Short-term and 1-year outcomes after MitraClip therapy in functional versus degenerative mitral regurgitation patients: a systematic review and meta-analysis
Introduction
Mitral regurgitation (MR) is among the most common valvular heart disease (1). MR is classified into degenerative MR (DMR) and functional MR (FMR), which are two distinct disease entities. DMR originates from a structural or degenerative abnormality of the mitral valve apparatus, while FMR is caused by a disease of the left ventricle, which results in malcoaptation of the mitral leaflets (2). As a result, different therapeutic strategies were adopted against different MR etiologies. For DMR, recommended interventions include surgical mitral valve repair and mitral valve replacement. Mitral valve repair is preferred over mitral valve replacement if a successful and durable repair can be achieved. For FMR, medical therapy for LV dysfunction is the first-line treatment and the role of surgery is not well defined (1).
The MitraClip (MC) system is a novel transcatheter treatment option drawing on the experience of the edge-to-edge mitral valve repair pioneered by Alfieri and colleagues and has been shown to be an effective treatment for patients with severe MR, especially for those with prohibitive surgical risk (3,4). A meta-analysis in 2014 demonstrated that the safety and efficacy are comparable between the MC system and open mitral valve repair (oMVR) at 1-year follow-up (5). In the 2017 European Society of Cardiology/European Association of the Cardio-Thoracic Surgery guidelines, percutaneous edge-to-edge repair is recommended for both DMR and FMR (recommendation class IIb, level of evidence C). However, the American Heart Association and American College of Cardiology only approved MC therapy for patients with severe, symptomatic DMR and at a high or prohibitive risk of surgery (recommendation class IIb, level of evidence B) and did not approve its clinical use for FMR (6-8). MITRA-FR study is the first ongoing, multi-center, randomized and open-label trial in France that compares the effectiveness and safety of MC system plus optimal medical therapy (OMT) versus only OMT (9). The outcome of the MITRA-FR trial can indicate the role of MC in FMR
As mentioned above, the guidelines in Europe and the United States differ in the use of MC in FMR. Moreover, no comparison regarding both short-term and 1-year outcomes after MC therapy has been reported in patients with DMR and FMR. The aim of this study was to perform a meta-analysis investigating the differences of short-term and 1-year outcomes after MC in these two different etiologies.
Methods
Search strategies
This systematic review and meta-analysis was conducted in accordance with the Guidelines for Meta-Analyses and Systematic Reviews of Observational Studies (10) and the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (11). Studies regarding MC were obtained through a systematic search of PubMed, EMBASE and Web of Science until November 2017 by two independent reviewers. The titles and abstracts were assessed carefully to include eligible studies.
Inclusion criteria
Studies that met the following criteria were included in this meta-analysis: (I) observational study design; (II) study with human subjects; (III) study that evaluated the use of MC in patients with DMR and FMR; (IV) observed data included a 30-day or one-year follow-up. We included studies without language restriction.
Study selection
Two reviewers screened the titles and abstracts of the studies to initially screen for eligible studies. Eligible studies from the initial screening were reviewed in full and assessed for compliance with the inclusion criteria. Any discrepancies between the two reviewers were resolved through consensus of a third reviewer.
Data extraction
Using a prepared data extraction form, two blinded reviewers independently performed data extraction. In the case where studies had overlapping populations, only the manuscript reporting the larger number of patients was selected. The following information was collected and tabulated: (I) publication details, including first author’s last name, country, publication year, study design and study population; (II) characteristics of the study population, including sample size, age, and gender; (III) baseline clinical and echocardiographic characteristics; (IV) outcomes during follow-up. Data extraction conflicts were discussed and resolved with another author.
Quality assessment
The quality of each study was assessed according to different study designs. We used the Newcastle-Ottawa scale to evaluate cohort and case-control studies.
