
Predicting the rate of oxygen consumption during the 3-minute constant-rate stair stepping and shuttle tests in people with chronic obstructive pulmonary disease
Introduction
The 3-minute constant-rate stair stepping (3-min CRSST) and constant-speed shuttle tests (3-min CSST) were developed and validated for standardized assessment of exertional breathlessness in people with chronic obstructive pulmonary disease (COPD) (1-3). That is, the 3-min CRSST and 3-min CSST require participants to stair-step or shuttle to the pace of an external audio beep, thereby standardizing the exercise stimulus and allowing the breathlessness response to the stimulus to be assessed. The 3-min CRSST and 3-min CSST have good test-retest reliability and are responsive to bronchodilator therapy in people with COPD (1,3,4). The key benefit of these tests over other tests of exertional breathlessness [e.g., cardiopulmonary exercise testing (CPET) on a cycle ergometer or treadmill] is that they are cheap and require limited resources, offering an opportunity for wide-spread use in both clinical and research settings (5). Further, unlike commonly used task-based questionnaires of breathlessness [e.g., Medical Research Council dyspnea scale (MRC) (6)] or self-paced tests of exercise tolerance (6-minute walk test), the 3-min CRSST and 3-min CSST specifically aim to quantify the breathlessness response to a direct and standardized exercise stimulus (2). This overcomes problems when the level of physical activity or walking speed during self-paced exercise tests are adjusted from one evaluation to another according to the level of breathlessness experienced (7). For example, a person with COPD who initiates effective bronchodilator therapy may engage in more physical activity, and thus, experience and report a similar (or even higher) level of breathlessness on a task-based questionnaire or at the end of a self-paced exercise test. In this situation, the benefits of bronchodilation may not be captured using common breathlessness assessment approaches, and true treatment effects may be overlooked. Assessment at a standardized level of exertion is, therefore, key for valid measurement of breathlessness and its response to therapy (5,7,8).
The ability to estimate the rate of oxygen consumption (V’O2) of the 3-min CRSST and 3-min CSST would enable healthcare providers to relate the stair stepping rate and shuttle speed that elicits a certain level of breathlessness in the clinical care setting to physical activities of daily life (e.g., household chores, gardening, leisure activities) with a similar metabolic demand (9). This could help healthcare providers conceptualize how activity-related breathlessness may impact daily life for their patients and facilitate a more personalized approach to symptom management. While the gold standard for assessment of V’O2 during these tests would be direct measurement of gas exchange with a CPET system, this is often not viable in many clinical and research settings due to resource constraints, both financial and personnel. The objectives of this study were to: (I) develop COPD-specific equations for predicting the V’O2 during the 3-min CRSST and 3-min CSST; and (II) compare the newly developed COPD-specific metabolic equations to the American College of Sports Medicine (ACSM) metabolic equations developed in the healthy general adult population (10) for predicting the V’O2 of these tests.
Methods
This was a retrospective analysis of participant data from three previous multi-centre studies conducted at the Montreal Chest Institute and the Institut Universitaire de Cardiologie et de Pneumologie de Québec by Perrault et al. (2), Sava et al. (3) and Borel et al. (1). Participants were included in each of these studies if they had at least moderate COPD according to the Global Initiative for Obstructive Lung Disease (GOLD) criteria [post-bronchodilator forced expiratory volume in one second (FEV1) to forced vital capacity (FVC) <0.7 and FEV1 ≤80% predicted (11)] and a cigarette smoking history of at least 10 pack years. Further methodological details on each of these studies are published elsewhere (1-3). The Biomedical Research Ethics Committee of both institutions approved the study protocols and informed consent was obtained from all participants.
Eligibility
Participants were included in the current study if they completed all three minutes of a 3-min CRSST and/or 3-min CSST as part of studies by Perrault et al. (2), Sava et al. (3) or Borel et al. (1). In each of these studies, several participants completed multiple trials of the same test at various stepping rates or shuttle speeds. Data from the 3-min CRSST and/or 3-min CSST completed at the highest stepping rate or fastest shuttle speed for each participant was included in the present analyses.
