
Minimally invasive embolectomy of HeartWare left ventricular assist device outflow graft
Introduction
Nowadays permanent mechanical circulatory support is a common treatment option for end-stage heart failure. Due to a shortage of donor heart transplantation not always being available for the patient in need to get life-saving treatment, a left ventricular assist devices (LVAD) is a suitable treatment option to overcome long waiting times on the transplant waiting list (1). Despite the technical evolution and better understanding of LVAD, there are still drawbacks in treating patients with an LVAD. Typical complications are bleeding complications, infections, right heart failure, and the risk of thrombus formation, which can lead to a device failure with a hemodynamic instability of the patient (2).
There a several treatment options a doctor can perform for a thrombus formation. Some authors recommend a lysis therapy, and some recommend early LVAD exchange (3,4). However, there is in the literature incoherence how to treat the patient in the best way with low risk of accompanied complications, which shows the difficulty and explains that every treatment should be tailored to each patient.
Here we present a minimally invasive surgical technique to remove a confirmed thrombus formation in the outflow graft in the setting of an LVAD complication.
Case presentation
A 28 years old man was presented with shortness of breath, new onset of weakness and signs of right heart failure while he was on LVAD. Laboratory tests did not reveal any signs of hemolysis or myocardial ischemia. Initial transthoracic echocardiography did not reveal any signs of a thrombus. The device (HeartWare) parameters, such as for power consumption were in range (no increase in power). However, the pump flow dropped to 1.5 L/min. The patient was treated in the past due to a candida bloodstream infection with antifungal drugs. However, he was again detected positive in the collected blood cultures for a candida bloodstream infection. Treatment with voriconazol was started, and inflammatory parameters were low under treatment. A progressive biventricular heart failure characterized the further clinical course. Inotropic support with Milrinone 0.5 µg/kg/min was established and pump flow increased sufficiently. Subsequent transthoracic echocardiography showed a new thrombus in the outflow graft with a partial and non-obstructive clot formation, which was confirmed by a CT-angiography. The patient was taken to theatre, and a small anterolateral right-sided thoracotomy was performed. The clot could be harvested from the outflow graft without any complications. Further investigations revealed a fungal thrombus (Figure 1A,B,C,D).
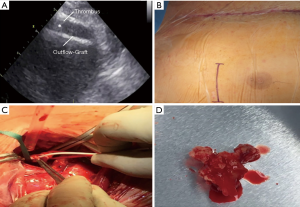
Treatment and follow up
Postoperative the pump flow rates were normalized, but the patient developed a severe vasoplegia which was treated with high vasopressor dosages and methylene blue and under treatment regressive. Three weeks after embolectomy the patient was transferred into the intermediate care unit but was weak after prolonged ICU therapy. Furthermore, the patient suffered from listlessness and was difficult to motivate for physical activity. With a BMI of 18, the patient was no candidate for heart transplant listing. The patient was subsequently readmitted to the ICU with a severe septic shock and multiorgan failure under high doses of inotropes and vasopressors and finally died.
Discussion
Despite life-saving aspects of LVAD therapy in patients with end-stage heart failure, LVAD thrombosis remains an important issue in the treatment of these patients. The diagnosis of LVAD thrombosis is complex, and many clinical considerations have to be taken into account, but LVAD malfunction is most commonly due to pump thrombosis. There are several algorithms published on how to diagnose LVAD thrombosis. Especially outflow graft obstruction or thrombosis is a poorly recognized complication. Patients presenting with common LVAD dysfunctional symptoms and LVAD low flow alarms like the patient in the presented case. A continuous or gradual decrease in flow rate can be used as a discriminator for differential diagnosis or the thrombosis origin. However, the diagnostic workup in LVAD thrombosis remains the same with transthoracic echocardiography and blood samples searching for signs of hemolysis. Finally, a CTA should be performed to complete diagnostic workup if the prior investigation reveals no signs of a thrombus. Another possibility which is described in the literature is intravascular ultrasound IVUS (5).
A fungal thrombosis of the LVAD is a rare and antifungal therapy, as well as a complete exchange or removal of the LVAD, might be the method of choice. Candida species are most commonly responsible for fungal thrombi and in the presented case the diagnosis was established based on the surgically removed obstructive mass and microbiological investigation. Prior re-occurrent candidemia was detected in the patients’ bloodstream which might be the cause of the development of the outflow graft thrombus.
Conclusions
Outflow graft thrombosis is difficult to detect and has to be taken into consideration especially in the setting of low flow signs and a continuous decrease in flow rates without pump power increase over a longer period. Candida thrombus is rare but in case of preexisting candidemia a possible explanation for the development of a fungal thrombus. A small anterolateral right-sided thoracotomy without reopening the complete chest is feasible to remove a clot from the outflow graft to establish a sufficient LVAD flow again.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Informed Consent: Written informed consent was obtained from the patient for publication of this manuscript and any accompanying images.
References
- Popov AF, Hosseini MT, Zych B, et al. Clinical experience with HeartWare left ventricular assist device in patients with end-stage heart failure. Ann Thorac Surg 2012;93:810-5. [Crossref] [PubMed]
- Sabashnikov A, Mohite PN, Simon AR, et al. HeartWare miniaturized intrapericardial ventricular assist device: advantages and adverse events in comparison to contemporary devices. Expert Rev Med Devices 2013;10:441-52. [Crossref] [PubMed]
- Scandroglio AM, Kaufmann F, Pieri M, et al. Diagnosis and Treatment Algorithm for Blood Flow Obstructions in Patients With Left Ventricular Assist Device. J Am Coll Cardiol 2016;67:2758-68. [Crossref] [PubMed]
- Luc JGY, Tchantchaleishvili V, Phan K, et al. Medical Therapy Compared With Surgical Device Exchange for Left Ventricular Assist Device Thrombosis: A Systematic Review and Meta-Analysis. ASAIO J 2018. [Epub ahead of print]. [Crossref] [PubMed]
- Muller Moran HR, Kass M, Ravandi A, et al. Diagnosis of Left Ventricular Assist Device Outflow Graft Obstruction Using Intravascular Ultrasound. Circ Heart Fail 2016.9. [PubMed]


