Efficacy of cardiovascular surgery for Marfan syndrome patients: a single-center 15-year follow-up study
Introduction
Marfan syndrome (MFS) is caused by fibrillin 1 gene mutation, which is characterized by autosomal dominant inherited connective tissue disease (1). A large portion of MFS patients develop cardiovascular disease during their lifetime. The most common causes include aortic root dilation with aortic valve insufficiency, thoracic aortic aneurysm, aortic dissection, and mitral valvular lesions. It has been demonstrated that the major cause of death in MFS patients is aortic dilatation or aortic dissection. In addition, heart failure due to heart valvular disease and arrhythmia is also another cause of death in MFS patients (2), while Murdoch et al. reported that the average life expectancy of MFS patients was 44 years, with cardiovascular disease being the main cause of death (3), and the lifespan of untreated patients with the classic MFS was approximately 44 years in 1972 but has markedly increased to 72 years in 1993 (4). Heart failure and arrhythmia caused by heart valvular disease are additional causes of death in MFS patients (5). Hence, surgical intervention is required timely for patients with acute aortic dissections (Stanford A, Stanford B) or more severe aortic and mitral valvular lesions (6). At present, there are only a few reports regarding the prognosis and long-term follow-up of MFS patients who underwent different cardiovascular surgical procedures. Therefore, in the present study, we retrospectively analyzed the results of hospitalization and long-term follow-up of MFS patients.
We present the following article in accordance with the STROBE reporting checklist (available at http://dx.doi.org/10.21037/jtd-20-2109).
Methods
Patient characteristics
The retrospective study was approved by the Ethics Committee of Changhai Hospital affiliated to Naval Medical University (No. 20200189) and informed consent was taken from all the patients. The study was conformed to the provisions of the Declaration of Helsinki (as revised in 2013). The study subjects included 145 patients hospitalized for MFS from January 2005 to December 2019. We used the revised Ghent nosology to definite the diagnosis of MFS. Age, gender, pre-surgical basic disease, New York heart association (NYHA) cardiac function classification, primary disease type, aortic sinus diameter, the timing of surgery, and preoperative complications were obtained. Ten patients were excluded from the study due to rupture of the dissection aneurysm and severe heart failure and hence were not suitable for surgery. MFS patients during hospitalization were divided into David, Bentall, and Arch surgery groups. Discharged MFS patients during follow-up were divided into David, Bentall, Proximal + Arch surgery, and Arch + Stent groups that of all kinds of surgery involved arch of aorta or needed surgery of intraoperative stents.
Follow up
Surgery-related death was defined as death within 30 days post-surgery or before final discharge. Adverse events were defined as surgical death, severe stroke or paraplegia. Preoperative, intraoperative and outcome data were collected according to reported standardized definitions. Postoperative follow-ups were performed through outpatient, telephone or text messages. The average follow-up period was 7.26±5.34 years.
Surgical procedures
Extracorporeal intubation was performed using the femoral artery and conventional right atrial intubation. Inferior and superior vena cava cannulas were used for mitral and tricuspid valve surgery. If circulatory arrest was required to treat aortic arch lesions during surgery, selective cerebral perfusion was performed using innominate artery intubation. After the initiation of extracorporeal circulation, if adverse perfusion events were observed, the right axillary artery was intubated. Aortic root aneurysm or dissections were limited to patients undergoing Bentall or David surgery under conventional low temperature (33 °C) cardiopulmonary bypass with aortic root surgery, while, all David surgery were performed with reimplantation (7,8). A family history of dissection, increased rate of aortic dilation (greater than 2 mm per year), severe aortic valve regurgitation with left ventricular dilation, and relative feasibility of aortic valve-sparing surgery are indicators for early surgical intervention (9). For patients in whom anticoagulation is not the recommendation, valve-sparing techniques have been adopted. The David technique involves reimplantation of the native aortic valve into a graft and is for patients with annular involvement. But there were 25 percent of patients with MFS undergoing valve-sparing procedures have aortic regurgitation at 10 years, secondary to disease progression (10). Some of the patients underwent intra-aortic isolation and repair for Stanford type B aortic dissection. To reduce the incidence of postoperative paraplegia, cerebrospinal fluid drainage tubes were placed in MFS patients who required surgical intervention for descending aorta. If the aortic dissection involved coronary, the coronary artery bypass graft was performed. For the selection of artificial vessels, a diameter 15% larger than the diameter of the aortic annulus was selected. For patients with hemostasis, the transverse sinus was closed using a suitable autologous pericardial or bovine pericardial patch, and right atrial drainage was performed.
