Peri-operative outcomes of bariatric surgery in obstructive sleep apnoea: a single-centre cohort study
Highlight box
Key findings
• In patients undergoing bariatric surgery, there is a similar rate of post-operative complication rates between those with significant OSA prescribed CPAP and those without OSA.
What is known and what is new?
• Post-operative complication rates are higher in patients with obesity with concomitant OSA compared to those without.
• The use of CPAP in the post-operative period in patients with obesity and OSA undergoing bariatric surgery mitigates the increased risk of complications in this period, when compared with obese patient without OSA.
What is the implication, and what should change now?
• Screening for OSA in patients undergoing bariatric surgery should become routine practice. Those identified as having clinically significant OSA should be provided CPAP in the post-operative period to mitigate the increased risk of complications.
Introduction
Obesity affects 13% of adults worldwide and its prevalence is predicted to increase (1). 2019 data from England showed that 36% of adults over the age of 16 were classified as overweight and 28% as obese (2). The prevalence of sleep-disordered breathing (SDB), including obstructive sleep apnoea (OSA), is higher in patients eligible for bariatric surgery than with normal weight (3,4), with a prevalence of around 70% (3,5).
OSA is characterised by repetitive desaturations during sleep due to obstruction of the upper airway, causing repetitive arousals, sleep fragmentation and resulting in daytime symptoms (e.g., excessive daytime sleepiness) (6). Management of OSA involves lifestyle modifications, weight loss, and primary airway therapy [e.g., mandibular advancement devices (MAD) or continuous positive airway pressure (CPAP)].
Substantial weight loss can significantly reduce the severity of OSA in patients living with obesity, and sometimes resolve it entirely (5). Following bariatric surgery, resolution or improvement of OSA is seen in over 75% of patients (7). In the UK, the National Institute for Health and Care Excellence (NICE) guidelines recommend bariatric surgery as an option for patients with a body mass index (BMI) above 40 kg/m2, or 35–40 kg/m2 and another significant co-morbidity, such as hypertension, after failing to achieve or maintain clinically significant weight loss following non-surgical interventions (8).
Guidelines recommend pre-operative screening for OSA prior to bariatric surgery (9), however the impact of OSA on peri-operative outcomes remains unclear. Most of the evidence suggests that patients with OSA are at higher risk of cardiovascular and respiratory complications, as well as transfer to intensive care units (ICU) (10,11) following surgery compared to patients without OSA. These include postoperative hypoxaemia (12), respiratory infections, atrial fibrillation, intubation and ventilation (13), as well as longer length of stay (LOS) (12). However, some studies found no difference between those two groups with regards to the frequency of ICU admissions postoperatively, overall LOS, and post-operative complications (14,15), or even found a slight benefit in those with OSA on CPAP (16). The use of primary airway therapy (e.g., CPAP) in patients with OSA in the peri-operative setting is felt to protect against some of the complications seen following surgery (14), most importantly cardiovascular and respiratory events, which seem to be more frequent with increasing severity of OSA (13).
We sought to analyse peri-operative outcomes (overall LOS, respiratory and non-respiratory post-operative complications, maximum level of respiratory support, the highest level of care, and mortality) of patients living with obesity undergoing bariatric surgery in our tertiary referral centre and compared clinical outcomes in those patients who had OSA and required CPAP therapy to those who did not. Here’s a video abstract to this study (Video 1). We present the following article in accordance with the STROBE reporting checklist (available at https://jtd.amegroups.com/article/view/10.21037/jtd-22-1501/rc).
