
Pathogenesis and management of gastroesophageal reflux disease-associated cough: a narrative review
Introduction
Chronic cough is a common extraesophageal symptom of gastroesophageal reflux disease (GERD) and occurs in about one-third of patients with GERD (1). GERD-associated cough, also known as reflux cough syndrome, is considered to be a special type of GERD, which is characterized by a predominance of cough (2,3). It accounts for 10–40% of chronic cough and has been increasingly recognized by physicians and researchers (1). The international guidelines for the diagnosis and treatment of cough have included the management of GERD-associated cough as an important component (4,5).
Although GERD-associated cough is a common cause of chronic cough worldwide, it was considered to be relatively rare in China. In 2013, a multi-center survey of the etiologies of chronic cough reported that GERD-associated cough only accounted for less than 5% of chronic cough in China (6). In our specialist cough clinic, GERD-associated cough was found in 9% of patients with chronic cough from 2004 to 2008 (7), which increased to 33% from 2009 to 2016 (8). The increasing prevalence of GERD-associated cough may reflect the changes in lifestyle and obesity rates due to social and economic development in China (8). This is also complicated by regional differences in the etiological distribution of chronic cough, the diagnostic thinking of physicians and the available diagnostic techniques in medical institutions.
This review summarizes our current understanding on the pathogenesis and management of GERD cough. We present the following article in accordance with the Narrative Review reporting checklist (available at https://jtd.amegroups.com/article/view/10.21037/jtd-22-1757/rc).
Methods
The main literatures regarding the pathogenesis and management of GERD-associated cough were searched in PubMed and reviewed comprehensively as detailed in Table 1. We derived our understanding and insights on these issues from the published data, especially our studies conducted between 2009 and 2023.
Table 1
| Items | Specification |
|---|---|
| Date of search | September 25th, 2022–January 5th, 2023 |
| Databases and other sources searched | PubMed |
| Search terms used | “Chronic cough”, “Gastroesophageal reflux”, “Cough due to reflux” |
| Timeframe | 1988–2023 |
| Inclusion and exclusion criteria | The main literature review about the pathogenesis and management of GERD-associated cough were included; case reports and similar review articles were excluded |
| Selection process | Li Zhang and Alimire Aierken searched and reviewed all initial articles, and conferred with Mengru Zhang, with additional selection by Zhongmin Qiu. Ultimate final article inclusion was determined by all authors |
GERD, gastroesophageal reflux disease.
Pathogenesis of GERD-associated cough
The mechanisms underlying GERD-associated cough mainly include reflux theory and reflex theory (2). Historically, reflux theory proposes that the acid refluxes (gastric acid and pepsin) rising to proximal esophagus directly injure laryngopharyngeal mucosa and are microaspirated into the lower airways, which irritate the innervating cough receptors and arise cough. Reflex theory suggests that the irritants derived from reflux in distal esophagus may also trigger cough indirectly through esophageal-tracheobronchial reflex. It is generally believed that reflex theory is more important than reflux theory in the pathogenesis of GERD-associated cough since only 32–37% of patients with GERD were observed having reflux reaching the upper esophagus (9,10), and the distal esophagus is the mandatory route for all refluxes traveling up to the proximal end of esophagus as well. Therefore, reflux theory actually contains the components of reflex theory. In addition, the disorder of esophageal dysmotility, which is also common in patients with GERD-associated cough (11), can amplify the noxious response of abnormal reflux by delaying esophageal clearance and prolonging acid exposure (Figure 1). Why only some patients develop chronic cough in the presence of GERD remains to be elucidated. One possible explanation is that patients with cough and abnormal reflux are highly responsive to the effects of both acid and non-acid reflux. Cough hypersensitivity underlied by airway neurogenic inflammation is responsible for the exaggerated cough reactivity to reflux, which is non-tussive in physical state but is violently tussive in patients with GERD-associated cough (12). Currently, how GERD-associated cough initiates remains unclear.