Measures
Participant height and body mass were obtained in accordance with anthropometric measurement standards. Participants performed routine post-bronchodilator spirometry, single-breath diffusing capacity of the lungs for carbon monoxide (DLCO) and body plethysmography testing on automated equipment according to recommended techniques (12). Pulmonary function measures were expressed in relation to recommended reference values (13-15). At the same visit, participant’s physiological and perceptual responses to symptom-limited exercise were assessed with an incremental CPET: electronically braked cycle ergometer with 10 W·min−1 increase in power output (1) or incremental shuttle walk test (ISWT) (2,3). Gas exchange parameters were collected on a breath-by-breath basis using a computerized CPET system [Vmax, Sensormedics, Carefusion (1); Jaeger Oxycon Mobile®, CareFusion (2,3)]. Peak V’O2 was taken as the average of the last 30-sec of exercise. At peak exercise, participants rated the intensity of their perceived breathlessness using the 0-10 modified Borg scale (16).
3-min constant-rate stair stepping test (3-min CRSST)
The 3-min CRSST requires participants to step up and down a 20 cm step to the pace of a constant external audio beep. Participants were instructed to step up with both feet, then down with both feet at the corresponding audio signal. Participants from Perrault et al. (2) completed testing at stepping rates of 18, 22, 26 and 32 steps·min−1; for Sava et al. (3), participants completed testing at stepping rates of 18, 22 and 26 steps·min−1. For each of these studies, testing was performed in a sequential order interspersed by 10 (2) or 30 (3) minutes of rest. For Borel et al. (1), participants performed testing at stepping rates of 14, 16, 20 and 24 steps·min−1 in a randomized order, with tests interspersed by 30 minutes of rest.
3-min constant-speed shuttle test (3-min CSST)
The 3-min CSST requires participants to walk on a flat surface to the pace of a constant external audio beep walking around two markers set 9 metres part. The marker placement allowed for a 0.5 m turning circle at each end (total course length =10 m). Participants were instructed to start walking at the first audio signal and to arrive at the marker by the next audio signal. Participants for Perrault et al. (2) performed testing at shuttle speeds of 1.5, 2.5, 4.0 and 6.0 km·hr−1 in a sequential order, with tests interspersed by 10 minutes of rest. Participants of Sava et al. (3) started the 3-min CSST at 4.0 km·hr−1. If participants completed all three minutes, they repeated the 3-min CSST at 6.0 km·hr−1 after 30-minutes of rest. If they did not complete all three minutes, they repeated the 3-min CSST at 2.5 km·hr−1 after 30 minutes of rest.
3-min CRSST and 3-min CSST physiological and perceptual responses
Participant’s V’O2 during the 3-min CRSST and/or 3-min CSST was assessed on a breath-by-breath basis with a computerized CPET system [Vmax, Sensormedics, Carefusion (1); Jaeger Oxycon Mobile®, CareFusion (1-3)]; data collected over the last 30-seconds of testing were averaged and used for the present analyses. Throughout testing, participant’s peripheral oxygen saturation (SpO2) was assessed with finger pulse oximetry. Participants rated the intensity of their perceived breathlessness at the end of each test using the 0–10 modified Borg scale (16).
American College of Sports Medicine (ACSM) metabolic equations
The V’O2 measured during the 3-min CRSST or 3-min CSST was compared to the V’O2 predicted for constant-rate stair climbing and constant-speed walking using the following ACSM metabolic equations (10):
- Stair climbing: V’O2 (mL·kg·min−1) = [0.2* step rate (steps·min−1)] + [1.33*1.8*step height (m) * step rate (steps·min−1)] + 3.5 mL·kg·min −1, or for a step height of 20 cm, V’O2 (mL·kg·min−1) = (0.6788* steps·min−1) + 3.5 mL·kg·min−1;
- Walking: V’O2 (mL·kg·min −1) = [0.1* speed (km·hr−1)] +3.5 mL·kg·min−1.