Statistical analysis
Stata 14.0 (StataCorp LP, College Station, Texas, USA) was utilized. Continuous variables were reported as the mean and standard deviation (SD). Pooled effect sizes for continuous variables were presented as the standard mean difference (SMD) with 95% confidence interval (CI). Pooled effect sizes for categorical variables were presented as the risk ratio (RR) values with 95% CI using a binary random effects model if I2 >50% and the fixed effects model if I2 <50% with the Mantel-Haenszel method. Statistical heterogeneity was assessed by the χ2 test and quantified using the I2 statistic. I2 values of <40%, 40–60% or >60% indicated low, moderate or substantial heterogeneity, respectively. The sensitivity analysis and subgroup analysis were conducted in a random predefined manner to search for sources of heterogeneity. Publication bias was evaluated using the funnel plot. Two-tailed P value less than 0.05 was considered statistically significant.
Results
Search results
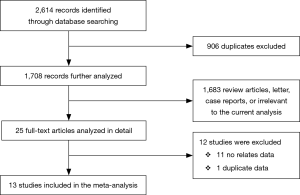
A flow chart of the study selection process is shown in Figure 1. A total of 2,614 articles were retrieved. After removal of irrelevant studies, 25 full-text studies were further assessed. Among these 25 studies, 11 were excluded because neither short-term nor 1-year results were reported. One duplicate study was also excluded. Thirteen studies were included in the final analysis.
Characteristics of the included studies
A total of 13 studies (12-24) were included in the meta-analysis, one of which was a conference report that fit the inclusion criteria (23). Among these studies, 11 were prospective while 2 were retrospective. The main features of the included studies were summarized in Table 1. A total of 2,351 patients (758 DMR and 1,593 FMR) experienced MC therapy in Europe (n=1,900, 80.8%), Asia (n=163, 6.9%) and the USA (n=288, 12.3%).

Full table
Baseline characteristics of the patients
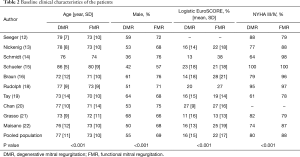
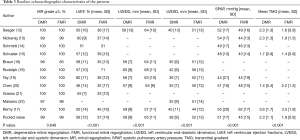
The baseline characteristics, including clinical and echocardiographic features, were shown in Tables 2 and 3. Results comparing the two groups, first report the DMR group result followed by the FMR group result. Patients in the DMR group were older (76.5 vs. 72.9 years, P<0.001). Males occupied a higher proportion in the FMR group (55.1% vs. 68.6%, P<0.001). Notably, FMR patients presented a higher risk profile. The Logistic EuroSCORE was significantly higher in the FMR group (16.0% vs. 22.3%, P<0.001) due to more comorbidities and previous interventions, including previous MI (98/616 vs. 460/1,311, P<0.001), diabetes mellitus (112/641 vs. 484/1,387, P<0.001), renal insufficiency (175/631 vs. 631/1,398, P<0.001), coronary artery bypass grafting (105/617 vs. 446 /1,327, P<0.001), percutaneous coronary intervention (124/539 vs. 366/1,167, P<0.001) and cardiac resynchronization therapy (8/311 vs. 98/681, P<0.001). No difference was observed in previous hypertension incidence (407/568 vs. 1,019/1,338, P=0.054) and AF (357/628 vs. 669/1,333, P=0.96). Moreover, more FMR patients were at NYHA grade III or IV (555/690 vs. 1,327/1,497, P<0.001). For the echocardiographic parameters, left ventricular ejection fractions (LVEF) (56.3% vs. 36.6%), systolic pulmonary artery pressure (50.8 vs. 45.3 mmHg) and mean transmitral gradient (mTMG) (2.29 vs. 1.88 mmHg) were significantly higher in the DMR group, while the left ventricular end-diastolic dimension (LVEDD) (57.6 vs. 65.1 mm) and left ventricular end-systolic dimension (LVESD) (37.8 vs. 52.0 mm) were higher in the FMR group. The number of patients with MR grade 3–4 was similar (680/688 vs. 1,430/1,449, P=0.646).