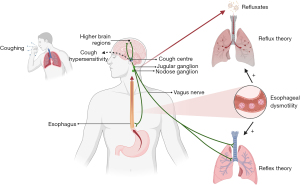
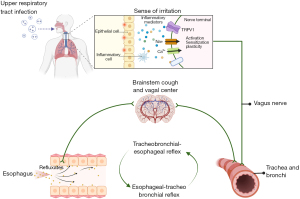
Both the tracheobronchial tree and the esophagus originate from the embryonic foregut and are innervated by the vagal C-fibers, which express the transient receptor potential vanilloid 1 (TRPV1) (13). The stimuli sensed and transmitted by the nerve endings innervating both esophagus and airway converge in the primary cough center and may have a neural crosstalk. This may produce the initiation of the cough reflex in response to the reflux-mediated irritation (14). In an intact vagal network, persistent excitation of one peripheral end may prime another distant peripheral end by sensitizing the neural circuit. There has been growing evidence to support that the esophageal-tracheobronchial reflex is mediated by the vagal C-fibers innervating the tracheobronchial and esophageal mucosa (15). Since GERD-associated cough is often provoked by a previous upper respiratory tract infection (16), it is conceivable that nociceptive irritants in the airway caused by the infection might enhance the excitability of C-fibers in the esophageal mucosa and elicit the esophageal hypersensitivity via the tracheobronchial-esophageal reflex (neuronal pathways that link the airway to the esophagus) as a mirror of esophageal-tracheobronchial reflex (neuronal pathways that link the esophagus to the airway) (Figure 2). Our study demonstrated tracheobronchial-esophageal reflex did exist and was mediated by TRPV1 signaling in the guinea pigs with repeated capsaicin inhalation (17). The tracheobronchial-esophageal reflex might provide a plausible explanation for how airway diseases, especially upper respiratory tract infection, initiated the reflux-related cough, thus producing a vicious cough-reflux loop (17).
Diagnosis of GERD-associated cough
Symptom features
Clinical features of patients with GERD-associated cough include diet-related cough (triggered or aggravated by meal), with or without typical reflux-associated symptoms such as regurgitation and heartburn. Moreover, hoarseness, postnasal drip and throat clearing may be a manifestation of reflux laryngitis. Generally, regurgitation and heartburn are present only in less than 60% of patients with GERD-associated cough and are more frequent in acid reflux-induced cough (18). Therefore, the absence of classic reflux-associated symptoms is not rare, and it is impossible to rely solely on these symptoms to diagnose GERD-associated cough.
Reflux monitoring
Abnormal reflux and its association with cough are important objective evidence for the diagnosis of GERD-associated cough. Currently, 24-hour ambulatory esophageal pH monitoring and impedance-pH monitoring are recommended as first choice to measure reflux in China (19). When these investigations are unavailable, upper endoscopy and barium esophagography can be alternative options. However, although these two methods have a relatively high specificity in detecting GERD-associated cough, the sensitivity remains quite poor (20).
High resolution manometry is worthy of attention and is now the most useful of the available esophageal manometry techniques due to its ability to describe the anatomy of the esophagus and accurately assesses esophageal peristalsis (3). European Respiratory Society (ERS) guideline for cough management has first advocated its application to the evaluation of GERD-associated cough (5). With this technology, esophageal dysmotility was found in two thirds of patients with chronic cough (11).
Esophageal impedance-pH monitoring is superior to 24-hour ambulatory esophageal pH monitoring. It has the ability to identify both acid and non-acid reflux, provide information on the temporal link between reflux and cough, accurately distinguish reflux from swallowing, precisely locate the height of the reflux reaching the esophagus (proximal and distal reflux) and determine the nature of the reflux (liquid, gas and liquid-gas mixture) (21). Furthermore, impedance-pH monitoring can be performed on or off the use of proton pump inhibitors, and in combination with high resolution manometry to assess the esophageal dysmotility. At present, it has been recommended as the gold standard for reflux detection by the Lyon Consensus (3). Since there is no general agreement on how to interpret the test of esophageal reflux monitoring in patients with chronic cough, esophageal impedance-pH monitoring is not accepted as a mandatory laboratory investigation for the diagnosis of GERD-associated cough in the international guidelines for the cough management (4,5).