The predicted V’O2 expressed in mL·kg·min−1 was converted to L·min−1 for each participant.
Statistical analysis
Participants were described by basic demographic and health characteristics. Peak V’O2 (L·min−1) and breathlessness intensity responses to the 3-min CRSST and 3-min CSST were reported for each stepping rate/shuttle speed. Multivariate regression analyses estimated predictors of the V’O2 response to the 3-min CRSST and 3-min CSST to build COPD-specific metabolic equations. Readily available participant characteristics of body size (height, mass, body mass index (BMI)), pulmonary function (FEV1, FVC, DLCO, residual volume, total lung capacity) and self-reported breathlessness (modified MRC) were considered as predictors. Univariate associations between potential predictors and the V’O2 measured during the 3-min CRSST or 3-min CSST were examined as scatter plots to evaluate the shape of association. Associations were then assessed for statistical significance. Predictor variables that were statistically significant (alpha <0.05) were added and estimated in the model. Variables that were non-significant in the final model were removed. Next, the V’O2 response to the 3-min CRSST and 3-min CSST was estimated using the corresponding ACSM metabolic equation. The mean squared error (MSE) of the COPD-specific metabolic equations and ACSM metabolic equations for predicting V’O2 was calculated and reported for comparisons. Bland-Altman analyses evaluated the level of agreement between the measured V’O2 and the V’O2 predicted for each test using (I) the newly developed COPD-specific metabolic equation and (II) the ACSM metabolic equation. The mean difference and 95% limits of agreement (LoA) were reported for comparison. All analyses were performed in the statistical software RStudio (version 1.2.1335). Statistical significance was considered at alpha <0.05.
Results
There were 98 and 69 participants who completed the 3-min CRSST and 3-min CSST, respectively, and had data for V’O2 during these tests. Participants in each group were mostly male with severe airflow limitation, static lung hyperinflation, pulmonary gas trapping and exercise intolerance (Table 1).
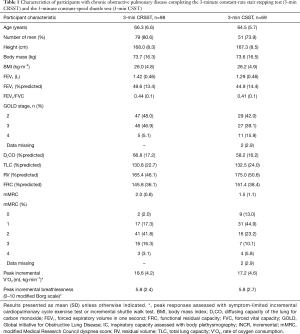
Full table
3-min constant-rate stair stepping test (3-min CRSST)
The stepping rates completed by participants are presented in Table 2; the average stepping rate completed was 24±4 steps·min−1. The V’O2 at the end of the 3-CRSST was, on average, 1.29±0.37 L·min−1, corresponding to 100%±21% of the peak V’O2 assessed during the CPET or ISWT. Breathlessness intensity ratings at the end of the 3-min CRSST were 5±2 units on the 0-10 modified Borg scale, corresponding to 91%±40% of the peak CPET or ISWT value. The average SpO2 at end exercise was 91%±4% (range, 77–99%).

Full table
Predictors of V’O2 in COPD
Stair stepping rate and participant body mass were the only significant predictors of the V’O2 (L·min−1) response over the last 30-sec of the 3-min CRSST. The COPD-specific metabolic equation developed was:
V’O2 (L·min−1) = [0.015286 * body mass (kg)] + [0.035605* step rate (steps·min−1)] –0.698449
This model estimated the V’O2 of the 3-min CRSST with a residual standard error of 0.24 L·min−1 and R2 of 0.61.
