A successful percutaneous mechanical vegetation debulking used as a bridge to surgery in acute tricuspid valve endocarditis
Introduction
The first line treatment for tricuspid valve endocarditis is medical treatment (1). Intravenous drug users with endocarditis present many issues not encountered in other endocarditis patients, including the high potential for recurrence due to addiction, poor compliance with therapy after mechanical valve replacement, and the risk of recurrence of endocarditis of the new valve (2). Here we present a case report of successful percutaneous transcatheter mechanical vegetation debulking followed by surgical valve replacement.
Case presentation
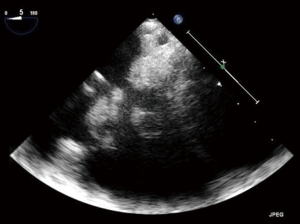
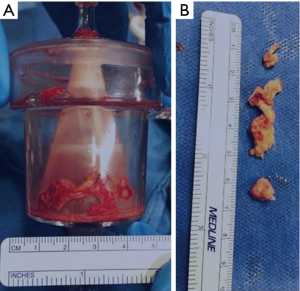
A 24-year-old previously healthy male with a history of intravenous drug abuse presented to an outside hospital with shorten of breath, mottled skin, lethargy, and lower extremities petechiae. A chest computed tomography (CT) eventually showed bilateral images suspicious of pulmonary infarcts. A brain magnetic resonance imaging (MRI) images suggests of brain infarct in the right parietal lobe. A transthoracic then transesophageal echocardiogram revealed large, mobile vegetations on the tricuspid valve (Figure 1) with severe tricuspid regurgitation. The blood cultures grew staph aureus was started on vancomycin 3 days later it was converted to Quinupristin/Dalfopristin for Methicillin-Resistant Staphylococcus Aureus (MRSA) bacteremia; however he continued to be septic, developing high fevers, chills with continuous positive culture, he developed hemodynamic instability led to intubation and inotropes were initiated. Subsequently he was transferred to our hospital for further management. Upon arrival, the patient was febrile and septic, he was requiring mechanical ventilatory support, and inotrope support, anuric in acute renal failure with creatinine of 4.4, and he had developed multi-organ failure. TTE on admission showed severe tricuspid valve regurgitation with bulky vegetation measuring 0.9 cm × 0.7 cm on the septal and 0.7 cm × 1 cm on the anterior leaflet, all other valves were of normal function and appearance. The patient was considered to be at high risk for surgical complications due hemodynamic instability, multi-organ failure, and the possibility of recurrence of endocarditis with the persistent sepsis and the positive cultures. A multi-disciplinary team decision was made to proceed with percutaneous transcatheter mechanical vegetectomy as a bridge to surgical valve replacement. The procedure was performed in hybrid operating room. A Vortex AngioVac™ (Vortex Inc.), the cannula was inserted via the right internal jugular vein and a reinfusion cannula was inserted via the right common femoral vein. Under fluoroscopic and transesophageal echocardiographic guidance, the vegetations were suctioned. In order to clear residual, adherent vegetations, a snaring device was introduced via a separate right internal jugular sheath and used to free pieces of vegetations from the septal side of the tricuspid annulus valve which were then captured by the AngioVac™ suction cannula. Approximate 80% of the vegetation load was removed (Figure 2). Post procedure transesophageal echocardiogram (TEE) continued to demonstrate severe tricuspid regurgitation with significant reduction of vegetation burden (Figure 3). The fevers were resolved, and subsequent blood cultures returned negative, the patient was extubated 2 days later, and as he became hemodynamically stable and the inotrope were weaned off. Seven days later, he underwent an open tricuspid valve replacement with a porcine valve as the tricuspid valve was not amenable for repair due to the endocarditis and the debulking procedure. His post-operative course was uneventful, and he was discharged to a rehabilitation facility 11 days post-surgery. Follow-up echocardiography and blood cultures at 12 months are without evidence of recurrent endocarditis.