Reflux diagnostic criteria
Diagnostic criteria for reflux usually utilize acid exposure time (AET), symptom associated probability (SAP) and total reflux episodes (3). These impedance-pH variables have individual inherent clinical values in establishing a causal relationship between cough and reflux. AET is defined as total percentage of time that the esophagus is exposed to pH <4.0 throughout monitoring time, with a value of <4% being definitively normal (physiological) and >6% being absolutely abnormal, and the intermediate value between these two limits being indeterminate. SAP reflects the likelihood of the reflux being the cause of coughing, and a value of ≥95% is considered positive. Total reflux episodes represent the total number of refluxes over 24 hours, with a number of >80 times being definitively abnormal, <40 being physiological and the intermediate values being indeterminate (3). In the early years, DeMeester score, a composite algorithmic score of six parameters measuring acid reflux, was commonly used to identify potential pathological esophageal acid exposure and predict response to PPI therapy (22), but now is recommended to be replaced by AET due to its poor repeatability (3,23).
According to the latest Chinese cough guidelines (updated in 2021), the diagnostic criteria for GERD-associated cough include (I) chronic cough, with or without concomitant classic reflux-association symptoms such as regurgitation and heartburn; (II) abnormal acid or non-acid reflux detected by esophageal reflux monitoring, as evidenced by AET >6% or SAP ≥95%; and (III) complete relief or marked improvement of cough in response to anti-reflux treatment (19). It is the only cough guideline worldwide that attempts to set up the clear reflux criteria for the diagnosis of GERD-associated cough. However, the application of the diagnostic criterion is only limited to a few cough research centers across China in real-world clinical practice because esophageal impedance-pH monitoring is expensive and time-consuming, In our cough clinic, approximately 70% of patients with suspected GERD-related cough have undergone esophageal impedance-pH monitoring for the establishment of the diagnosis (8,21), which is much higher than in the other clinics in China and in the other countries where the empirical anti-reflux therapy is used as the preferred mainstream diagnostic approach.
Our previous study has demonstrated that AET has a high sensitivity but low specificity for the prediction of GERD-associated cough, while SAP has a low sensitivity but high specificity (21). AET and SAP have limited diagnostic efficacy when used alone, however, this can be improved greatly by combined use. Overall, the diagnostic value of AET and SAP remains suboptimal (21). Although AET and DeMeester present a comparable diagnostic efficacy and AET is able to replace DeMeester score, AET is not superior to DeMeester score in identifying GERD-associated cough (24). Compared to AET >6%, AET >4.8% appears to be a more optimal cut-off point to predict GERD-associated cough with a higher sensitivity and roughly equivalent specificity (24). Therefore, 6% may be a too stringent cut-off point for diagnosing GERD-associated cough. An optimal pathological AET needs to be sought to further improve its diagnostic accuracy.
There are two methods of imputation for SAP: the first is to count all the recorded reflux-related symptoms which include cough; and the second is to count only the cough symptom. While the former is widely used in the diagnosis of GERD, the latter has only been used for determining the relationship between reflux and chronic cough in several studies (25,26), but has not been applied in clinical practice to identify GERD-associated cough. Our recent study on 105 patients showed that SAP calculated only by cough was superior to SAP involving all the reflux-related symptoms, with the former demonstrating a higher sensitivity and diagnostic accuracy for both acid reflux and non-acid reflux induced cough (27). In contrast, SAP is inferior to AET in terms of diagnostic efficacy, but is particularly suitable for predicting GERD-associated cough with obvious non-acid reflux or without abnormal acid reflux.
Our previous study also demonstrated a limited predictive value of the total reflux episodes in GERD-associated cough, with the cut-off point >97 times (28). Mean nocturnal baseline impedance and post-reflux swallow-induced peristaltic wave index have demonstrated the ability to enhance the diagnostic yield of impedance-pH monitoring in GERD-associated cough as well (29).
The summary of reflux diagnostic criteria for GERD-associated cough can be seen in Table 2.