Comparison of COPD-specific and ACSM metabolic equations
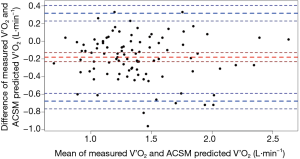
The MSE of the COPD-specific metabolic equation for predicting V’O2 of the 3-min CRSST was 0.05 L·min−1. Bland-Altman analyses revealed a mean difference between the V’O2 measured and the V’O2 predicted using the COPD-specific metabolic equation of 0.00 L·min−1 (95% LoA −0.46, 0.46) (Figure 1). There was equivalent bias between measured and predicted values with increasing V’O2.
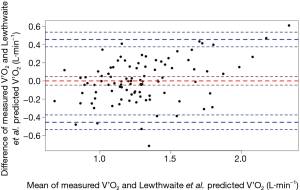
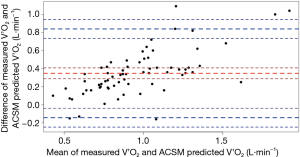
For the ACSM metabolic equation, the MSE for predicting V’O2 was 0.10 L·min−1. Bland-Altman analyses revealed a mean difference (measured-predicted) of −0.18 L·min−1 (95% LoA −0.68, 0.32) (Figure 2). That is, on average, the ACSM metabolic equation almost consistently overestimated the V’O2 of the 3-min CRSST. The bias was relatively homoscedastic (Figure 2).
3-min constant-speed shuttle test (3-min CSST)
The shuttle speeds completed by participants is presented in Table 2; the average shuttle speed completed was 4±1 km·hr−1. The V’O2 at the end of the 3-min CSST was, on average, 1.13±0.39 L·min−1, corresponding to 93%±25% of peak V’O2 assessed during the CPET or ISWT. Breathlessness intensity ratings at the end of the 3-min CSST were 4±2 units on the 0–10 modified Borg scale, corresponding to 66%±37% of the peak CPET or ISWT value. The average SpO2 at end exercise was 91%±4% (range, 72–98%).
Predictors of V’O2 in COPD
Shuttle speed and participant body mass were the only significant predictors of the V’O2 (L·min−1) response over the last 30-sec of the 3-min CSST in people with COPD. The COPD-specific metabolic equation developed was:
V’O2 (L·min−1) = [0.012039 * body mass (kg)] + [0.217654* shuttle speed (km·hr−1)] – 0.691760
This model estimated the V’O2 of the 3-min CSST with a residual standard error of 0.23 L·min−1 and R2 of 0.68.
Comparison of COPD-specific and ACSM metabolic equations
The MSE of the COPD-specific metabolic equation for predicting V’O2 of the 3-min CSST was 0.05 L·min−1. Bland-Altman analyses revealed a mean difference between the V’O2 measured and the V’O2 predicted using the COPD-specific metabolic equation of 0.00 L·min−1 (95% LoA −0.44, 0.44) (Figure 3). The bias was relatively homoscedastic.
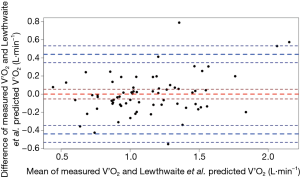
For the ACSM metabolic equation, the MSE for predicting V’O2 of the 3-min CSST was 0.18 L·min−1. Bland-Altman analyses revealed a mean difference (measured-predicted) of 0.35 L·min−1 (95% LoA −0.14, 0.84). That is, on average, the ACSM metabolic equation underestimated the V’O2 of the 3-min CSST. There was substantial heteroscedasticity, with the underestimation of V’O2 becoming more pronounced with increasing V’O2 (Figure 4).