Full table
Full table
Acute procedural outcomes and 30-day follow-up results
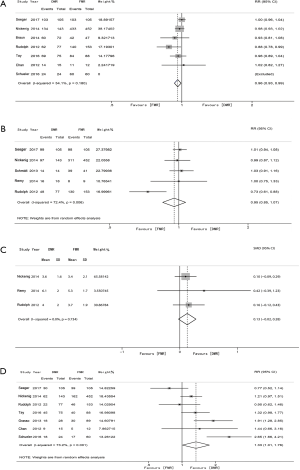
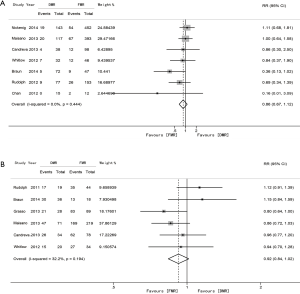
The acute procedural success (APS) rate was defined as successful and stable MC placement with residual MR ≤2+ upon discharge. APS was reported in 7 studies, which demonstrated a higher APS rate in the FMR group (91.2% vs. 95.2%; RR 0.96; 95% CI: 0.93 to 0.99; P=0.016) with evidence of low heterogeneity among the studies (I2 =34.1%) (Figure 2A). An MR grade of 0–2 after the MC procedure was reported in 5 studies without a significant difference (76.8% vs. 77.1%; RR 0.95; 95% CI: 0.85 to 1.07; P=0.428) and with evidence of substantial heterogeneity (I2 =72.4%) (Figure 2B). Post-implantation mTMG was reported in 3 studies (3.92 vs. 3.50 mmHg; SMD 0.13; 95% CI: −0.02 to 0.28; P=0.098), with evidence of no heterogeneity (I2=0.0%) (Figure 2C). The proportion of patients who had received two clips or more were significantly higher in the DMR group in the 7 studies reporting this index (41.4% vs. 35.7%; RR 1.33; 95% CI: 1.01 to 1.76; P=0.043) with evidence of substantial heterogeneity (I2 =73.2%) (Figure 2D). Procedural death was investigated in 7 studies where 25 deaths were reported, of which 12 were in DMR and 13 in FMR.
Both the 30-day mortality rate and NYHA I–II were reported in 4 studies. Although the trend suggested the mortality rate was higher in the DMR group, the result was not statistically significant (0.05% vs. 0.03%; RR 1.68; 95% CI: 0.88 to 3.22; P=0.118; I2=0.0%) (Figure 3A). No difference was observed in the rate of 30-day NYHA grade I–II (85.3% vs. 78.7%; RR 1.08; 95%CI: 0.96 to 1.22; P=0.211; I2 =50.7%) (Figure 3B).
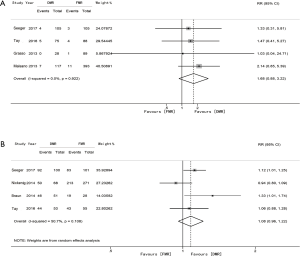
One-year follow-up outcomes
One-year mortality was reported in 7 studies. As shown in Figure 4A, the rate did not differ significantly between DMR and FMR (13.0% vs. 15.2%; RR 0.86; 95% CI: 0.67 to 1.12; P=0.268), with no evidence of heterogeneity (I2 =0.0%).
At 1-year follow-up, the number of patients with MR ≤2 was 156 out of 208 in the DMR group and 389 out of 482 in the FMR group (75.0% vs. 80.7%; RR 0.92; 95% CI: 0.84 to 1.02; P=0.106), with evidence of low heterogeneity (I2 =32.2%) (Figure 4B). LVEF (57.7% vs. 36.0%) remained higher and LVEDV (114.6 vs. 177.7 mL) and LVESV (50.7 vs. 109.6 mL) remained lower in the DMR group (P<0.001). The proportion of patients achieving NYHA grade I–II at 1-year follow-up was not examined because less than 3 studies reported this index.
Furthermore, we derived funnel-plot distributions of the 1-year mortality rate and MR ≤2 from the SE of the logRR plotted against the logRR confirming the absence of publication bias and a small study effect (Figure 5).