Table 2
| Diagnostic indicator | Cut-off value | AUC | Sensitivity (%) | Specificity (%) | References |
|---|---|---|---|---|---|
| AET | 6% | 0.613–0.768 | 42.5–63.2 | 63.6–93.0 | (21,24,29) |
| 4.8% | 0.827 | 73.3 | 88.4 | (24) | |
| SAPa | 95% | 0.561–0.714 | 22.1–25.7 | 87.8–100 | (21,24) |
| SAPb | 95% | 0.757 | 53.9 | 97.5 | (27) |
| Total reflux episodes | 97 | 0.684 | 46.6 | 83.1 | (28) |
| DeMeester score | 14.7 | 0.818 | 70.0 | 89.6 | (24) |
| AET and SAP | 6%, 95% | 0.863 | 92.6 | 63.6 | (21) |
| DeMeester score and AET | 14.7, 4.8 | 0.809 | 72.7 | 75.3 | (24) |
| MNBI | 2,292 Ω | 0.748 | 65.0 | 75.5 | (29) |
| PSWI index | 61% | 0.732 | 79.3 | 87.5 | (29) |
a, SAP involving all the reflux-related symptoms; b, SAP calculated only by cough. GERD, gastroesophageal reflux disease; AUC, area under the curve; AET, acid exposure time; SAP, symptom associated probability; MNBI, mean nocturnal baseline impedance; PSWI, post-reflux swallow-induced peristaltic wave.
Treatment of GERD-associated cough
The aim of treatment for GERD-associated cough is to relieve cough symptoms and improve the patients` quality of life by minimizing reflux episodes and eliminating the factors precipitating reflux (2). Relief of cough that responds to anti-reflux therapy is a key outcome to definitely confirm the diagnosis of GERD-associated cough (Table 3).
Table 3
| Treatment | Patient selection | Representatives | Outcomes | Side effects |
|---|---|---|---|---|
| Lifestyle modification | Overweight, unhealthy eating and lifestyle habits | Healthy eating and living, head elevation (30,31) | Unclear | N/A |
| Medicinal treatment | ||||
| PPI | Typical reflux-related symptoms or abnormal acid reflux | Omeprazole, rabeprazole, esomeprazole | Controversial; benefits 10–70% of cough patients (32,33) | No available data |
| Prokinetics | Esophageal dysmotility and impaired gastric emptying | Metoclopramide, domperidone and mosapride | Lack of sufficient evidence for cough patients; when added to PPI, may be effective (34) | No available data |
| Double dose of PPI | Refractory GERD-associated cough, severe esophageal acid exposure | Omeprazole, rabeprazole, esomeprazole | Cough controlled in 20–40% of patients (35) | No available data |
| Neuromodulators | Refractory GERD-associated cough | Baclofen, gabapentin | 50–70% of success rates for cough patients (36-38) | Central nervous system-related side effects (somnolence, dizziness, fatigue) |
| Anti-reflux surgery | Refractory GERD-associated cough | laparoscopic fundoplication | Cough resolved in 85% of patients (39) | Complications (dysphagia, dyspepsia, reflux esophagitis) |
GERD, gastroesophageal reflux disease; PPI, proton pump inhibitor; N/A, not applicable.
Lifestyle modification
Lifestyle modifications are essential for the long-term management of GERD-associated cough, which include weight loss for patients who are overweight or have recently gained weight, quitting smoking and alcohol, and avoiding foods or drugs which may trigger or aggravate reflux (30). Head elevation during sleep may also reduce reflux (31), but whether it can improve GERD-associated cough remains to be proven.
Anti-reflux medicinal treatment
Several studies have confirmed that acid suppressive therapy can significantly reduce the damage and irritation of the esophagus and airway mucosa from gastric acid and pepsin in the refluxates, and alleviate cough by inhibiting micro-aspiration and esophageal-tracheobronchial reflex (2,4). Proton pump inhibitors are the first choice since they potently block the secretion of gastric acid and provide rapid relief of reflux-associated symptoms, such as cough (30). However, the antitussive efficacy of proton pump inhibitors on GERD-associated cough is inconsistent between clinical observations and randomized clinical trials (40,41). Meta-analysis also showed that proton pump inhibitors have not been universally beneficial for GERD-associated cough (32) and only work in some patients with pathologic esophageal acid exposure (33). Therefore, the overall benefits of proton pump inhibitors have been controversial (34). The latest American College of Chest Physicians (ACCP) and ERS guidelines for cough management did not recommend the universal use of proton pump inhibitors to treat GERD-associated cough. Proton pump inhibitors have been considered only suitable for patients with typical reflux-related symptoms (e.g., regurgitation and heartburn) or with the subjective evidence of abnormal acid reflux. However, their therapeutic benefits may also be limited (4,5). In contrast, the updated Chinese, Japanese and Korean guidelines for the management of cough advocated acid suppression therapy as the first-line treatment for GERD-associated cough (19,42,43). Since all the guidelines for GERD have recommended anti-acid therapy as the primary treatment (30), it is difficult to understand why proton pump inhibitors are ineffective for GERD-associated cough (34). Even for cough due to weakly acidic reflux, a slight increase in the pH value of refluxates in response to a trial of proton pump inhibitors may significantly reduce the acid signaling in the hypersensitive esophagus and relieve cough (12). Ideally, the first-best option for GERD-associated cough is to minimize the excessive reflux burden by rebuilding normal anti-reflux barrier. Therefore, acid suppressive therapy is currently the second-best option because the structural and functional abnormalities of the lower esophageal sphincter and esophagogastric junction do not get treated (34). The failed response to proton pump inhibitors should not rule out the possibility that patient’s cough is triggered by reflux.