Discussion
This study was the first to explore different methods for predicting the V’O2 of the 3-min CRSST and 3-min CSST in people with moderate to very severe COPD. The main findings of this study were:
It could be argued that the 95% LoA between the measured V’O2 and the V’O2 predicted using the newly developed COPD-specific metabolic equations for the 3-min CRSST (±0.46 L·min−1, equivalent to 36% of the average V’O2 for all trials) and 3-min CSST (±0.44 L·min−1, equivalent to 39% of the average V’O2 for all trials) are broad, limiting their clinical utility. In terms of daily activity, these LoA would equate to around 1.9 metabolic equivalents of task (MET) for a 70 kg person, where 1 MET is the V’O2 of quiet sitting, equal to 3.5 ml of O2·kg·min−1. However, the mean difference between measured and predicted V’O2 was 0.00 L·min−1 for both the 3-min CRSST and 3-min CSST, and ±1 SD (68% LoA) was ±0.23 L·min−1 (3-min CRSST) and ±0.22 L·min−1 (3-min CSST), which equate to <1 MET for a 70 kg person. There was no clear pattern with participant characteristics that could inform when the V’O2 of these tests would likely be over- or underestimated. Nevertheless, compared to the ACSM metabolic equations developed in healthy adults, the newly generated COPD-specific metabolic equations enable the V’O2 to be predicted with narrower LoA and less intensity-dependent bias, particularly during the 3-min CSST.
In our participants with moderate to very severe COPD, stair stepping during the 3-min CRSST elicited a lower V’O2 than predicted using the ACSM metabolic equation for stair climbing in healthy adults. In contrast, shuttle walking during the 3-min CSST elicited a higher V’O2 than predicted using the ACSM metabolic equation for straight line walking in healthy adults, particularly with increasing shuttle speeds. Factors that may have contributed to these differences in our sample of people with COPD include differences in the: (I) study populations (i.e., the ACSM metabolic equations were developed in a sample of healthy adults); and/or (II) specific nature of the stepping and shuttle tasks. It seems unlikely that the V’O2 during the 3-min CRSST and 3-min CSST would differ in people with COPD compared to healthy adults. Whole-body metabolic efficiency is reportedly unaffected during submaximal (17) and maximal (18) cycle exercise testing in people with vs. without COPD. Previous studies have also reported no difference in the V’O2 of daily life activities (e.g., sitting, standing, walking, stair stepping, carrying a load, dressing/undressing, self-care and household chores) between people with vs. without COPD (19,20) or between people of different COPD disease severities (21).
As for the difference in specific task type, for the 3-min CRSST, the participant is required to repeatedly step up and down a single 20 cm step. This differs quite significantly to the stepping task that the ACSM metabolic equations are intended, where the participant ascends continuously up a flight of stairs. The V’O2 of ascending stairs is around twice that of descending stairs in healthy adults (22). For the 3-min CSST, the participant is required to shuttle walk around two marker cones placed 9.5 m apart. The change in gait and deacceleration/acceleration with each turn may increase the V’O2 needed to complete this test. The ACSM metabolic equations are intended to predict the V’O2 of constant horizontal acceleration (10). Like with this study in people with COPD, people with cardiovascular disease are reported to have a higher V’O2 during shuttle walking compared to the V’O2 predicted using the ACSM metabolic equation for walking (23), and difficulty manoeuvring around the marker cones has been reported by people with cardiovascular disease as limiting shuttle test performance (24). An increase in V’O2 resulting from deacceleration/acceleration, and/or change in gait while manoeuvring around the marker cones, may also explain why the underestimation of V’O2 by the ACSM metabolic equations became more pronounced with increasing shuttle speeds. Therefore, the metabolic equations developed in the current study to predict the V’O2 of the 3-min CRSST and 3-min CSST for people with COPD may not be COPD-specific per se but rather specific to the task requirements of these particular tests. It follows that additional research is required to determine whether the metabolic equations developed in this study for people with COPD can be used to accurately predict the V’O2 of the 3-min CRSST and 3-min CSST in healthy adults or people with other malignant and non-malignant conditions.