Heterogeneity among studies
Significant heterogeneity was observed in studies investigating the number of patients with implanted MC ≥2, MR grade 0–2 after MC procedure and 30-day NYHA I–II (I2 =73.2%, 72.4%, and 50.7%, respectively). First, to identify the source of heterogeneity among the studies investigating the number of patients with implanted MC ≥2, a subgroup analysis was conducted based on the proportion of DMR patients and the subject number (Figure 6). The studies by Schueler et al. and Grasso et al. (15,21), which the subject number is small and the proportion of DMR patients is less than 30%, were pooled into subgroup 1. Subgroup 1 showed that the number of DMR patients with implanted MC ≥2 was more than twice that of the FMR patients with a mild heterogeneity (RR =2.20; 95% CI: 1.60 to 3.02; P<0.001; I2=8.9%). Meanwhile, the remaining 5 cross-sectional studies was pooled into subgroup 2. Subgroup 2 showed the number of DMR patients with implanted MC ≥2 was not significantly greater than that of the FMR patients (RR =1.11; 95% CI: 0.90 to 1.36; P=0.322; I2 =35.7%) (12,13,18-20).
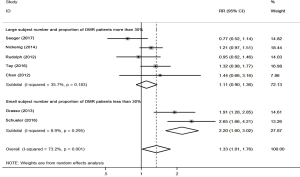
Sensitivity analyses were also performed to identify other sources of heterogeneity. When examining the heterogeneity between studies examining the MR grade 0–2 after MC procedure, the study by Rudolph et al. (18) was excluded since the APS rate was lower than that of others and the resulting I2 was 0%. For examining heterogeneity across studies on the 30-day NYHA I–II, the study with the largest ratio of patients lost to follow-up (13), was excluded, resulting in no significant influences on heterogeneity.
Discussion
The present study is a novel meta-analysis investigating the differences of both short-term and 1-year results between DMR and FMR patients receiving MC implantation. The main findings are as follows. First, patients with FMR were at a higher baseline risk profile. Second, similar mTMG and the percentage of patients with MR grade 0–2 immediately after MC implantation were reported between DMR and FMR. Third, a significantly higher rate of APS and lower number of implanted MC were found in patients with FMR. Fourth, no differences were found in the 30-day mortality rate and percentage of patients with NYHA grade I–II. Finally, no differences were reported in terms of the 1-year mortality rate and MR grade 0–2.
MR is divided into DMR and FMR. There are ongoing discussions about the appropriateness of the MC system in FMR (25,26). Only one randomized controlled trial, the EVEREST II, has investigated 1-year outcomes after MC treatment. The EVEREST II trial showed MC had higher safety compared with surgery, but was less effective at 1 year. However, it did not directly compare the differences in terms of effectiveness and safety between the two etiologies (27). To date, the use of MC in these two etiologies remains unclear. The ESC/EACTS guidelines recommend for MC for both DMR and FMR, whereas the AHA/ACC guidelines recommend MC only for DMR patients at a high or prohibitive risk of surgery (6,8). Chiarito et al. demonstrated that MC is likely to be an effective and safe option in patients with both function and degenerative MR. However, short-term results was not compared. Thus, we investigated both short-term and 1-year results between DMR and FMR patients who underwent MC implantation in order to further demonstrate the effectiveness and safety of MC in these two etiologies (28).
This meta-analysis demonstrates a higher overall risk profile in FMR patients. There are three contemporary surgical scores for predicting mortality after percutaneous mitral valve repair with MC- Logistic EuroSCORE, Society of Thoracic Surgeons Predicted Risk of Mortality, and EuroSCORE II. The most commonly used scoring system is the Logistic EuroSCORE (LES); therefore, we adapted LES as the metric to assess baseline overall risk (3). Patients in the FMR group had a higher LES, a lower LVEF, a higher proportion at NYHA grade III or IV and more comorbidities at baseline. However, we need to consider that DMR patients were generally older in our study, which may have counterbalanced the factors that were negative for baseline characteristics in FMR patients.
The short-term results were comparable between DMR and FMR and the APS rate was higher in patients with FMR. Schillinger et al. reported the impact of the learning curve on APS, which was 80% and 92% from early to advanced stages, respectively (29). Thus, it is reasonable to speculate that in our study, the use of the MC system was in a relatively mature period. Interestingly, we found that more DMR patients were implanted with two or more MCs. From a clinical indication perspective, it is unclear what the optimal number of implanted MC devices is to achieve a residual MR grade of 2 or lower in the absence of mitral stenosis. Currently, the MC procedure is individualized according to valve anatomy, and several parameters need to be considered for the procedural strategy, such as the number of clips to implant, including the mitral valve opening area, the degree of mobility of the mitral leaflets, the extent of the mitral regurgitant jet and the pre-procedure TMG (30,31). However, it is important to note that the residual MR grade and post-implantation TMG rather than the number of MCs are more meaningful parameters associated with 1-year prognosis as they are more likely to reflect the risk of persistent symptoms after the procedure (30). A high TMG or residual MR can be observed after one clip, whereas the gradients may remain normal even after 3 implanted clips. In our study, both post-implantation TMG and residual MR grades from 0–2 were comparable, which may indicate similar outcomes between FMR and DMR over a relatively long period.