Statistical analysis
Continuous variables were expressed as the standard deviation of the mean when normally distributed, and as the median and quartile range when they were not normally distributed. Categorical variables were expressed as numbers and percentages. T-test was used to compare continuous variables that were normally distributed. The U test was used to compare continuous variables that were not normally distributed. Categorical variables were analyzed using chi-square or Fisher precise tests. Kaplan-Meier method was used to describe the overall survival rate and the re-surgical rate. Using univariate logistic regression analysis, potential risk factors for major adverse outcomes were initially identified, and significant univariate related factors were included in the inverse stepwise multivariate logistic regression model to determine whether they were independent factors (11). SPSS 22.0 software was used for statistical analysis.
Results
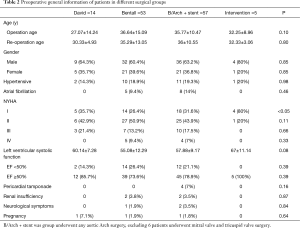
A total of 135 MFS patients underwent surgery. Preoperative general clinical data are shown in Table 1. The study included 86 males (63.7%) and 49 females (36.3%), aged 8–71 years old, with an average age of 34.74±13.48 years. 26 (19.3%) patients had hypertension and 13 (9.6%) patients had atrial fibrillation prior to surgery. Preoperative NYHA classifications were as follows: 47 cases with grade I (34.8%), 59 cases with grade II (43.7%), 20 cases with grade III (14.8%), 9 cases with grade IV (6.7%). Preoperative echocardiography indicated 30 (22.2%) cases with a left ventricular ejection fraction (LVEF) lower than 50%, with an average of 57.42%±10.81%. Arterial lesion types were as follows: 58 (43%) cases with aortic aneurysm expansion, 61 (45.2%) cases with Stanford type A aortic dissection, 10 (7.4%) cases with Stanford type B aortic dissection, and 2 cases with atrial septal defect. There were 17 cases with mitral valvular lesions. Preoperative ultrasound examination showed that 74 cases (54.8%) had severe aortic valvular regurgitation, 33 (24.4%) had mild aortic valvular regurgitation, 28 (20.7%) had no reflux and aortic sinus. The diameter ranged from 34–89 mm, with an average of 53.3±11.6 mm. 41 (30.3%) patients underwent emergency surgery and 94 (69.6%) patients underwent elective surgery. Pericardial tamponade occurred in 6 (4.4%) cases, renal insufficiency in 5 (3.7%) cases, neurological symptoms in 6 (4.4%) cases, and 3 (2.2%) were pregnant cases (Tables 1,2). There were 61 cases with Stanford A dissection, 10 cases with Stanford B dissection, and 58 cases with thoracic aortic aneurysm. There were 40 (29.6%) cases of emergency operation and 95 (70.4%) cases of elective operation. Surgical procedures were as follows: Bentall surgery was performed in 53 cases, David surgery in 14 cases, Bentall + half arch replacement/total aortic arch replacement + descending aortic elephant trunk surgery in 57 cases, aortic isolation and repair in 5 cases. Other concurrent surgeries include mitral annuloplasty in 12 cases, mitral valvular replacement in 5 cases, coronary artery bypass grafts in 15 cases, and atrial septal defect repair in 2 cases. Thirty-three cases underwent re-surgeries and included 28 cases with a history of open surgery and 5 cases with a history of interventional surgery. Thirty-six patients were treated using a right atrial shunt for aortic anastomotic bleeding (Table 2).