Clinical implications
The COPD-specific metabolic equations developed in the present study increase the clinical utility of the 3-min CRSST and 3-min CSST in people with COPD. Specifically, use of the COPD-specific metabolic equations enable the 3-min CRSST stepping rate and 3-min CSST shuttle speed to be related to daily life physical activities with a similar metabolic demand. For example, the average participant of the present study was 74 kg and completed the 3-min CRSST at 24 steps·min−1 and the 3-min CSST at 4.0 km·hr−1 with a breathlessness intensity response of 4 (somewhat severe) on the 0–10 modified Borg scale. For this ‘average’ participant with COPD, the stair stepping rate of 24 steps·min−1 would equate to an estimated V’O2 of 17.4 mL·kg·min−1 (or 5 METs), which is comparable to the whole-body metabolic cost of shovelling snow (~5.3 METs) (25). The shuttle speed of 4.0 km·hr−1 would equate to an estimated V’O2 of 14.5 mL·kg·min−1 (or 4.1 METs), which is comparable to the whole-body metabolic cost of washing laundry by hand/hanging washing (4 METs) (25). It could then be interpreted that this ‘average’ participant with COPD would likely experience somewhat severe breathlessness when performing these daily physical activities in his/her real life for periods of 3-minutes or more. With this information, the healthcare provider could start explicit conversations with the patient around their expectations with breathlessness, and realistic goals could be set for the patient around performing daily life physical activities while living with breathlessness. To facilitate clinical use of the COPD-specific metabolic equations in this context, a tool is provided in Supplementary File (http://fp.amegroups.cn/cms/4a99372cea9b8d2d25b7033f88b7fcc2/jtd.2020.03.13-1.xlsx).
Strengths and limitations
This study was strengthened by the comprehensive gas exchange and symptom data available for people with COPD who performed the 3-min CRSST and 3-min CSST using a standardized protocol across three studies at two research centres in Québec, Canada. Data were available for people with COPD across a range of COPD and symptom severities, which reflects the typical COPD population managed by healthcare providers in primary and secondary care (26,27). The metabolic equations developed in this study enable the V’O2 of the 3-min CRSST and 3-min CSST to be estimated for people with COPD who perform these tests using the same standardized protocol as the current study. It is unknown whether the V’O2 of the 3-min CRSST and 3-min CSST would differ for people with other acute or chronic health conditions. To this end, the reliability, validity and responsiveness of the 3-min CRSST and 3-min CSST for assessment of breathlessness in other non-COPD conditions have not yet been explored. Finally, there was an overrepresentation of men in the present study; the effect of this sex imbalance on the COPD-specific metabolic equations is unknown.
Conclusions
The development and prospective validation of the 3-min CRSST and 3-min CSST has provided a means to effectively evaluate breathlessness and its response to therapy at a standardized level of exertion in people with COPD, without the need for high cost resources or skills (1,3,5,8,17). This study was the first to develop metabolic equations to predict the V’O2 of the 3-min CRSST and 3-min CSST for people with moderate to very severe COPD. Being able to accurately predict the V’O2 of the 3-min CRSST and 3-min CSST further extends the use of these tests in clinical practice, enabling the clinician to equate stair stepping rates/shuttle walking speeds that elicit a certain level of breathlessness to common physical activities of daily life with a similar whole-body metabolic demand.
Acknowledgments
We acknowledge the work done by those who helped collect and collate data used in the present analyses as part of earlier studies by Perrault et al. (2009), Sava et al. (2012) and Borel et al. (2016): Helene Perrault, Jacinthe Baril, Sara Henophy, Ashley Rycroft, Francesco Sava, Cynthia Brouillard, Carmen Darauay, Courtney A Wilkinson-Maitland, Marthe Bélanger, Marie-Josée Breton, Brigitte Jean, Josée Picard, Eric Nadreau, Julie Milot, Yves Lacasse, Steeve Provencher, Pierre Leblanc, Alisha Maini, Sara Abdallah.