Differences at baseline must be considered when investigating 1-year clinical endpoints. The characteristics of the sample with FMR showed a much higher risk profile. A worse prognosis was associated with the natural history of the disease, and we expected a worse 1-year result in the FMR group. However, in our study, the 1-year mortality rate was similar. Meanwhile, the rate of mortality was unexpectedly high, which could be related to severely depressed EF and other comorbidities (32,33). Another concern regarding the use of MC in FMR patients is MR recurrence, but a similar percentage of patients with a 1-year MR grade 0–2 indicated that interruption of the LV dilation-MR worsening loop did help prevent MR recurrence (34).
It is still too early to determine the use of MC in different MR etiologies. A recent data analysis report suggested that indications based on current guidelines might underestimate the opportunities of MC treatment. In comparison, the anatomical criteria might evolve due to increasing experience of MC therapy and new imaging technologies. The new technologies would provide MC a new way to visualize and facilitate its clinical development (35). Perhaps we can reconsider MC as a treatment option in patients with a relatively lower risk profile instead of a rescue therapy in patients at prohibitive surgical risk.
Study limitations
First, the patients in the sample were mostly European. Based on previous publications, including large registries such as ACCESS (23), SENTINEL (14) and TRAMI (36) (excluded from our study because it did not compare DMR and FMR), the main indication of MC in Europe is FMR, whereas in the USA, it is the DMR (27). This difference is reflected in our study in which the number of cases of FMR was twice that of DMR. The results may be influenced by more experience with treating FMR and lead to potential bias. Second, DMR patients have a diseased mitral valve but a generally normal myocardium; thus, they are expected to have a relatively favorable outcome after MC therapy. Conversely, FMR patients have no underlying valvular disorder but advanced heart failure with LV remodelling and dysfunction (1). Their outcomes should be that of advanced heart failure. With the progression of heart failure, one can expect a progression of MR. As a result, a follow-up longer than one year is warranted. For example, the EVEREST II 5-year results identified that FMR was a predictor of decreased survival regardless of percutaneous or surgical treatment, which indicates that the etiology of FMR may play a more important role along with the progression of the disease (27). Third, the lack of RCTs reflect the continued development of MC and a lack of standard indices restricted the uniformity of data collection. Finally, this study is a study-level rather than a patient-level analysis, thus the generalizability is limited.
Conclusions
This meta-analysis pooled 13 studies totalling 2,351 patients receiving MC therapy. The study found that outcomes after MC therapy is similar between FMR and DMR patients up until 1-year follow-up, despite a higher baseline risk profile in FMR patients. The APS rate was higher in FMR patients. Large randomized controlled trials are warranted to fully and further assess the clinical impact of the procedure in these two different MR etiologies over a longer period of time, and clinical trials such as MITRA-FR are expected to disclose the role of MC in FMR in the future.
Acknowledgements
Funding: This work was supported by the National Natural Science Foundation of China [No. 81470426 to W.C.]; and the PUMCH Young and Middle-aged Investigation Fund, Key Project [No. PUMCH-2016-1.12 to W.C.].