There is no clear consensus regarding the course of acid suppressive therapy, with little evidence on the optimal duration of treatment for GERD-associated cough. Generally, a trial of proton pump inhibitor for 8–12 weeks is recommended to define extraesophageal GERD symptoms (30). In our practice, we usually shorten the duration of treatment to 4–8 weeks for suspected GERD-associated cough. Since a variety of conditions other than GERD can also produce a cough, a prolonged anti-reflux trial may delay the diagnosis and treatment of the true cause (18,21). If the initial trial could provide cough resolution, a further 3–6 months’ supply can be given. The treatment can be maintained indefinitely or used as needed afterwards, depending on the relapse and persistence of cough symptoms (30).
Prokinetics should be a reasonable option of anti-reflux medicinal therapy since they increase the pressure of lower esophageal sphincter and promote gastric emptying. However, there is no evidence currently to show that metoclopramide, domperidone and mosapride can alleviate GERD-associated cough. Prokinetics are usually used in combination with antacids, and are especially indicated in patients with esophageal dysmotility and impaired gastric emptying (5,34).
For the refractory GERD-associated cough, intensified anti-reflux medicinal therapy such as the double dose of proton pump inhibitors or neuromodulators can be tried (35). Besides the adjustments in dosage and brand, the optimization of proton pump inhibitor therapy also includes ensuring good compliance and correct usage, such as taking the drugs 30–60 minutes before meals, switching to a different proton pump inhibitor and co-administration with a histamine H2 receptor antagonist (35). This proved effective in the management of refractory GERD-associated cough (44). The other antiacids, such as novel potassium-competitive acid blockers and alginates, have the potential to achieve effective and durable acid suppression as well, thus addressing the specific therapeutic needs of GERD patients (30). Although there is a wealth of available data on the intractable GERD, studies on refractory GERD-associated cough are absolutely lacking. Baclofen and gabapentin are the neuromodulators commonly off-label used for cough, but they do not work perfectly (45). Theoretically, baclofen should be superior to gabapentin since it inhibits the transient relaxation of the lower esophageal sphincter and blocks both acid and non-acid reflux in addition to having a comparable antitussive effect to gabapentin (45). However, our previous study showed that as the add-on therapy of omeprazole, baclofen and gabapentin achieved a similar therapeutic success in 53–58% of patients with refractory GERD-associated cough, but gabapentin had fewer central nervous system-related side effects and was more tolerable than baclofen (36). Given the failure of the treatment with baclofen or gabapentin in approximately 40% of patients with refractory GERD-associated cough and the limitations of frequent central nervous system-related side effects, it is of great clinical value to find a way to screen patients most likely to respond to baclofen and gabapentin to improve the success rates to avoid drug abuse or minimize potential adverse effects. Our previous studies have demonstrated that the pressure (less than 11 mmHg) and length (less than 2.35 cm) of lower esophageal sphincter measured by esophageal manometry are useful indicators to predict the therapeutic success of baclofen treatment, while Hull airway reflux questionnaire score (more than 21.5) is a promising tool to predict a more favorable response to gabapentin treatment in patients with refractory GERD-associated cough (37,38).