Methods
Data collection
The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). This was a retrospective study registered as a service review and approved by the institutional review board of Guy’s and St Thomas’ NHS Foundation Trust (Approval No. LIRB: 2021/12240) and individual consent for this retrospective analysis was waived. Data were collected from the electronic patient records of all patients referred to the Lane Fox Unit (Respiratory High Dependency Unit, St Thomas’ Hospital, London, UK) and to the Sleep Disorders Centre (Guy’s Hospital, London, UK) for pre-operative screening of SDB prior to bariatric surgery (June 2014 to February 2021). The strategy employed in the pre-operative screening process has been outlined elsewhere (3). Patients referred for bariatric surgery completed a STOP-BANG questionnaire and those who scored >4 points were referred for a nocturnal pulse oximetry for two consecutive nights (3). Data collected included demographic data, sleep study data, co-morbidities, post-operative complications, maximal level of respiratory support, LOS, mortality, and the highest level of care (Appendix 1). Respiratory support data were included only if there was available documentation for the daily oxygen requirements during the whole inpatient stay. Complications are presented according to the Clavien-Dindo classification of post-operative complications. The follow up period was until discharge from hospital and these data were obtained from the electronic patient records.
All sleep data were analysed and reviewed by a sleep technician and a physician with a specialist interest in sleep who recommended the appropriate treatment. OSA was classified based on the 4% oxygen desaturation index (ODI); it was mild with an ODI of 5.0–14.9 events/hour, moderate with an ODI of 15.0–29.9 events/hour, and an ODI of 30 events/hour and above classified as severe OSA. Patients were generally offered CPAP therapy if they had moderate or severe OSA, or mild OSA with significant associated symptoms. Patients with no OSA, or mild OSA without significant symptoms were not offered CPAP therapy. The CPAP device offered was a ResMed AirSense 10 Autoset (ResMed, San Diego, California, USA) and the non-invasive ventilation (NIV) device offered was a ResMed Lumis 150 VPAP ST-A (ResMed, San Diego, California, USA).
In- and exclusion criteria
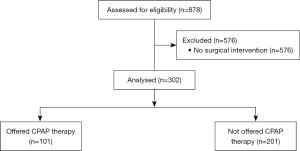
Cases were included if they were older than 18 years and had been referred for bariatric surgery and undergone pre-operative screening for OSA. Cases were excluded if they had not had their surgery at the time of data collection (Figure 1).
Outcomes
The primary outcome parameters were the frequency of post-operative complications and the maximum level of respiratory support required during the inpatient stay. Secondary outcomes were the overall LOS, the maximum level of care received during the inpatient stay, and mortality.
Statistical analysis
Data are presented as mean [standard deviation (SD)], or median [interquartile range (IQR)] following testing for normality. Statistical significance was calculated using Student’s t-test for continuous variables, and a chi-squared test or a Mann-Whitney U test for non-continuous variables. Data were collected and analysed using Microsoft Excel v16.43 (Microsoft Corporation, Washington, USA) and SPSS (Version 28.0.1.0, IBM, New York, USA). Statistical significance was defined as P value of <0.05. Where data were missing, calculations were performed with the subjects with available data only.
Results
A total of 878 patients were referred for assessment, of which 302 patients underwent bariatric surgery during the observation period (Figure 1). Of the 302 patients, 34.1% had no OSA, 33.8% had mild, 17.2% moderate, and 11.6% severe OSA. 3.3% had concomitant OSA and obesity hypoventilation syndrome (OHS). A total of 101 (33.4%) patients had either moderate or severe OSA, or had mild OSA with significant symptoms, and were offered CPAP. Two hundred and one (66.6%) patients either had no OSA, or had mild OSA without significant symptoms, and were not offered CPAP (Table 1).
