BRCA1 germline mutations dominate familial breast cancer patients in Henan China
Introduction
At present, the incidence of breast cancer in the world is obviously increasing, which seriously affects women’s life and health. According to epidemiologic data, breast cancer exist familial clustering individual and race differences (1). These suggest that genetic factors play an important role in the initiation, promotion, and progression of mammary carcinogenesis. As rapid advances have been made in molecular biology and bioinformatic analyses, a lot of mutations were detected in BRCA1 genes. One study has shown that gene mutations of BRCA could be inherited as an autosomal dominant (2). More than 80% of hereditary breast cancer cases carried gene mutations of BRCA by age 80, they had an early age at onset (3). Role of genetic testing for BRCA genes is to find hotspot pathogenesis and screen for high-risk groups. It is a manifestation of assessing the risk of familial breast cancer and its early diagnosis. Although a few studies have been carried out for investigating BRCA mutations in familial breast cancer in Chinese women (4,5), the spectrums of BRCA gene mutation are different due to other environmental and geographical factors (6,7). In this cohort study the aim was to analyze BRCA1 gene mutations of familial breast cancer patients with Han nationality in Henan, China.
Methods
Study participants
Our study was approved by the Ethics Committee of the Institute for Henan Population and Family Planning (No. 2014YC005). Written informed consent was obtained for all patients.
This study enrolled 59 breast cancer patients and 122 healthy donors whom were unrelated to each other. All breast cancer samples were diagnosed according to histopathologic examination and came from the Center of Human Genetic Resources, Institute for Henan Population and Family Planning.
Blood sampling, DNA preparation
Venous blood was collected into two 4.5-mL sodium citrate anticoagulant tubes and stored at +4 °C until processing. Within 4 h after blood collecting, genomic DNA was extracted using QIAamp DNA Mini Kit (QIAGEN GmbH, Hilden, Germany) and stored at −80 °C.
Gene mutations testing using denaturing high-performance liquid chromatography (DHPLC) assay
This assay included: all PCR primers were designed using the software PRIMER3-OUTPUT according to BRCA1 and BRCA2 gene exon sequences reported in Gene Bank. There were 24 primers on BRCA1 gene. Multiplex PCR was performed with a thermal cycler and PCR products were purified. Purified PCR were denaturalized at 94 °C for 30 seconds and cooled down to room temperature within 30–40 minutes. This could contribute to forming hetero-duplex between normal BRCA gene and mutated gene. Denaturation temperature of target DNA fragments was predicted by WAVE Maker/Investigator software. Equal sample loading was 5 μL. According to changes of peak at 254 nm ultraviolet wavelengths, BRCA gene mutations were detected. Samples with BRCA gene mutations were PCR amplified again and confirmed by sequencing at Sangon Biotechnology Limited Company.
Statistical analysis
The difference in the rate of BRCA gene mutation between patient and healthy donor was analyzed χ2 test. P values <0.05 was considered as statistically significant.
Results
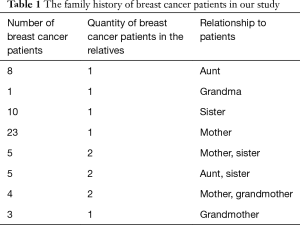
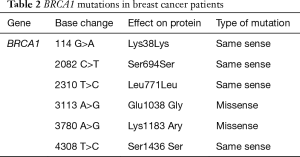
The breast cancer patients were a mean age of 42.5±7.9 years. They have at least one or more first-degree relatives affected with breast cancer (Table 1). Among these patients the percent that both mother and daughter had breast cancer was 39% and the percent that three generations all had breast cancer was 6.8%. These patients BRCA1 gene mutations were detected in 31 of 59 breast cancer patients. These mutations are mostly located in exon 11 of BRCA1 gene and the mutation rate is 52.54% (31/59). Mutation types are same sense mutations and missense mutation (Table 2). One novel missense mutation (3780 A>G) was found in this study.