Anti-reflux surgery
Anti-reflux surgery removes abnormal gastroesophageal reflux by artificially rebuilding the mechanical barrier at the gastro-esophageal junction, and may be a promising approach for refractory GERD-associated cough (4). As a commonly used anti-reflux operation, laparoscopic fundoplication can resolve coughing in 85% of patients who undergo this treatment (39). Given its invasive nature and the fact that efficacy cannot be guaranteed if GERD-associated cough is not definitively established, strict surgical indications are required. The following criteria may be useful in patient selection: patients who have (I) a clear diagnosis of GERD-associated cough and responded well to anti-reflux agents, but do not wish to be on long-term medications and would like the anti-reflux surgery; (II) failed anti-reflux trials, but have objective evidence of abnormal reflux which is associated with cough, as indicated by various laboratory investigations such as esophageal impedance-pH monitoring; (III) no peristaltic dysfunction of the esophagus (4,19).
Conclusions
Tracheobronchial-esophageal reflex might initiate reflux-induced cough provoked by the upper respiratory tract infection through a central crosstalk between the neural pathways linking the airway and esophagus in a manner similar but opposite to esophageal-tracheobronchial reflex. Esophageal reflux monitoring provides the main diagnostic criteria for GERD-associated cough. AET and SAP have demonstrated significant diagnostic efficiency as reflux diagnostic parameters, but they are not yet perfect and far from being the gold standard. It is necessary to optimize the current standards and explore new criteria with higher diagnostic potency. Acid suppressive therapy is the first choice for GERD-associated cough but needs further evaluation. Neuromodulators may be potential therapeutic options for refractory GERD-associated cough, for which anti-reflux surgery may be also a promising treatment option.
Acknowledgments
Funding: This study was supported in part by the Project of Science and Technology Commission of Shanghai Municipality (Nos. 20Y11902500 and 21140903400) and Tongji Hospital [Nos. ITJ(ZD)2006 and TJ1801].
Footnote
Provenance and Peer Review: This article was commissioned by the Guest Editors (Woo-Jung Song and Kian Fan Chung) for the series “Novel Insights into Chronic Cough” published in Journal of Thoracic Disease. The article has undergone external peer review.
Reporting Checklist: The authors have completed the Narrative Review reporting checklist. Available at https://jtd.amegroups.com/article/view/10.21037/jtd-22-1757/rc
Peer Review File: Available at https://jtd.amegroups.com/article/view/10.21037/jtd-22-1757/prf
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://jtd.amegroups.com/article/view/10.21037/jtd-22-1757/coif). The series “Novel Insights Into Chronic Cough” was commissioned by the editorial office without any funding or sponsorship. The authors have no other conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Jaspersen D, Kulig M, Labenz J, et al. Prevalence of extra-oesophageal manifestations in gastro-oesophageal reflux disease: an analysis based on the ProGERD Study. Aliment Pharmacol Ther 2003;17:1515-20. [Crossref] [PubMed]
- Irwin RS. Chronic cough due to gastroesophageal reflux disease: ACCP evidence-based clinical practice guidelines. Chest 2006;129:80S-94S. [Crossref] [PubMed]
- Gyawali CP, Kahrilas PJ, Savarino E, et al. Modern diagnosis of GERD: the Lyon Consensus. Gut 2018;67:1351-62. [Crossref] [PubMed]
- Kahrilas PJ, Altman KW, Chang AB, et al. Chronic Cough Due to Gastroesophageal Reflux in Adults: CHEST Guideline and Expert Panel Report. Chest 2016;150:1341-60. [Crossref] [PubMed]