Table 1
| Demographics and sleep data | CPAP (n=101) | Non-CPAP (n=201) | P value |
|---|---|---|---|
| Age, years, mean (SD) | 50 (10.0) | 46 (12.0) | 0.004 |
| Male sex, n (% total cohort) | 34 (33.7) | 30 (14.9) | <0.001 |
| BMI, kg/m2, mean (SD) | 50.2 (9.0) | 47.0 (6.9) | 0.002 |
| Pre-operative weight, kg, mean (SD) | 142.2 (29.3) | 131.0 (24.1) | 0.001 |
| Weight loss at 12 months, kg, mean (SD) | 40.3 (18.6) | 31.0 (16.1) | 0.003 |
| Weight loss at 12 months, % pre-operative weight, mean (SD) | 27.5 (9.2) | 23.3 (10.5) | 0.015 |
| No OSA, n (% total cohort) | 0 | 103 (51.2) | <0.001 |
| Mild OSA, n (% total cohort) | 4 (4.0) | 98 (48.8) | <0.001 |
| Moderate OSA, n (% total cohort) | 52 (51.5) | 0 | <0.001 |
| Severe OSA, n (% total cohort) | 35 (34.7) | 0 | <0.001 |
| Combined OSA & OHS, n (% total cohort) | 10 (9.9) | 0 | <0.001 |
| Epworth Sleepiness Score, points, mean (SD) | 9.7 (4.9) | 6.9 (4.6) | <0.001 |
| Mean SpO2, %, mean (SD) | 92.1 (4.5) | 95.5 (1.2) | <0.001 |
| 3% ODI, events/hour (SD) | 37.7 (22.8) | 8.8 (4.7) | <0.001 |
| 4 % ODI, events/hour (SD) | 29.8 (21.0) | 5.7 (3.2) | <0.001 |
| PRI, events/hour (SD) | 42.7 (20.8) | 29.2 (14.3) | <0.001 |
| Time < SpO2 of 90%, % total sleep study (SD) | 17.6 (18.8) | 1.4 (2.0) | <0.001 |
CPAP, continuous positive airway pressure; BMI, body mass index; SD, standard deviation; OSA, obstructive sleep apnoea; OHS, obesity hypoventilation syndrome; ODI, oxygen desaturation index; PRI, pulse rise index; SpO2, peripheral blood oxygen saturations; time < SpO2 of 90%, time spent with oxygen saturations less than 90% as a percentage of the total sleep study time.
CPAP vs. no CPAP
Of the 101 patients in the CPAP group, 85 (84.2%) were prescribed long-term CPAP, seven (6.9%) were recommended to be extubated onto CPAP following surgery, and nine (8.9%) were referred for further assessment due to suspected OHS. Of those, seven were prescribed CPAP and two were established on NIV. Of the 201 patients in the non-CPAP group, 184 (91.5%) were prescribed no airway therapy, and 17 (8.5%) were recommended a MAD independent of any surgery. Patients requiring CPAP therapy were older, more frequently male, had a higher BMI, and lost more weight following bariatric intervention than those in the non-CPAP group (Table 1).
Comorbidities
The most common comorbidities were hypertension and type 2 diabetes, all of which were more frequent in the CPAP group. Anxiety and depression, asthma and gastro-oesophageal reflux disease were similarly common in both groups (Table 2).
Table 2
| Co-morbidity | CPAPa (n=101) | Non-CPAPa (n=201) | P value |
|---|---|---|---|
| Hypertension | 47 (46.5) | 54 (26.9) | <0.001 |
| Type 2 diabetes mellitus | 43 (42.6) | 58 (28.9) | 0.017 |
| Depression and/or anxiety | 35 (34.7) | 51 (25.4) | 0.09 |
| Asthma | 11 (10.9) | 18 (9.0) | 0.59 |
| GORD | 8 (7.9) | 8 (4.0) | 0.15 |
a, data are presented as total numbers of patients with the comorbidity (% of total cohort). CPAP, continuous positive airway pressure; GORD, gastro-oesophageal reflux disease.
Type of surgery
The most frequent surgical intervention was a sleeve gastrectomy (n=170, 56.3% of total cohort), followed by roux-en-y gastric bypass (n=112, 37.1%), gastric band insertion (n=17, 5.6%), and three (1.0%) gastric band removals due to intolerable side-effects. There was no significant difference in the type of surgery undergone between both groups (Table 3).
Table 3
| Type of surgery | CPAPa (n=101) | Non-CPAPa (n=201) | P value |
|---|---|---|---|
| Sleeve gastrectomy | 55 (54.5) | 115 (57.2) | 0.65 |
| RYGB | 43 (42.6) | 69 (34.3) | 0.16 |
| Gastric band | 2 (2.0) | 15 (7.5) | 0.05 |
| Gastric band removal | 1 (1.0) | 2 (1.0) | 0.997 |
a, data are presented as the total number of patients undergoing each surgery (% of total cohort). CPAP, continuous positive airway pressure; RYGB, Roux-en-Y gastric bypass.