Funding: Each of these previous studies was supported by an unrestricted and investigator-initiated grant from Boehringer Ingelheim to FM. The sponsor of these studies had no role in their design, data collection and interpretation, preparation of the manuscript or decision to submit the manuscript for publication. Similarly, Boehringer Ingelheim had no role in (i) the conception, analysis and interpretation of data of the present study or (ii) preparation of this manuscript or the decision to submit for publication. HL was supported by an Endeavour Research Fellowship by the Department of Education and Training, Australia. EMK was supported by a Canada Graduate Scholarship – Master Award (CGS-M) from the Canadian Institutes of Health Research (CIHR). FM holds a GlaxoSmithKline (GSK)/CIHR Research Chair on COPD at Université Laval. JB holds a GSK/CIHR Research Chair on Better Understanding of COPD and Intervention Guides at McGill University. DJ holds a Canada Research Chair in Clinical Exercise & Respiratory Physiology (Tier 2) from the CIHR.
Footnote
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/jtd.2020.03.13). AH is an employee of Boehringer Ingelheim (Canada) Ltd. DJ reports grants from Boehringer Ingelheim, during the conduct of the study; grants and personal fees from AstraZeneca, grants from Boehringer Ingelheim, grants from Novartis, grants from Tilray, outside the submitted work. FM reports grants from AstraZeneca and GlaxoSmithKline, Boehringer Ingelheim, GSK, Sanofi, and Novartis during the conduct of this study, and personal fees for serving on speaker bureaus and consultation panels from Boehringer Ingelheim, Grifols, and Novartis outside the submitted work. He is financially involved with Oxynov, a company which is developing an oxygen delivery system. JB reports other from GlaxoSmithKline, during the conduct of the study; grants from CIHR, grants from Canadian Respiratory Research Network (CRRN), personal fees from Canadian Thoracic Society, personal fees from CHEST, grants from Foundation of the MUHC, grants from Aerocrine, grants and personal fees from AstraZeneca, grants and personal fees from Boehringer Ingelheim, grants and personal fees from Grifols, grants and personal fees from GlaxoSmithKline, grants and personal fees from Novartis, grants and personal fees from Trudell, outside the submitted work. The other authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The Biomedical Research Ethics Committee of both institutions approved the study protocols and informed consent was obtained from all participants.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Borel B, Wilkinson-Maitland CA, Hamilton A, et al. Three-minute constant rate step test for detecting exertional dyspnea relief after bronchodilation in COPD. Int J Chron Obstruct Pulmon Dis 2016;11:2991-3000. [Crossref] [PubMed]
- Perrault H, Baril J, Henophy S, et al. Paced-walk and step tests to assess exertional dyspnea in COPD. COPD 2009;6:330-9. [Crossref] [PubMed]
- Sava F, Perrault H, Brouillard C, et al. Detecting improvements in dyspnea in COPD using a three-minute constant rate shuttle walking protocol. COPD 2012;9:395-400. [Crossref] [PubMed]
- Maltais F, Aumann JL, Kirsten AM, et al. Dual bronchodilation with tiotropium/olodaterol further reduces activity-related breathlessness versus tiotropium alone in COPD. Eur Respir J 2019;53:1802049. [Crossref] [PubMed]
- Lewthwaite H, Koch EM, Tracey L, et al. Standardized measurement of breathlessness during exercise. Curr Opin Support Palliat Care 2019;13:152-60. [Crossref] [PubMed]
- Fletcher C. Standardised questionnaire on respiratory symptoms: a statement prepared and approved by the MRC Committee on the Aetiology of Chronic Bronchitis (MRC breathlessness score). BMJ 1960;2:1665.
- Ekström M, Elmberg V, Lindow T, et al. Breathlessness measurement should be standardised for the level of exertion. Eur Respir J 2018;51:1800486. [Crossref] [PubMed]
- Ekström M. Why treatment efficacy on breathlessness in laboratory but not daily life trials? The importance of standardized exertion. Curr Opin Support Palliat Care 2019;13:179-83. [Crossref] [PubMed]
- Annegarn J, Meijer K, Passos VL, et al. Problematic activities of daily life are weakly associated with clinical characteristics in COPD. J Am Med Dir Assoc 2012;13:284-90. [Crossref] [PubMed]
- American College of Sports Medicine. ACSM's Metabolic Calculations Handbook. USA: Lippincott, Williams & Wilkins; 2007.