Full table
Full table
Short-term results
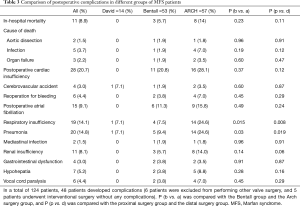
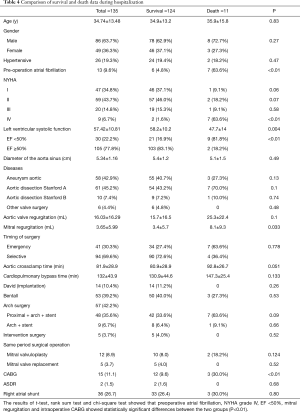
Forty-eight (35.6%) patients had varying degrees of complications and included 11 patients with renal failure and 3 requiring dialysis at discharge (Table 3). Vocal cord paralysis was observed in 6 (4.4%) cases, and cerebrovascular accident occurred in 4 (3%) cases, which included 2 cases with postoperative cerebral infarction and 2 cases with cerebral hemorrhage. Gastrointestinal dysfunction was observed in 2 cases, which included gastrointestinal bleeding in 1 case and intestinal obstruction in the other. The most common complications were respiratory failure (19 cases, 14.1%) and pulmonary infections (20 cases, 14.8%). From the short-term results, patients who underwent arch and distal surgery were more likely to have respiratory dysfunction compared to patients who underwent proximal aortic surgery (P=0.008). The type of surgical procedure had no significant effect on the incidence of complications during hospitalization. There were 11 (8.1%) deaths during hospitalization. No statistical differences in mortality were observed between the different groups (P=0.11). Two patient deaths were due to descending aortic dissection, 3 cases to heart failure, and 5 cases to septic shock. Because symptoms from aortic dissection generally takes a long time, 3 patients had multiple organ failure, however, cardiac function recovered to a satisfactory level post-surgery. This may be related to “hyperperfusion syndrome” (8). The type of surgical procedure was not significantly associated with the incidence of complications during hospitalization. Pre-surgical cardiac function classification IV(P=0.047), EF <50% (P=0.047), pre-surgical atrial fibrillation (P=0.042), and the lesions in the coronary arteries (P=0.02) were independent risk factors for post-surgical mortality (Tables 4,5).
Full table
Full table

Full table
Long-term results
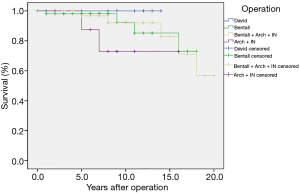
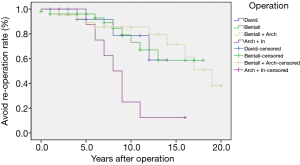
A total of 10 deaths were recorded during the 15-year follow-up period, with 7 cases lost during follow-up. Of the 10 discharged patients who died, 3 had unknown causes, 2 died due to rupture of the distal aortic dissection, 1 died due to accidental cerebrovascular disease, 3 had graft infections, and 2 patients had renal failure that required long-term hemodialysis (Table 6). 29 (24.8%) cases required re-surgery due to aortic dissection recurrence. The probability of not requiring re-surgery for patients (Figure 1) in the David group was 91.7%±8% at 5-year, 78.6%±14% at 10 years, and 58.9%±20% at 15 years. The probability of not requiring re-surgery for patients in the Bentall group was 96%±2.8% at 5 years, 73.1%±9.5% at 10 years, and 58.7%±12.1% at 15 years. For patients in the Bentall + Arch group, the probability of not requiring re-surgery at 5 years was 92.2%±4.4%, at 10 years was 85.6%±6.1%, and at 15 years was 71.5%±10.5%. The probability of not requiring re-surgery for patients in the Arch + Stent group at 5 years was 85.7%±11.7%, at 10 years was 25.0%±15.3%, and at 15 years was 12.5%±11.7% (P=0.007). Concerning long-term survival (Figure 2), there were no deaths in the David group. The 1-year survival rate for patients in the Bentall group was 98.1%±1.9%, the 5-year survival rate was 92.3%±5.9%, the 10-year survival rate was 85.2%±8.7%, and the 15-year survival rate 73%±13.5%. The 1-year survival rate for patients who underwent Bentall + Arch surgery was 96.7%±3.3%, the 5-year survival rate was 92.1%±5.5%, the 10-year survival rate was 82.9%±10%, and the 15-year survival rate was 71%±13.9%. The 1-year survival rate for patients who underwent Arch + Stent surgery was 100%, the 5-year survival rate was 87.5%±11.1%, the 10-year survival rate was 72.9%±16.5%, and the 15-year survival rate was 72.9%±16.5% (P=0.42).