Post-operative complications
The observed post-operative respiratory complications included respiratory infections, exacerbation of airways disease, and development of acidotic hypercapnic respiratory failure requiring the use of acute NIV. Non-respiratory complications included acute kidney injury (AKI), acute liver injury (ALI), upper gastrointestinal bleeding, intra-abdominal bleeding, and wound infections requiring antibiotic therapy.
There was no between group difference in the rate of individual complications. However, when analysed according to the Clavien-Dindo grades there was a difference in grade I complications that were more frequent in the CPAP group, with no difference in the grade II and IVa complications. There were no complications for the other grades. No patient required any organ support and there were no deaths during the follow up period in either cohort (Table 4).
Table 4
| Post-operative complication | CPAP (n=89)a | Non-CPAP (n=190)a | P value |
|---|---|---|---|
| Grade 1 (total combined) | 8 (9.0) | 5 (2.6) | 0.02 |
| AKI | 3 (3.4) | 1 (0.5) | 0.06 |
| Upper GI bleed | 1 (1.1) | 1 (0.5) | 0.58 |
| Intra-abdominal bleed | 2 (2.2) | 1 (0.5) | 0.19 |
| Acute liver injury | 2 (2.2) | 2 (1.1) | 0.43 |
| Grade II (total combined) | 4 (4.5) | 4 (2.1) | 0.27 |
| Intra-abdominal bleed | 1 (1.1) | 2 (1.1) | 0.96 |
| Respiratory infection | 2 (2.2) | 1 (0.5) | 0.19 |
| Wound infection | 1 (1.1) | 0 | 0.14 |
| Exacerbation of OAD | 0 | 1 (0.5) | 0.49 |
| Grade IVa (total combined) | 3 (3.4) | 1 (0.5) | 0.06 |
| T2RF | 1 (1.1) | 0 | 0.14 |
| Intra-abdominal bleed | 1 (1.1) | 0 | 0.14 |
| Respiratory infection | 1 (1.1) | 1 (0.5) | 0.58 |
a, data are presented as total number of patients with each complication (% of total cohort with available data). CPAP, continuous positive airway pressure; AKI, acute kidney injury; GI, gastrointestinal; OAD, obstructive airways disease; T2RF, type 2 respiratory failure.
Respiratory support
Patients in the CPAP group patients more frequently required the composite of any oxygen or respiratory support than those in the non-CPAP group (60.6% vs. 26.3%, P<0.001) (Table 5).
Table 5
| Respiratory support | CPAPa (n=33) | Non-CPAPa (n=76) | P value |
|---|---|---|---|
| No oxygen | 13 (39.4) | 56 (73.7) | <0.001 |
| Nasal cannula | 15 (45.5) | 17 (22.4) | 0.02 |
| Other oxygen | 3 (9.1) | 2 (2.6) | 0.14 |
| CPAP | 1 (3.0) | 1 (1.3) | 0.54 |
| NIV | 1 (3.0) | 0 | 0.13 |
| Reintubation | 0 | 0 | n/a |
a, data are presented as total number of patients requiring each level of support (% of total cohort with available data). CPAP, continuous positive airway pressure; NIV, non-invasive ventilation; n/a, not applicable.
Ward level requirements and LOS
The majority of patients were managed in a general surgical ward and there was no between group difference (P=0.06): 85 (96.6%) and 187 (99.5%) patients were managed in a general surgical ward, whilst three (4.5%) and one (0.5%) required unplanned ICU level care, in the CPAP (one patient for severe type 1 respiratory failure due to infection requiring high-flow oxygen, one due to de novo hypercapneic respiratory failure requiring NIV, and one due to major intra-abdominal bleeding) and non-CPAP (one patient developed severe type 1 respiratory failure due to respiratory infection requiring high-flow oxygen) groups, respectively. The overall median LOS was 2 (1.0) days, with patients in the CPAP cohort having a longer median LOS [3 (1.5) vs. 2 (1.0) days; P=0.002].