Full table
Full table
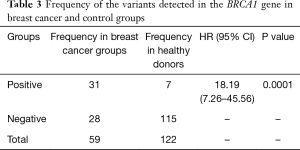
BRCA1 mutations were detected in 7 of 122 healthy donors, and the mutation rate is 5.74%. Comparing to two rates of BRCA gene mutations between patients and healthy donors there were statistically significant difference (P=0.0001) (Table 3).
Full table
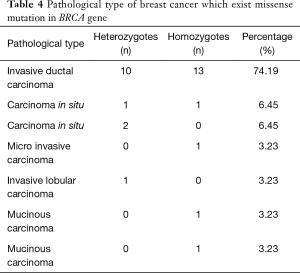
The BRCA1 mutation rates are varied regarding the subtypes of breast cancer. The mutation rate with in situ and tubule ductal carcinoma is 6.45% (2/31), respectively. One mutated case was found in micro invasive carcinoma, invasive lobular carcinoma, medullary carcinoma and mucinous carcinoma, and the mutation rate is 3.22% (1/31). Most significantly, 23 patients with invasive ductal carcinoma were shown BRCA1 mutations and the mutation rate was 74.19% (23/31) (Table 4). According to AJCC Cancer Staging Manual (the sixth edition) clinic stage was shown in the Table 5.
Full table

Full table
Discussion
BRCA are tumor suppressors and play roles in DNA impair and posttranscriptional control. BRCA gene is closely related to the occurrence and development of breast cancer (8). They can inhibit cell proliferation and induce apoptosis (9,10). Loss of function of BRCA by mutations might lead to abnormal cell cycle and cell growth, and then cancers. Our result in this study shown the rate of BRCA1 gene mutation is 52.54% among familiar breast cancer patients, which is higher than published result (11). A possible reason is that invasive ductal carcinoma is the main histopathologic types of breast cancer in this cohort of patients, in which the mutation rates in BRCA1 even as high as 74.19%. One research have shown that BRCA1 gene mutation invasive ductal carcinoma exist high correlations (4). So BRCA1 gene mutation is more likely to harbor invasive ductal carcinoma. On the other hand, there was a high incidence of breast cancer in the family the percent of BRCA1 mutation was more than 80% (12). In our study the rate that two generations both had breast cancer were 39% and the rate that three generations all had breast cancer was 6.8%. So it had shown that family history was correlation to BRCA1 gene mutation.
Our result had shown the rate of BRCA gene mutation among healthy donors was 5.74%, which was consistent with published result (4). There was statistical significance between breast cancer patients and healthy donors. So the result in this study suggested that women with familial breast cancer are good candidates for BRCA testing and women who carry a BRCA mutation should gain necessary inspections in order to find breast cancer.
Rebbeck et al.’s research (13) have shown that carcinogenesis of BRCA is associated with specific location and type of gene mutations. The localization of BRCA gene mutation in breast cancer cluster region (BCCR) will increase risk of breast cancer, and female carrier whose BRCA1 gene mutation is located in EXON 11 will suffer from breast cancer in early youth. Our study had shown that BRCA 1 gene mutation was missense mutation at EXON 11 and located in BCCR. A mean age of patients group was 42.5±7.9 years. The hotspots of BRCA1 gene mutation in familial breast cancer with Han nationality in Henan, China might locate in 3113 A>G and 3780 A>G. There was no BRCA2 mutation in this study. So BRCA1 mutation is more frequent than BRCA2 mutation in Henan, China. Furthermore, we found one novel mutation 3780 A>G in BRCA1 in this study.