Full table
Discussion
The majority of patients in this study were males. The incidence of aortic dissection and the number of preventive surgeries performed were higher in males (6). With regards to the type of surgical procedure, there were no statistically significant differences in post-surgical hospital mortality. Risk factors, such as NYHA classification, preoperative mitral regurgitation, and preoperative atrial fibrillation, were predictive indicators for valvular lesions in MFS patients during late-stage disease. Aortic root aneurysm expansion induces a large amount of aortic valvular regurgitation, increases left ventricular end-diastolic diameter, leads to left heart failure, and ultimately affects patient survival after surgery (12). MFS patients with heart failure have been associated with mitral valvular lesions. These patients are generally more severe and have been associated with low cardiac output syndrome after surgery. Hence, MFS patients with mitral valvular lesions have a higher surgical risk and need to be carefully prepped during the perioperative period.
However, patients with emergency aortic dissection often do not have enough time to improve their cardiac function. Systemic inflammatory response syndrome is more serious in these patients compared to patients undergoing elective surgery. This is due to the long surgical time, intensive surgery, prolonged cardiopulmonary bypass time, and heavy bleeding that occurs. In addition, coronary artery damage could have a serious effect on survival during hospitalization (12,13). Samadi et al. suggested that elective surgery could be performed instead of emergency surgery if early use of color doppler ultrasound, preventive surgery, and drug therapy was established for MFS patients (14). Hence, early follow-up examinations are recommended for MFS patients. If progressive cardiovascular disease is observed, early surgical intervention should be considered, even if the aortic root diameter is less than 45 mm (15,16).
With regards to postoperative hospital complications, respiratory complications mainly occurred in patients who underwent aortic arch surgery. This may be due to intraoperative circulatory arrest, hypothermic extracorporeal circulation, massive blood transfusion, and systemic inflammatory response syndrome induced by surgical shock in patients undergoing aortic arch surgery, which severely affects the respiratory system (15). Despite advances in surgical techniques in recent years, the risk of aortic arch surgery remains high (17). In contrast, the David (reimplantation) surgical procedure had lower complications and mortality (16,18,19). The number of vocal cord paralysis was only 6 (4.4%) cases in our study and was lower compared to previous studies 26% (20). This may have been because the left common carotid artery and the left subclavian artery were dissociated sufficiently, so that the recurrent laryngeal nerve could be seen clearly, and great care was taken in blocking to avoid damage to the recurrent laryngeal nerve. The incidence of postoperative paraplegia in this study was very low. This could have been due to cerebrospinal fluid drainage tubes being placed on all patients with descending aortic dissection before surgery. This surgical technique reduces spinal cord injury by prolonging the spinal cord ischemic tolerance period and increasing collateral perfusion during and after surgery (21). It should be noted that two (1.5%) patients had mediastinal infections after surgery, which may have been attributed to bacterial strains that were present in the intensive care unit of our hospital. Thoracic drainage procedures performed in patients with aortic dissections may have been responsible for these infections. Ultrasound probes that were placed under the xiphoid during bedside ultrasounds may not have been disinfected before examinations, resulting in infections along the thorax tube that entered the mediastinum.