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
- Nishimura RA, Vahanian A, Eleid MF, et al. Mitral valve disease—current management and future challenges. Lancet 2016;387:1324-34. [Crossref] [PubMed]
- Kar S. Percutaneous transcatheter mitral valve repair: adding life to years. J Am Coll Cardiol 2013;62:1062-4. [Crossref] [PubMed]
- Adamo M, Capodanno D, Cannata S, et al. Comparison of Three Contemporary Surgical Scores for Predicting All-Cause Mortality of Patients Undergoing Percutaneous Mitral Valve Repair With the MitraClip System (from the Multicenter GRASP-IT Registry). Am J Cardiol 2015;115:107-12. [Crossref] [PubMed]
- Mendirichaga R, Singh V, Blumer V, et al. Transcatheter Mitral Valve Repair With MitraClip for Symptomatic Functional Mitral Valve Regurgitation. Am J Cardiol 2017;120:708-15. [Crossref] [PubMed]
- Bail DH. (Meta)-Analysis of Safety and Efficacy Following Edge-to-Edge Mitral Valve Repair Using the MitraClip System. J Interv Cardiol 2015;28:69-75. [Crossref] [PubMed]
- Falk V, Baumgartner H, Bax JJ, et al. 2017 ESC/EACTS Guidelines for the Management of Valvular Heart Disease. Eur J Cardiothorac Surg 2017;52:616-64. [Crossref] [PubMed]
- Nishimura RA, Otto CM, Bonow RO, et al. 2017 AHA/ACC Focused Update of the 2014 AHA/ACC Guideline for the Management of Patients With Valvular Heart Disease: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. J Am Coll Cardiol 2017;70:252-89. [Crossref] [PubMed]
- Nishimura RA, Otto CM, Bonow RO, et al. 2014 AHA/ACC Guideline for the Management of Patients With Valvular Heart Disease: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. Circulation 2014;129:e521-643. [Crossref] [PubMed]
- Obadia JF, Armoiry X, Iung B, et al. The MITRA-FR study: design and rationale of a randomized study of percutaneous mitral valve repair compared with optimal medical management alone for severe secondary mitral regurgitation. EuroIntervention 2015;10:1354-60. [Crossref] [PubMed]
- Stroup DF, Berlin JA, Morton SC, et al. Meta-analysis of observational studies in epidemiology: a proposal for reporting. Meta-analysis Of Observational Studies in Epidemiology (MOOSE) group. JAMA 2000;283:2008-12. [Crossref] [PubMed]
- Moher D, Liberati A, Tetzlaff J, et al. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. Ann Intern Med 2009;151:264-9, w64.
- Seeger J, Müller P, Gonska B, et al. Percutaneous mitral valve repair in primary compared with secondary mitral valve regurgitation using the Mitral Valve Academic Research Consortium criteria. J Invasive Cardiol 2017;29:145-50. [PubMed]
- Nickenig G, Estevez-Loureiro R, Franzen O, et al. Percutaneous mitral valve edge-to-edge repair: in-hospital results and 1-year follow-up of 628patients of the 2011-2012 Pilot European Sentinel Registry. J Am Coll Cardiol 2014;64:875-84. [Crossref] [PubMed]
- Schmidt FP, von Bardeleben RS, Nikolai P, et al. Immediate effect of the MitraClipw procedure on mitral ring geometry in primary and secondary mitral regurgitation. Eur Heart J Cardiovasc Imaging 2013;14:851-7. [Crossref] [PubMed]
- Schueler R, Kaplan S, Melzer C, et al. Impact of interventional edge-to-edge repair on mitral valve geometry. Int J Cardiol 2017;230:468-75. [Crossref] [PubMed]
- Braun D, Lesevic H, Orban M, et al. Percutaneous Edge-to-Edge Repair of the Mitral Valve in Patients With Degenerative Versus Functional Mitral Regurgitation. Catheter Cardiovasc Interv 2014;84:137-46. [Crossref] [PubMed]
- Remy T, Bertog SC, Wunderlich N, et al. Change in Mitral Annular Size and Geometry after MitraClipW Implantation in Patients with Functional and Degenerative Mitral Regurgitation. J Interv Cardiol 2014;27:516-24. [Crossref] [PubMed]
- Rudolph V, Lubos E, Schlüter M, et al. Aetiology of mitral regurgitation differentially affects 2-year adverse outcomes after MitraClip therapy in high-risk patients. Eur J Heart Fail 2013;15:796-807. [Crossref] [PubMed]
- Tay E, Muda N, Yap J, et al. The MitraClip Asia-Pacific Registry: Differences in Outcomes Between Functional and Degenerative Mitral Regurgitation. Catheter Cardiovasc Interv 2016;87:E275-81. [Crossref] [PubMed]
- Chan PH, She HL, Alegria-Barrero E, et al. Real-world experience of MitraClip for treatment of severe mitral regurgitation. Circ J 2012;76:2488-93. [Crossref] [PubMed]
- Grasso C, Capodanno D, Scandura S, et al. One- and Twelve-Month Safety and Efficacy Outcomes of Patients Undergoing Edge-to-Edge Percutaneous Mitral Valve Repair (from the GRASP Registry). Am J Cardiol 2013;111:1482-7. [Crossref] [PubMed]
- Maisano F, Franzen O, Baldus S, et al. Percutaneous Mitral Valve Interventions in the Real World: Early and 1-Year Results From the ACCESS-EU, A Prospective, Multicenter, Nonrandomized Post-Approval Study of the MitraClip Therapy in Europe. J Am Coll Cardiol 2013;62:1052-61. [Crossref] [PubMed]
- Candreva A, Latib A, Taramasso M, et al. MitraClip therapy in patients with functional or degenerative mitral regurgitation: a single-centre clinical experience. EuroPCR Abstracts 2013.
