely sinus rhythm is to be maintained; the smaller the LAD, the better the sinus rhythm.
Acknowledgments
Funding: This study was supported by the PLA funds (16BJZ28) to SLJ.
Footnote
Reporting Checklist: The authors have completed the STROBE reporting checklist. Available at http://dx.doi.org/10.21037/jtd-20-2953
Data Sharing Statement: Available at http://dx.doi.org/10.21037/jtd-20-2953
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/jtd-20-2953). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The study was carried out in accordance with the Declaration of Helsinki (as revised in 2013). The study was approved by the Institutional Ethics Committee of the First Medical Center of PLA General Hospital. All personal data have been protected and secured according to current national and international laws. Individual consent for this retrospective analysis was waived.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Benjamin EJ, Levy D, Vaziri SM, et al. Independent risk factors for atrial fibrillation in a population-based cohort. The Framingham Heart Study. JAMA 1994;271:840-4. [PubMed]
- Bum Kim J, Suk Moon J, Yun SC, et al. Long-term outcomes of mechanical valve replacement in patients with atrial fibrillation: impact of the maze procedure. Circulation 2012;125:2071-80. [PubMed]
- Cheng S, Lu X, Wang J, et al. Thoracoscopic lobectomy for massive hemoptysis caused by complete pulmonary vein occlusion after radiofrequency ablation for atrial fibrillation. J Thorac Dis 2018;10:E296-300. [Crossref] [PubMed]
- Ad N, Holmes SD, Massimiano PS, et al. Long-term outcome following concomitant mitral valve surgery and Cox maze procedure for atrial fibrillation. J Thorac Cardiovasc Surg 2018;155:983-94. [Crossref] [PubMed]
- Jurin I, Hadzibegovic I, Durlen I, et al. Left atrium size and red cell distribution width predict atrial fibrillation progression from paroxysmal or persistent to permanent. Acta Clin Belg 2020;75:205-11. [Crossref] [PubMed]
- Mont L, Bisbal F, Hernandez-Madrid A, et al. Catheter ablation vs. antiarrhythmic drug treatment of persistent atrial fibrillation: a multicentre, randomized, controlled trial (SARA study). Eur Heart J 2014;35:501-7. [Crossref] [PubMed]
- Van Gelder IC, Haegeli LM, Brandes A, et al. Rationale and current perspective for early rhythm control therapy in atrial fibrillation. Europace 2011;13:1517-25. [Crossref] [PubMed]
- Marrouche NF, Wilber D, Hindricks G, et al. Association of atrial tissue fibrosis identified by delayed enhancement MRI and atrial fibrillation catheter ablation: the DECAAF study. JAMA 2014;311:498-506. [Crossref] [PubMed]
- Flegel KM, Shipley MJ, Rose G. Risk of stroke in non-rheumatic atrial fibrillation. Lancet 1987;1:526-9. [Crossref] [PubMed]
- Bryan AJ, Rogers CA, Bayliss K, et al. Prospective randomized comparison of CarboMedics and St. Jude Medical bileaflet mechanical heart valve prostheses: ten-year follow-up. J Thorac Cardiovasc Surg 2007;133:614-22. [Crossref] [PubMed]
- Medi C, Hankey GJ, Freedman SB. Stroke risk and antithrombotic strategies in atrial fibrillation. Stroke 2010;41:2705-13. [Crossref] [PubMed]
- Miyairi T, Miura S, Kigawa I, et al. Mid-term results of a closed biatrial procedure using bipolar radiofrequency ablation concomitantly performed with non-mitral cardiac operations. Interact Cardiovasc Thorac Surg 2009;9:169-72. [Crossref] [PubMed]
- Gillinov AM, McCarthy PM, Blackstone EH, et al. Surgical ablation of atrial fibrillation with bipolar radiofrequency as the primary modality. J Thorac Cardiovasc Surg 2005;129:1322-9. [Crossref] [PubMed]
(English Language Editor: J. Reynolds)