After long-term follow-up, the overall survival rate was satisfactory with 82.7% of the patients alive 15 years after surgery. Survival rates were higher in patients who underwent proximal (David surgery) aortic interventions, while patients who underwent extensive replacements such as aortic arches had a relatively low 15-year survival rate. Inpatient mortality was similar between the groups, with differences in survival rates mainly related to long-term mortality. A recent study from Johns Hopkins Hospital compared long term follow-up data and found that patients who underwent the David procedure (98 cases) had reduced cardiogenic mortality and valve-related complications compared to patients who underwent the Bentall procedure (67 cases) (8). After adjusting for clinical covariates, the aortic valve-sparing operation was associated with reduced cardiac mortality and valve-related complications compared with aortic root replacement with a mechanical or tissue valve. David operation has made a major difference in the lives of young adults with aortic root aneurysms.
During the follow-up period, we observed that the re-surgical rates of MFS patients undergoing the Bentall or David surgical procedure were higher compared to patients in the arch surgery group. The reason could be that aortic lesions occur naturally in MFS patients. Although the proximal aortic disease was eliminated, the distal disease continued to develop. The reason for re-surgery at the distal end is usually for aortic dissection rather than an aneurysm. This is because the vessel walls of the aortic arch and the distal end are weak and the arterial pressure is high, hence a risk of dissection remains (8). The incidence of aortic dissection recurrence in MSF patients has been reported to range from 5% to 35%, which depends on the scope of the previous surgery and the quality of follow-up (22). It should be noted that, of the 9 patients who underwent arch replacement and elephant trunk stent implantation, 7 underwent Bentall surgery within 15 years after surgery due to aortic root tumors or aortic dissection. In MFS patients with aneurysms, the most common cause of re-surgery was aortic dissection (23). Hence, proximal vascular disease in MFS patients is still the main reason for re-surgery. Because some patients died before re-surgery or refused re-surgery, the true incidence of patients who required re-surgery could be much higher (24).
With the development of endovascular repair and treatment methods from 2014 to 2019, the number of re-surgeries for aortic dissection caused by stent grafts has increased significantly. The continuous longitudinal pressure exerted by the stent graft on the diseased aortic wall creates a risk for aortic rupture and stent-graft movement. Hence, it may not be suitable for MFS patients to undergo endovascular repair (24). Ghanta et al. suggested that stent-grafts should be limited to patients with a high risk of surgery and hence should be closely monitored.
With the improvement of modern medical examination methods and the popularity of MFS testing, the age of hospitalized patients with non-acute aortic dissection has become lower. This highlights the importance of using advanced imaging techniques and routine monitoring after proximal aortic surgery (25,26). Given the reduced aortic valve leaflet toughness and strength in MFS patients, some researchers believe that the David surgical procedure is not suitable for the treatment of MFS patients. However, some researchers believe that aortic valvular organic lesions are rare in MFS patients, and the David surgical procedure could produce better long-term results (27). From our short-term and long-term follow-up data, we found that 14 patients who underwent the David procedure in our center achieved satisfactory results. However, due to the small number, the research results still need to be further supported by multi-center data.
Conclusions
In this study, MFS patients were followed-up for an extended period. The prognosis of MFS patients who underwent different types of cardiovascular surgical procedures was then evaluated. The limitation of this study was that it was a single-center study with a relatively small cohort of patients. In addition, patient quality of life was not measured during the long-term follow-up period. Additional multi-center studies should be performed using larger patient cohorts to validate our findings.