- Morice AH, Millqvist E, Bieksiene K, et al. ERS guidelines on the diagnosis and treatment of chronic cough in adults and children. Eur Respir J 2020;55:1901136. [Crossref] [PubMed]
- Lai K, Chen R, Lin J, et al. A prospective, multicenter survey on causes of chronic cough in China. Chest 2013;143:613-20. [Crossref] [PubMed]
- Yu L, Wei WL, Lü HJ, et al. Changes in the spectrum and frequency of causes for chronic cough: a retrospective analysis. Zhonghua Jie He He Hu Xi Za Zhi 2009;32:414-7. [PubMed]
- Ding H, Xu X, Wen S, et al. Changing etiological frequency of chronic cough in a tertiary hospital in Shanghai, China. J Thorac Dis 2019;11:3482-9. [Crossref] [PubMed]
- Lee JH, Park SY, Cho SB, et al. Reflux episode reaching the proximal esophagus are associated with chronic cough. Gut Liver 2012;6:197-202. [Crossref] [PubMed]
- Patterson N, Mainie I, Rafferty G, et al. Nonacid reflux episodes reaching the pharynx are important factors associated with cough. J Clin Gastroenterol 2009;43:414-9. [Crossref] [PubMed]
- Sykes DL, Crooks MG, Hart SP, et al. Investigating the diagnostic utility of high-resolution oesophageal manometry in patients with refractory respiratory symptoms. Respir Med 2022;202:106985. [Crossref] [PubMed]
- Qiu Z, Yu L, Xu S, et al. Cough reflex sensitivity and airway inflammation in patients with chronic cough due to non-acid gastro-oesophageal reflux. Respirology 2011;16:645-52. [Crossref] [PubMed]
- Cortright DN, Szallasi A. Biochemical pharmacology of the vanilloid receptor TRPV1. An update. Eur J Biochem 2004;271:1814-9. [Crossref] [PubMed]
- Canning BJ. Central regulation of the cough reflex: therapeutic implications. Pulm Pharmacol Ther 2009;22:75-81. [Crossref] [PubMed]
- Mazzone SB, Mori N, Canning BJ. Synergistic interactions between airway afferent nerve subtypes regulating the cough reflex in guinea-pigs. J Physiol 2005;569:559-73. [Crossref] [PubMed]
- Lai K, Zhan W, Li H, et al. The Predicative Clinical Features Associated with Chronic Cough That Has a Single Underlying Cause. J Allergy Clin Immunol Pract 2021;9:426-432.e2. [Crossref] [PubMed]
- Chen Q, Dong R, Zhang L, et al. Tracheobronchial-esophageal reflex initiates esophageal hypersensitivity and aggravates cough hyperreactivity in guinea pigs with esophageal acid infusion. Respir Physiol Neurobiol 2022;301:103890. [Crossref] [PubMed]
- Xu X, Yang Z, Chen Q, et al. Comparison of clinical characteristics of chronic cough due to non-acid and acid gastroesophageal reflux. Clin Respir J 2015;9:196-202. [Crossref] [PubMed]
- Asthma Group of Chinese Thoracic Society. Chinese national guideline on diagnosis and management of cough (2021). Zhonghua Jie He He Hu Xi Za Zhi 2022;45:13-46. [PubMed]
- Kopeo S, Ma J, Ferrucci J. The sensitivity and specificity of barium swallow in diagnosing gastroesophageal reflux in patients with chronic cough. Chest 2013;144:525A. [Crossref]
- Li N, Chen Q, Wen S, et al. Diagnostic accuracy of multichannel intraluminal impedance-pH monitoring for gastroesophageal reflux-induced chronic cough. Chron Respir Dis 2021;18:14799731211006682. [Crossref] [PubMed]
- Mainie I, Tutuian R, Castell DO. Comparison between the combined analysis and the DeMeester Score to predict response to PPI therapy. J Clin Gastroenterol 2006;40:602-5. [Crossref] [PubMed]
- Wiener GJ, Morgan TM, Copper JB, et al. Ambulatory 24-hour esophageal pH monitoring. Reproducibility and variability of pH parameters. Dig Dis Sci 1988;33:1127-33. [Crossref] [PubMed]
- Zhu Y, Tang J, Shi W, et al. Can acid exposure time replace the DeMeester score in the diagnosis of gastroesophageal reflux-induced cough? Ther Adv Chronic Dis 2021;12:20406223211056719. [Crossref] [PubMed]
- Sifrim D, Dupont L, Blondeau K, et al. Weakly acidic reflux in patients with chronic unexplained cough during 24 hour pressure, pH, and impedance monitoring. Gut 2005;54:449-54. [Crossref] [PubMed]