In conclusion, in a cohort of 59 familiar breast cancer patients and 122 healthy donors in Henan China, we found that the mutation rate of BRCA1 was as high as 52.54% in patients, and do not identify any mutation in BRCA2. Mutation rates were also varied in different subtypes of breast cancer. The subtype with the highest mutation rate was invasive ductal carcinoma, which was 74.19%. Our report here indicates that BRCA1 is the major mutated gene in this cohort of patients. Therefore, the preventing and diagnosis of breast cancer with inherited patients should be mainly focus on detecting BRCA1 germline mutations at least in Henan area of China.
Acknowledgements
We thank Dong Wang, Ph.D. at Investigator of Cancer Genomics, Department of Basic Medical Sciences School of Medicine Tsinghua University for the article writing.
Funding: This work was supported by the grant from the key project for public benefit of Henan (No. 081100911100).
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: Our study was approved by the Ethics Committee of the Institute for Henan Population and Family Planning (No. 2014YC005). Written informed consent was obtained for all patients.
References
- Mavaddat N, Antoniou AC, Easton DF, et al. Genetic susceptibility to breast cancer. Mol Oncol 2010;4:174-91. [Crossref] [PubMed]
- Newman B, Austin MA, Lee M, et al. Inheritance of human breast cancer: evidence for autosomal dominant transmission in high-risk families. Proc Natl Acad Sci U S A 1988;85:3044-8. [Crossref] [PubMed]
- Mavaddat N, Peock S, Frost D, et al. Cancer risks for BRCA1 and BRCA2 mutation carriers: results from prospective analysis of EMBRACE. J Natl Cancer Inst 2013;105:812-22. [Crossref] [PubMed]
- Zhang J, Pei R, Pang Z, et al. Prevalence and characterization of BRCA1 and BRCA2 germline mutations in Chinese women with familial breast cancer. Breast Cancer Res Treat 2012;132:421-8. [Crossref] [PubMed]
- Chen W, Pan K, Ouyang T, et al. BRCA1 germline mutations and tumor characteristics in Chinese women with familial or early-onset breast cancer. Breast Cancer Res Treat 2009;117:55-60. [Crossref] [PubMed]
- Yablonski-Peretz T, Paluch-Shimon S, Gutman LS, et al. Screening for germline mutations in breast/ovarian cancer susceptibility genes in high-risk families in Israel. Breast Cancer Res Treat 2016;155:133-8. [Crossref] [PubMed]
- de Oliveira ES, Soares BL, Lemos S, et al. Screening of the BRCA1 gene in Brazilian patients with breast and/or ovarian cancer via high-resolution melting reaction analysis. Fam Cancer 2016;15:173-81. [Crossref] [PubMed]
- Rebbeck TR, Friebel TM, Mitra N, et al. Inheritance of deleterious mutations at both BRCA1 and BRCA2 in an international sample of 32,295 women. Breast Cancer Res 2016;18:112. [Crossref] [PubMed]
- Einbeigi Z, Enerbäck C, Wallgren A, et al. BRCA1 gene mutations may explain more than 80% of excess number of ovarian cancer cases after breast cancer - a population based study from the Western Sweden Health Care region. Acta Oncol 2010;49:361-7. [Crossref] [PubMed]
- Privat M, Aubel C, Arnould S, et al. AKT and p21 WAF1/CIP1 as potential genistein targets in BRCA1-mutant human breast cancer cell lines. Anticancer Res 2010;30:2049-54. [PubMed]
- Cavallone L, Arcand SL, Maugard CM, et al. Comprehensive BRCA1 and BRCA2 mutation analyses and review of French Canadian families with at least three cases of breast cancer. Fam Cancer 2010;9:507-17. [Crossref] [PubMed]
- Scully R, Livingston DM. In search of the tumour-suppressor functions of BRCA1 and BRCA2. Nature 2000;408:429-32. [Crossref] [PubMed]
- Rebbeck TR, Mitra N, Wan F, et al. Association of type and location of BRCA1 and BRCA2 mutations with risk of breast and ovarian cancer. JAMA 2015;313:1347-61. [Crossref] [PubMed]