MFS patients underwent different surgical procedures due to different arterial lesions, and the results during hospitalization were not significantly different. When an aneurysm or dissection is confined to the proximal aorta, intervention on the proximal aorta alone could still achieve high long-term survival rates. However, we observed from long-term follow-ups that the risk of re-surgeries due to distal aortic dissection was high. The benefits of elective surgery were the highest for patients who underwent the David surgical procedure. This was because the complications caused by long-term use of anticoagulants could be avoided and hence long-term survival rates were high. Satisfactory results could be obtained by constant follow-ups after surgery so that timely interventional treatments for possible distal dissection aneurysms could be performed.
Acknowledgments
Funding: This study was funded by Shanghai Committee of Science and Technology, China (Grant No. 2014ZYJB0401).
Footnote
Reporting Checklist: The authors have completed the STROBE reporting checklist. Available at http://dx.doi.org/10.21037/jtd-20-2109
Data Sharing Statement: Available at http://dx.doi.org/10.21037/jtd-20-2109
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/jtd-20-2109). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The retrospective study was approved by the Ethics Committee of Changhai Hospital affiliated to Naval Medical University (No. 20200189) and informed consent was taken from all the patients. The study was conformed to the provisions of the Declaration of Helsinki (as revised in 2013).
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Schoenhoff FS, Jungi S, Czerny M, et al. Acute aortic dissection determines the fate of initially untreated aortic segments in Marfan syndrome. Circulation 2013;127:1569-75. [Crossref] [PubMed]
- Price J, Magruder JT, Young A, et al. Long-term outcomes of aortic root operations for Marfan syndrome: A comparison of Bentall versus aortic valve-sparing procedures. J Thorac Cardiovasc Surg 2016;151:330-6. [Crossref] [PubMed]
- Murdoch JL, Walker BA, Halpern BL, et al. Life expectancy and causes of death in the Marfan syndrome. N Engl J Med 1972;286:804-8. [Crossref] [PubMed]
- Finkbohner R, Johnston D, Crawford ES, Coselli J, Milewicz DM. Marfan syndrome. Long-term survival and complications after aortic aneurysm repair. Circulation 1995;91:728-33. [Crossref] [PubMed]
- Knadler JJ, LeMaire S, McKenzie ED, et al. Thoracic Aortic, Aortic Valve, and Mitral Valve Surgery in Pediatric and Young Adult Patients With Marfan Syndrome: Characteristics and Outcomes. Semin Thorac Cardiovasc Surg 2019;31:818-25. [Crossref] [PubMed]
- Treasure T, King A, Hidalgo Lemp L, et al. Developing a shared decision support framework for aortic root surgery in Marfan syndrome. Heart 2018;104:480-6. [Crossref] [PubMed]
- Maddalo S, Beller J, DeAnda A. A Bentall Is Not a Bentall Is Not a Bentall: The Evolution of Aortic Root Surgery. Aorta (Stamford) 2014;2:169-78. [Crossref] [PubMed]
- David TE, David CM, Feindel CM, et al. Reimplantation of the aortic valve at 20 years. J Thorac Cardiovasc Surg 2017;153:232-8. [Crossref] [PubMed]
- Svensson LG, Adams DH, et al. Aortic valve and ascending aorta guidelines for management and quality measures: executive summary. Ann Thorac Surg 2013;95:1491-505. [Crossref] [PubMed]
- Patel ND, Weiss ES, et al. Aortic root operations for Marfan syndrome: a comparison of the Bentall and valve-sparing procedures. Ann Thorac Surg 2008;85:2003-10; discussion 2010-1. [Crossref] [PubMed]