Discussion
About one third of the patients undergoing bariatric surgery required respiratory support with CPAP for OSA. Those with a higher BMI and symptom burden were more likely to have significant OSA requiring CPAP therapy. They were also more likely to have comorbidities, but also benefitted more from the intervention, as they lost more weight independent of the type of surgery. In this study, the number of peri-operative complications following bariatric surgery was low, with a rate of 2–3%, and balanced between the two groups, with only minor complications being more common in patients with more severe OSA and CPAP therapy. Patients prescribed CPAP therapy were more likely to require supplemental oxygen postoperatively. Although there was no difference in the required level of ward care between the two groups, patients with CPAP therapy had a slightly longer LOS.
Clinical significance
Patients with OSA seem to be at higher risk of post-operative complications than those without OSA, with respiratory and cardiovascular complications predominating (10,11). Post-operative hypoxaemia and ventilatory disturbances are common in the general surgical population (17). Several factors, including pre-operative apnoea-hypopnoea index, have been associated with the severity of post-operative SDB (18). Increased airway collapsibility and airway oedema in patients with untreated OSA are likely to pose added difficulties to the anaesthetist for intubation (19) and ventilation (20), with OSA an independent risk factor for both (21). Anaesthetic drugs exacerbate these problems by reducing muscle tone, impairing the arousal response, and reducing ventilatory drive (22). Optimal positioning at induction, extubating patients when they are completely or almost awake, and CPAP use in the immediate post-operative period, are recommended by consensus guidelines (9).
A systematic review of studies assessing peri-operative complications of bariatric and non-bariatric surgery described associations between OSA and hypoxaemia and reintubation (22), whilst in another study there was no impact of prior CPAP use on outcomes following non-cardiac surgery, albeit post-operative CPAP use was not assessed (12). Pre-operative use of CPAP may help reduce post-operative complications through a reduction in airway oedema (23). Having at least mild OSA with clinical symptoms leads to higher rates of respiratory and other complications, and the risk seems to increase with ODI (24). Mokhlesi et al. showed how patients with SDB had higher rates of emergency intubation, acute CPAP/NIV use, atrial fibrillation, and respiratory failure following bariatric surgery; data on the post-operative CPAP use was not available (16). Another prospective study found that OSA increased the risk of a composite endpoint of death, venous thromboembolism, re-intervention, and failure to be discharged at 30 days following bariatric surgery, despite routine post-operative use of CPAP in all patients with OSA (25). Two meta-analyses concluded that respiratory and cardiac complications, as well as ICU transfers, were more frequent in patients with OSA (10,11). LOS and infection rates also seem to be more frequent in patients with OSA (22). In the current study, higher risk ratios for use of oxygen and LOS in patients with significant OSA may also be confounded by the higher BMI and increased level of comorbidities in general, not attributable directly to the OSA.
The use of CPAP might mitigate against these deleterious effects. Weingarten et al. found no association between the diagnosis of OSA and post-operative complications following bariatric surgery, where 93% of OSA patients used post-operative CPAP therapy and were closely monitored (14). In a different study, the need for ICU-level care did not differ between those with and without OSA where patients with OSA were given CPAP if they desaturated post-operatively (15). A previous analysis of a smaller dataset also showed no increased risk of respiratory complications in patients with significant OSA compared to those without, with the authors concluding that the use of CPAP contributed to this outcome (26). In the current analysis, there was no difference in post-operative complications between groups and the pragmatic approach to offer CPAP to patients with moderate and severe OSA, or symptomatic milder OSA, seems to provide safe peri-operative care. Appropriate identification of OSA by screening all surgical candidates, including using nocturnal oximetry alone without a full polysomnogram (27), and institution of CPAP in those with OSA, is therefore likely to improve outcomes for these patients in the post-operative period (28). Using CPAP after bariatric surgery is safe and does not lead to higher rates of surgery-related complications, such as anastomotic leaks (29). Weight loss was highest in the CPAP group, and this was an unexpected finding which differs from that of a recent study (30), in which the OSA group also had a higher BMI but lost less weight. It is likely that this observation was due to the different weight at baseline and not due to the use of CPAP as patients with OSA do not typically experience a beneficial change in weight (31), they more likely experience a weight gain (32), in patients who do not undergo bariatric surgery.