- Whitlow PL, Feldman T, Pedersen WR, et al. Acute and 12-Month Results With Catheter-Based Mitral Valve Leaflet Repair: The EVEREST II (Endovascular Valve Edge-to-Edge Repair) High Risk Study. J Am Coll Cardiol 2012;59:130-9. [Crossref] [PubMed]
- Jilaihawi H, Hussaini A, Kar S. New Horizons: MitraClip: a novel percutaneous approach to mitral valve repair. J Zhejiang Univ Sci B 2011;12:633-7. [Crossref] [PubMed]
- Siegel RJ, Luo H, Biner S. Transcatheter valve repair/ implantation. Int J Cardiovasc Imaging 2011;27:1165-77. [Crossref] [PubMed]
- Feldman T, Kar S, Elmariah S, et al. Randomized comparison of percutaneous repair and surgery for mitral regurgitation: 5-year results of EVEREST II. J Am Coll Cardiol 2015;66:2844-54. [Crossref] [PubMed]
- Chiarito M, Pagnesi M, Martino EA, et al. Outcome after percutaneous edge-to-edge mitral repair for functional and degenerative mitral regurgitation: a systematic review and meta-analysis. Heart 2018;104:306-12. [PubMed]
- Schillinger W, Athanasiou T, Weicken N, et al. Impact of the learning curve on outcomes after percutaneous mitral valve repair with MitraClipw and lessons learned after the first 75 consecutive patients. Eur J Heart Fail 2011;13:1331-9. [Crossref] [PubMed]
- Armstrong EJ, Rogers JH, Swan CH, et al. Echocardiographic predictors of single versus dual MitraClip device implantation and long-term reduction of mitral regurgitation after percutaneous repair. Catheter Cardiovasc Interv 2013;82:673-9. [PubMed]
- Alegria-Barrero E, Chan PH, Foin N, et al. Concept of the central clip: when to use one or two MitraClips®. EuroIntervention 2014;9:1217-24. [Crossref] [PubMed]
- Puls M, Tichelbäcker T, Bleckmann A, et al. Failure of acute procedural success predicts adverse outcome after percutaneous edge-to-edge mitral valve repair with MitraClip. EuroIntervention 2014;9:1407-17. [Crossref] [PubMed]
- Taramasso M, Maisano F, Latib A, et al. Clinical outcomes of MitraClip for the treatment of functional mitral regurgitation. EuroIntervention 2014;10:746-52. [Crossref] [PubMed]
- Grayburn PA, Foster E, Sangli C, et al. Relationship between the magnitude of reduction in mitral regurgitation severity and left ventricular and left atrial reverse remodeling after MitraClip therapy. Circulation 2013;128:1667-74. [Crossref] [PubMed]
- Deuschl F, Schofer N, Lubos E, et al. MitraClip—data analysis of contemporary literature. J Thorac Dis 2015;7:1509-17. [PubMed]
- Puls M, Lubos E, Boekstegers P, et al. One-year outcomes and predictors of mortality after MitraClip therapy in contemporary clinical practice: results from the German transcatheter mitral valve interventions registry. Eur Heart J 2016;37:703-12. [Crossref] [PubMed]