- Hickey GL, Dunning J, Seifert B, et al. Statistical and data reporting guidelines for the European Journal of Cardio-Thoracic Surgery and the Interactive CardioVascular and Thoracic Surgery. Eur J Cardiothorac Surg 2015;48:180-93. [Crossref] [PubMed]
- Pyeritz RE. Recent progress in understanding the natural and clinical histories of the Marfan syndrome. Trends Cardiovasc Med 2016;26:423-8. [Crossref] [PubMed]
- Estrera AL, Miller CC 3rd, Villa MA, et al. Proximal reoperations after repaired acute type A aortic dissection. Ann Thorac Surg 2007;83:1603-8; discussion 1608-9. [Crossref] [PubMed]
- Samadi A, Detaint D, Roy C, et al. Surgical management of patients with Marfan syndrome: evolution throughout the years. Arch Cardiovasc Dis 2012;105:84-90. [Crossref] [PubMed]
- Esaki J, Leshnower BG, Binongo JN, et al. Risk Factors for Late Aortic Valve Dysfunction After the David V Valve-Sparing Root Replacement. Ann Thorac Surg 2017;104:1479-87. [Crossref] [PubMed]
- de Kerchove L, Boodhwani M, Glineur D, et al. Valve sparing-root replacement with the reimplantation technique to increase the durability of bicuspid aortic valve repair. J Thorac Cardiovasc Surg 2011;142:1430-8. [Crossref] [PubMed]
- Omura A, Tanaka A, Miyahara S, et al. Early and late results of graft replacement for dissecting aneurysm of thoracoabdominal aorta in patients with Marfan syndrome. Ann Thorac Surg 2012;94:759-65. [Crossref] [PubMed]
- Urbanski PP, Luehr M, Di Bartolomeo R, et al. Multicentre analysis of current strategies and outcomes in open aortic arch surgery: heterogeneity is still an issue. Eur J Cardiothorac Surg 2016;50:249-55. [Crossref] [PubMed]
- Vallabhajosyula P, Szeto WY, Habertheuer A, et al. Bicuspid Aortic Insufficiency With Aortic Root Aneurysm: Root Reimplantation Versus Bentall Root Replacement. Ann Thorac Surg 2016;102:1221-8. [Crossref] [PubMed]
- Lodewyks CL, White CW, Bay G, et al. Vocal Cord Paralysis After Thoracic Aortic Surgery: Incidence and Impact on Clinical Outcomes. Ann Thorac Surg 2015;100:54-8. [Crossref] [PubMed]
- Wynn M, Acher C, Marks E, et al. The effect of intercostal artery reimplantation on spinal cord injury in thoracoabdominal aortic aneurysm surgery. J Vasc Surg 2016;64:289-96. [Crossref] [PubMed]
- Puluca N, Burri M, Cleuziou J, et al. Consecutive operative procedures in patients with Marfan syndrome up to 28 years after initial aortic root surgery. Eur J Cardiothorac Surg 2018;54:504-9. [Crossref] [PubMed]
- Carrel T, Beyeler L, Schnyder A, et al. Reoperations and late adverse outcome in Marfan patients following cardiovascular surgery. Eur J Cardiothorac Surg 2004;25:671-5. [Crossref] [PubMed]
- Hui SK, Fan CS, Christie S, et al. The aortic root does not dilate over time after replacement of the aortic valve and ascending aorta in patients with bicuspid or tricuspid aortic valves. J Thorac Cardiovasc Surg 2018;156:5-13.e1. [Crossref] [PubMed]
- Luehr M, Peterss S, Zierer A, et al. Aortic events and reoperations after elective arch surgery: incidence, surgical strategies and outcomes. Eur J Cardiothorac Surg 2018;53:519-24. [Crossref] [PubMed]
- Kobuch R, Hilker M, Rupprecht L, et al. Late reoperations after repaired acute type A aortic dissection. J Thorac Cardiovasc Surg 2012;144:300-7. [Crossref] [PubMed]
- Shrestha ML, Beckmann E, Abd Alhadi F, et al. Elective David I Procedure Has Excellent Long-Term Results: 20-Year Single-Center Experience. Ann Thorac Surg 2018;105:731-8. [Crossref] [PubMed]