Primary pericardial synovial sarcoma
Introduction
Primary synovial sarcoma is a rare malignant soft tissue tumor that commonly occurs in the vicinity of the extremities of young adults. While broad surgical resection is the first choice for treatment, chemotherapy and radiotherapy are carried out when surgical resection is unsuccessful; however, the prognosis of this disease is poor. We herein report our experience with an extremely rare case of synovial sarcoma occurring in the pericardium.
Case report
A 57-year-old man was admitted to our hospital with cardiomegaly on a chest roentgenogram and signs of cardiac tamponade on echocardiography.
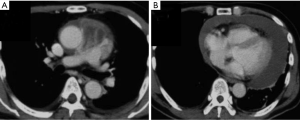
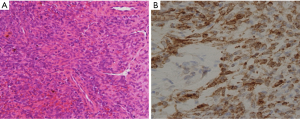
Furthermore, a mediastinal tumor was observed directly above the pericardium on a chest computed tomographic scan (Figure 1). As a result of a tumor biopsy through parasternotomy, spindle-shaped tumor cells were observed, and the results from an immunohistochemical analysis showed the expression of epithelial membrane, keratin, CD99, and bcl-2 antigen (Figure 2). A cytogenetic analysis for t(X;18) by through a reverse transcriptase-polymerase chain reaction analysis of SYT/SSX transcripts by was negative. According to these findings, surgical resection through median sternotomy was performed with a diagnosis of pericardial synovial sarcoma; however, while the tumor developed in the center of the pericardium, it further invaded the basal part of the pulmonary artery, leading to incomplete resection. Subsequently, three cycles of ifosfamide (3 g/m2/d on three subsequent days for 3-week intervals) and adriamycin (20 mg/m2, 3 times every 3 weeks) were administered along with radiation therapy (50 Gy). As a result, the tumor size temporarily reduced (reduction rate: approximately 32%), and exclusion of the pulmonary artery improved. However, the patient refused further therapy, and the tumor gradually increased. Consequently, the patient died approximately 3 years following the initial medical examination. No autopsy was performed.
Discussion
Synovial sarcoma often occurs in patients aged 10 to 40 years of age, with most occurring in the vicinity of the joints of the extremities, such as the joint capsule, synovial bursa, tendon sheath, and tendons (1). However, occurrence in the head and neck, as well as the torso, which are unrelated to these tissues, may be observed. Among these cases, only a few case reports have been reported on pericardial synovial sarcoma (2). A definite diagnosis of synovial sarcoma is generally carried out by a biopsy; however, the histopathological images widely vary, and thus the diagnosis is not necessarily simple. Immunohistochemically, the epithelial component reflects true epithelial differentiation, while keratin, which is an endothelial marker, becomes positive; moreover, the occurrence of endothelial markers at varying degrees is observed in the spindle system cells as well (3,4). Furthermore, in recent cytogenetic analyses using established techniques, the chimeric genes SYT/SSX1 or SYT/SSX2 derived from the characteristic translocation of chromosome t(X;18) (p11; q11) are observed in most synovial sarcomas; therefore, a genetic diagnosis is also currently becoming more useful (5). In our case, histology was carried out by a tumor biopsy via a parasternal incision, with a sarcoma suspected according to hyperplasia of spindle-shaped or oval tumor cells upon hematoxylin and eosin staining. On the immunohistochemical analysis, keratin and epithelial membrane antigen were positive among the endothelial markers, α-smooth muscle actin and actin antigen (which are muscular markers) were positive among non-epithelial markers, while CD56 was positive along with neurofilament, and S100 antigen was slightly positive among the neurological markers. The patient was diagnosed with haplotype synovial sarcoma according to the shape of the tumor and the finding that the tumor was endothelial, with non-endothelial markers positive upon immunostaining; however, the cytogenetic analysis for t(X;18) by demonstration of SYT/SSX transcripts and SYT/SSX2 was negative.
Broad surgical resection is the optimal standard treatment for synovial sarcomas; however, this treatment leaves the patient prone to local recurrence and distant metastasis, with a poor prognosis of a 5-year survival rate of 35% to 50%. Therefore, concomitant chemotherapy is actively carried out. Regarding chemotherapy, the therapeutic effect using a single drug is generally low, thus combination chemotherapy using dacarbazine, vincristine, and cisplatin with mainly ifosfamide and adriamycin are attempted (1,2). In the present case, retention of pericardial effusion and invasion to the pulmonary artery was already observed at the initial examination, thus broad surgical resection with negative margins was impossible. Therefore, chemotherapy and radiotherapy were carried out; however, although the tumor dramatically decreased in size, the patient ultimately died approximately 3 years following the initial examination. Currently, there are no standard treatments established for synovial sarcoma, and it is believed that an early detection and diagnosis, in addition to complete resection, are necessary to obtain a long-term survival.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
- Spillane AJ, A’Hern R, Judson IR, et al. Synovial sarcoma: a clinicopathologic, staging, and prognostic assessment. J Clin Oncol 2000;18:3794-803. [PubMed]
- Al-Rajhi N, Husain S, Coupland R, et al. Primary pericardial synovial sarcoma: a case report and literature review. J Surg Oncol 1999;70:194-8. [PubMed]
- Suster S, Fisher C, Moran CA. Expression of bcl-2 oncoprotein in benign and malignant spindle cell tumors of soft tissue, skin, serosal surfaces, and gastrointestinal tract. Am J Surg Pathol 1998;22:863-72. [PubMed]
- van de Rijn M, Hendrickson MR, Rouse RV. CD34 expression by gastrointestinal tract stromal tumors. Hum Pathol 1994;25:766-71. [PubMed]
- Kawai A, Woodruff J, Healey JH, et al. SYT-SSX gene fusion as a determinant of morphology and prognosis in synovial sarcoma. N Engl J Med 1998;338:153-60. [PubMed]