Discussion
Isolated right-sided endocarditis constitutes 5–10% of all cases of endocarditis (3). Among these approximately 20% requires surgical intervention. Large vegetations are common in patients with right-sided endocarditis because of the lower pressure of the right-sided heart chambers, which might be the source of septic pulmonary embolisms (4) as in our case. There is limited published data for management of active infection (2-5). Valvectomy without replacement was presented as a possible option in 1971 by Arbulu (5), which leaves the patient with iatrogenic open tricuspid regurgitation. There are some case reports of percutaneous removal of cardiac lead vegetations (6) and aright atrial thrombus (7). Divekar et al. (8) reported a case of percutaneous vegetation debulking as a bridge to surgery for refractory infective endocarditis of a bioprosthetic pulmonary valve.
This case introduces a new option in the management of right-sided endocarditis in critically ill patient with high operative morbidities. By debulking the infectious site, and achieving lower bacterial load more effective antibiotic efficacy could be achieved, this allows for clearing of the blood stream by continuing effective antibiotics treatment, which reduces the risk of post-operative recurrence. In addition, resolution of the septic state allowed for improved hemodynamics in preparation for surgical, decreased inflammation of the infected annulus may also allow for better technical implantation and healing of the valve and annulus. One of the limitation of this procedure is the risk of pulmonary embolization of the vegetation fragments this can be decrease by using the aspiration and a good manipulation of the aspiration cannula to be adjacent to the snaring device. This case is the first reported case of the use of percutaneous transcatheter mechanical vegetectomy aspiration and snaring as a bridge to surgery in acute tricuspid valve endocarditis. This paper could be considered as a “proof of concept” of a debulking/debridement procedure in a patient who is critically ill and high associated surgical risks and complications. This procedure subsequently led to improvement in patient’s condition, and he was able to undergo definitive surgery in better operative condition. It also represents an example of a multispecialty team approach to the management of a complex patient to achieve a successful outcome.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
- Baddour LM, Wilson WR, Bayer AS, et al. Infective endocarditis: diagnosis, antimicrobial therapy, and management of complications: a statement for healthcare professionals from the Committee on Rheumatic Fever, Endocarditis, and Kawasaki Disease, Council on Cardiovascular Disease in the Young, and the Councils on Clinical Cardiology, Stroke, and Cardiovascular Surgery and Anesthesia, American Heart Association: endorsed by the Infectious Diseases Society of America. Circulation 2005;111:e394-434. [PubMed]
- Byrne JG, Rezai K, Sanchez JA, et al. Surgical management of endocarditis: the society of thoracic surgeons clinical practice guideline. Ann Thorac Surg 2011;91:2012-9. [PubMed]
- Musci M, Siniawski H, Pasic M, et al. Surgical treatment of right-sided active infective endocarditis with or without involvement of the left heart: 20-year single center experience. Eur J Cardiothorac Surg 2007;32:118-25. [PubMed]
- Cook RJ, Ashton RW, Aughenbaugh GL, et al. Septic pulmonary embolism: presenting features and clinical course of 14 patients. Chest 2005;128:162-6. [PubMed]
- Arbulu A, Thoms NW, Chiscano A, et al. Total tricuspid valvulectomy without replacement in the treatment of Pseudomonas endocarditis. Surg Forum 1971;22:162-4. [PubMed]
- Patel N, Azemi T, Zaeem F, et al. Vacuum assisted vegetation extraction for the management of large lead vegetations. J Card Surg 2013;28:321-4. [PubMed]
- Nickel B, McClure T, Moriarty J. A Novel Technique for Endovascular Removal of Large Volume Right Atrial Tumor Thrombus. Cardiovasc Intervent Radiol 2015;38:1021-4. [PubMed]
- Divekar AA, Scholz T, Fernandez JD. Novel percutaneous transcatheter intervention for refractory active endocarditis as a bridge to surgery-angiovac aspiration system. Catheter Cardiovasc Interv 2013;81:1008-12. [PubMed]