- From the Global Strategy for the Diagnosis, Management and Prevention of COPD. Global Initiative for Chronic Obstructive Lung Disease (GOLD). 2019 [cited 27 October 2019]. Available online: .http://www.goldcopd.org/
- Miller MR, Crapo R, Hankinson J, et al. General considerations for lung function testing. Eur Respir J 2005;26:153-61. [Crossref] [PubMed]
- Quanjer PH, Stanojevic S, Cole TJ, et al. Multi-ethnic reference values for spirometry for the 3-95-yr age range: the global lung function 2012 equations. Eur Respir J 2012;40:1324-43. [Crossref] [PubMed]
- Crapo RO, Morris AH, Clayton PD, et al. Lung volumes in healthy nonsmoking adults. Bull Eur Physiopathol Respir 1982;18:419-25. [PubMed]
- Stanojevic S, Graham BL, Cooper BG, et al. Official ERS technical standards: Global Lung Function Initiative reference values for the carbon monoxide transfer factor for Caucasians. Eur Respir J 2017;50:1700010. [Crossref] [PubMed]
- Borg GA. Psychophysical bases of perceived exertion. Med Sci Sports Exerc 1982;14:377-81. [Crossref] [PubMed]
- Perrault H, Gravel G, Ofir D, et al. Cycling efficiency is not compromised for moderate exercise in moderately severe COPD. Med Sci Sports Exerc 2007;39:918-25. [Crossref] [PubMed]
- Lewis MI, Belman MJ, Monn SA, et al. The relationship between oxygen consumption and work rate in patients with airflow obstruction. Chest 1994;106:366-72. [Crossref] [PubMed]
- Jeng C, Chang W, Wai PM, et al. Comparison of oxygen consumption in performing daily activities between patients with chronic obstructive pulmonary disease and a healthy population. Heart Lung 2003;32:121-30. [Crossref] [PubMed]
- Vaes AW, Wouters EFM, Franssen FME, et al. Task-related oxygen uptake during domestic activities of daily life in patients with COPD and healthy elderly subjects. Chest 2011;140:970-9. [Crossref] [PubMed]
- Castro AA, Porto EF, Iamonti VC, et al. Oxygen and ventilatory output during several activities of daily living performed by COPD patients stratified according to disease severity. PLoS One 2013;8:e79727. [Crossref] [PubMed]
- Teh KC, Aziz AR. Heart rate, oxygen uptake, and energy cost of ascending and descending the stairs. Med Sci Sports Exerc 2002;34:695-9. [PubMed]
- Almodhy M, Beneke R, Cardoso F, et al. Pilot investigation of the oxygen demands and metabolic cost of incremental shuttle walking and treadmill walking in patients with cardiovascular disease. BMJ Open 2014;4:e005216. [Crossref] [PubMed]
- Woolf-May K, Meadows S. Exploring adaptations to the modified shuttle walking test. BMJ Open 2013;3:e002821. [Crossref] [PubMed]
- Ainsworth BE, Haskell WL, Herrmann SD, et al. 2011 Compendium of Physical Activities: a second update of codes and MET values. Med Sci Sports Exerc 2011;43:1575-81. [Crossref] [PubMed]
- Bednarek M, Maciejewski J, Wozniak M, et al. Prevalence, severity and underdiagnosis of COPD in the primary care setting. Thorax 2008;63:402-7. [Crossref] [PubMed]
- Jones PW, Nadeau G, Small M, et al. Characteristics of a COPD population categorised using the GOLD framework by health status and exacerbations. Respir Med 2014;108:129-35. [Crossref] [PubMed]