Finally, patients living with obesity and OSA represent a complex cohort who, as observed in our dataset, frequently have multiple other co-morbidities that could impact on clinical outcomes following surgery. Whether there is a causal relationship between OSA and clinical outcomes in the peri-operative setting, or whether other comorbidities confound the results, either individually or in conjunction with each other, is not entirely clear from the current evidence. However, CPAP would be expected to have the most meaningful impact on respiratory and cardiovascular complications (22). In our analysis, the use of post-operative CPAP in patients with OSA might have led to an assimilation in clinical outcomes between the two cohorts and helped to minimise risks.
Limitations
Our study was limited by a number of factors. There were data missing for complications and level of respiratory support. Data for complications were available for 89 (88.1%) patients and 190 (94.5%), on respiratory support were available in 33 (32.7%) and 76 (37.8%) patients, and on wards managed in 88 (87.1%) and 188 (93.5%), in the CPAP and non-CPAP groups, respectively. Respiratory support data were only used if there was documented evidence for each day of the inpatient stay. Several patients were operated in the independent sector or were operated before full electronic documentation was implemented at our institution. For these there was only access to discharge summaries noting whether there had been any complications, with no daily information on respiratory support. We included this data as evidence for complications but could not conclude the level of respiratory support required. However, it would be reasonable to assume that were patients to have been intubated, developed an acute need for NIV, or required high levels of oxygen, this would have been documented in a discharge summary. None did, however, so it is likely that none needed this level of support. Pre- and post-operative data on CPAP compliance were limited, and the overall number of complications was small; however, our sample size approximates to those in other studies (10) and, as a result, our findings should be considered with caution but reflect valid findings in clinical services. The limited differences observed in the complications according to the Clavien-Dindo grades may also be due to the low sample size, as, when grouped together, there was a more marked effect. Differences in respiratory outcomes might have been attributable to CPAP use, although we cannot infer a causal relationship. Better compliance by some patients in the CPAP group might have further contributed to more beneficial outcomes compared to others with poor compliance. OSA was diagnosed using nocturnal oximetry and not the gold-standard polysomnography which is likely to have underdiagnosed the prevalence of OSA in our cohort. Nonetheless, this is normal practice at our institution and in the wider NHS and other healthcare systems and has been shown to be effective (27) and supported in NICE (33) and other consensus guidelines (9). Our data only refers to bariatric surgery and therefore cannot be generalizable to other types of surgery.
Conclusions
Significant OSA is common in morbidly obese patients. Complications following bariatric surgery are relatively rare but require close monitoring, particularly in the at-risk cohort of patients with higher weight and increased level of comorbidities. Peri-operative CPAP therapy might minimise associated risks in their management. Further prospective studies would be useful to test the best strategic use of resources in the management of morbidly obese patients with OSA perioperatively.
Acknowledgments
Professor Steier’s contributions were partially supported by the National Institute for Health Research (NIHR) Biomedical Research Centre based at Guy’s and St Thomas’ NHS Foundation Trust and King’s College London, UK. The views expressed are those of the authors and not necessarily those of the NHS, the NIHR or the Department of Health.
Funding: None.
Footnote
Provenance and Peer Review: This article was commissioned by the editorial office, Journal of Thoracic Disease for the series “Clinical Update Sleep 2023”. The article has undergone external peer review.