- Smith JA, Decalmer S, Kelsall A, et al. Acoustic cough-reflux associations in chronic cough: potential triggers and mechanisms. Gastroenterology 2010;139:754-62. [Crossref] [PubMed]
- Zhang L, Zhang M, Chen Q, et al. Improved diagnostic yield of symptom association probability involving only cough for gastroesophageal reflux-induced chronic cough. J Thorac Dis 2023; [Crossref]
- Wang S, Wen S, Bai X, et al. Diagnostic value of reflux episodes in gastroesophageal reflux-induced chronic cough: a novel predictive indicator. Ther Adv Chronic Dis 2022;13:20406223221117455. [Crossref] [PubMed]
- Ribolsi M, Luca Guarino MP, Balestrieri P, et al. The Results From Up-Front Esophageal Testing Predict Proton Pump Inhibitor Response in Patients With Chronic Cough. Am J Gastroenterol 2021;116:2199-206. [Crossref] [PubMed]
- Katz PO, Dunbar KB, Schnoll-Sussman FH, et al. ACG Clinical Guideline for the Diagnosis and Management of Gastroesophageal Reflux Disease. Am J Gastroenterol 2022;117:27-56. [Crossref] [PubMed]
- Khan BA, Sodhi JS, Zargar SA, et al. Effect of bed head elevation during sleep in symptomatic patients of nocturnal gastroesophageal reflux. J Gastroenterol Hepatol 2012;27:1078-82. [Crossref] [PubMed]
- Kahrilas PJ, Howden CW, Hughes N, et al. Response of chronic cough to acid-suppressive therapy in patients with gastroesophageal reflux disease. Chest 2013;143:605-12. [Crossref] [PubMed]
- Chang AB, Lasserson TJ, Gaffney J, et al. Gastro-oesophageal reflux treatment for prolonged non-specific cough in children and adults. Cochrane Database Syst Rev 2005;CD004823. [PubMed]
- Chen R, Qiu Z, Lai K. 2019 ERS cough guideline: consensus and controversy. J Thorac Dis 2020;12:7504-14. [Crossref] [PubMed]
- Lv HJ, Qiu ZM. Refractory chronic cough due to gastroesophageal reflux: Definition, mechanism and management. World J Methodol 2015;5:149-56. [Crossref] [PubMed]
- Dong R, Xu X, Yu L, et al. Randomised clinical trial: gabapentin vs baclofen in the treatment of suspected refractory gastro-oesophageal reflux-induced chronic cough. Aliment Pharmacol Ther 2019;49:714-22. [Crossref] [PubMed]
- Zhu Y, Xu X, Zhang M, et al. Pressure and length of the lower esophageal sphincter as predictive indicators of therapeutic efficacy of baclofen for refractory gastroesophageal reflux-induced chronic cough. Respir Med 2021;183:106439. [Crossref] [PubMed]
- Zhang M, Chen Q, Dong R, et al. Prediction of therapeutic efficacy of gabapentin by Hull Airway Reflux Questionnaire in chronic refractory cough. Ther Adv Chronic Dis 2020;11:2040622320982463. [Crossref] [PubMed]
- Tustumi F, Bernardo WM, Mariano da Rocha JR, et al. Effectiveness of antireflux surgery for the cure of chronic cough associated with gastroesophageal reflux disease. Asian J Surg 2021;44:2-10. [Crossref] [PubMed]
- Chandra KM, Harding SM. Therapy Insight: treatment of gastroesophageal reflux in adults with chronic cough. Nat Clin Pract Gastroenterol Hepatol 2007;4:604-13. [Crossref] [PubMed]
- Faruqi S, Molyneux ID, Fathi H, et al. Chronic cough and esomeprazole: a double-blind placebo-controlled parallel study. Respirology 2011;16:1150-6. [Crossref] [PubMed]
- Mukae H, Kaneko T, Obase Y, et al. The Japanese respiratory society guidelines for the management of cough and sputum (digest edition). Respir Investig 2021;59:270-90. [Crossref] [PubMed]
- Joo H, Moon JY, An TJ, et al. Revised Korean Cough Guidelines, 2020: Recommendations and Summary Statements. Tuberc Respir Dis (Seoul) 2021;84:263-73. [Crossref] [PubMed]
- Xu X, Lv H, Yu L, et al. A stepwise protocol for the treatment of refractory gastroesophageal reflux-induced chronic cough. J Thorac Dis 2016;8:178-85. [PubMed]
- Zhang M, Zhu Y, Dong R, et al. Gabapentin versus baclofen for treatment of refractory gastroesophageal reflux-induced chronic cough. J Thorac Dis 2020;12:5243-50. [Crossref] [PubMed]