Reporting Checklist: The authors have completed the STROBE reporting checklist. Available at https://jtd.amegroups.com/article/view/10.21037/jtd-22-1501/rc
Data Sharing Statement: Available at https://jtd.amegroups.com/article/view/10.21037/jtd-22-1501/dss
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://jtd.amegroups.com/article/view/10.21037/jtd-22-1501/coif). The series “Clinical Update Sleep 2023” was commissioned by the editorial office without any funding or sponsorship. JS serves as the unpaid Guest Editor of the series and an unpaid editorial board member of Journal of Thoracic Disease. The authors have no other conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- World Health Organization. Obesity and Overweight [Internet]. Geneva: World Health Organization; 2021 June 9 [cited 19 December 2021]. Obesity and Overweight. Available online: https://www.who.int/news-room/fact-sheets/detail/obesity-and-overweight
- NHS Digital. Statistics on Obesity, Physical Activity and Diet, England 2021 [Internet]. Leeds: NHS Digital; 2020 (published 18 May 2021, accessed 16 March 2022). Available online: https://digital.nhs.uk/data-and-information/publications/statistical/statistics-on-obesity-physical-activity-and-diet/england-2021/appendices
- Reed K, Pengo MF, Steier J. Screening for sleep-disordered breathing in a bariatric population. J Thorac Dis 2016;8:268-75. [Crossref] [PubMed]
- Dacal Quintas R, Tumbeiro Novoa M, Alves Pérez MT, et al. Obstructive sleep apnea in normal weight patients: characteristics and comparison with overweight and obese patients. Arch Bronconeumol 2013;49:513-7. [Crossref] [PubMed]
- Peromaa-Haavisto P, Tuomilehto H, Kössi J, et al. Obstructive sleep apnea: the effect of bariatric surgery after 12 months. A prospective multicenter trial. Sleep Med 2017;35:85-90. [Crossref] [PubMed]
- Veasey SC, Rosen IM. Obstructive Sleep Apnea in Adults. N Engl J Med. 2019;380:1442-9. [Crossref] [PubMed]
- Wolfe BM, Kvach E, Eckel RH. Treatment of Obesity: Weight Loss and Bariatric Surgery. Circ Res 2016;118:1844-55. [Crossref] [PubMed]
- National Institute for Health and Care Excellence. Obesity: identification, assessment and management [Internet]. (London): NICE; 2014 (published 27 November 2014, accessed 24 February 2022). Clinical guideline [CG189]. Available online: https://www.nice.org.uk/guidance/cg189
- de Raaff CAL, Gorter-Stam MAW, de Vries N, et al. Perioperative management of obstructive sleep apnea in bariatric surgery: a consensus guideline. Surg Obes Relat Dis 2017;13:1095-109. [Crossref] [PubMed]
- Kaw R, Chung F, Pasupuleti V, et al. Meta-analysis of the association between obstructive sleep apnoea and postoperative outcome. Br J Anaesth 2012;109:897-906. [Crossref] [PubMed]
- Hai F, Porhomayon J, Vermont L, et al. Postoperative complications in patients with obstructive sleep apnea: a meta-analysis. J Clin Anesth 2014;26:591-600. [Crossref] [PubMed]
- Kaw R, Pasupuleti V, Walker E, et al. Postoperative complications in patients with obstructive sleep apnea. Chest 2012;141:436-41. [Crossref] [PubMed]
- Chung F, Nagappa M, Singh M, et al. CPAP in the Perioperative Setting: Evidence of Support. Chest 2016;149:586-97. [Crossref] [PubMed]
- Weingarten TN, Flores AS, McKenzie JA, et al. Obstructive sleep apnoea and perioperative complications in bariatric patients. Br J Anaesth 2011;106:131-9. [Crossref] [PubMed]
- Grover BT, Priem DM, Mathiason MA, et al. Intensive care unit stay not required for patients with obstructive sleep apnea after laparoscopic Roux-en-Y gastric bypass. Surg Obes Relat Dis 2010;6:165-70. [Crossref] [PubMed]
- Mokhlesi B, Hovda MD, Vekhter B, et al. Sleep-disordered breathing and postoperative outcomes after bariatric surgery: analysis of the nationwide inpatient sample. Obes Surg 2013;23:1842-51. [Crossref] [PubMed]
- Rosenberg J, Rasmussen GI, Wøjdemann KR, et al. Ventilatory pattern and associated episodic hypoxaemia in the late postoperative period in the general surgical ward. Anaesthesia 1999;54:323-8. [Crossref] [PubMed]
- Chung F, Liao P, Elsaid H, et al. Factors associated with postoperative exacerbation of sleep-disordered breathing. Anesthesiology 2014;120:299-311. [Crossref] [PubMed]
- Stierer TL, Wright C, George A, et al. Risk assessment of obstructive sleep apnea in a population of patients undergoing ambulatory surgery. J Clin Sleep Med 2010;6:467-72.
- Kheterpal S, Healy D, Aziz MF, et al. Incidence, predictors, and outcome of difficult mask ventilation combined with difficult laryngoscopy: a report from the multicenter perioperative outcomes group. Anesthesiology 2013;119:1360-9. [Crossref] [PubMed]
- Memtsoudis SG, Cozowicz C, Nagappa M, et al. Society of Anesthesia and Sleep Medicine Guideline on Intraoperative Management of Adult Patients With Obstructive Sleep Apnea. Anesth Analg 2018;127:967-87. [Crossref] [PubMed]
- Vasu TS, Grewal R, Doghramji K. Obstructive sleep apnea syndrome and perioperative complications: a systematic review of the literature. J Clin Sleep Med 2012;8:199-207. [Crossref] [PubMed]
- Adesanya AO, Lee W, Greilich NB, et al. Perioperative management of obstructive sleep apnea. Chest 2010;138:1489-98. [Crossref] [PubMed]
- Hwang D, Shakir N, Limann B, et al. Association of sleep-disordered breathing with postoperative complications. Chest 2008;133:1128-34. [Crossref] [PubMed]
- Longitudinal Assessment of Bariatric Surgery (LABS) Consortium. Perioperative safety in the longitudinal assessment of bariatric surgery. N Engl J Med 2009;361:445-54. [Crossref] [PubMed]
- Meurgey JH, Brown R, Woroszyl-Chrusciel A, et al. Peri-operative treatment of sleep-disordered breathing and outcomes in bariatric patients. J Thorac Dis 2018;10:S144-52. [Crossref] [PubMed]
- Chung F, Liao P, Elsaid H, et al. Oxygen desaturation index from nocturnal oximetry: a sensitive and specific tool to detect sleep-disordered breathing in surgical patients. Anesth Analg 2012;114:993-1000. [Crossref] [PubMed]
- Verbraecken J, Hedner J, Penzel T. Pre-operative screening for obstructive sleep apnoea. Eur Respir Rev 2017;26:160012. [Crossref] [PubMed]
- Weingarten TN, Kendrick ML, Swain JM, et al. Effects of CPAP on gastric pouch pressure after bariatric surgery. Obes Surg 2011;21:1900-5. [Crossref] [PubMed]
- Guggino J, Tamisier R, Betry C, et al. Bariatric surgery short-term outcomes in patients with obstructive sleep apnoea: the Severe Obesity Outcome Network prospective cohort. Int J Obes (Lond) 2021;45:2388-95. [Crossref] [PubMed]
- Ou Q, Chen B, Loffler KA, et al. The Effects of Long-term CPAP on Weight Change in Patients With Comorbid OSA and Cardiovascular Disease: Data From the SAVE Trial. Chest 2019;155:720-9. [Crossref] [PubMed]
- Quan SF, Budhiraja R, Clarke DP, et al. Impact of treatment with continuous positive airway pressure (CPAP) on weight in obstructive sleep apnea. J Clin Sleep Med 2013;9:989-93. [Crossref] [PubMed]
- National Institute for Health and Care Excellence. Obstructive sleep apnoea/hypopnoea syndrome and obesity hypoventilation syndrome in over 16s [Internet]. (London): NICE; 2021 (published 20 August 2021, accessed 24 February 2022). NICE Guideline [NG202]. Available online: https://www.nice.org.uk/guidance/ng202/chapter/1-Obstructive-sleep-apnoeahypopnoea-syndrome#diagnostic-tests-for-osahs