Sub-solid lung adenocarcinoma in Asian versus Caucasian patients: different biology but similar outcomes
Introduction
Lung cancer is the most commonly diagnosed cancer and the most common cause of cancer-related death in the United States (1) and worldwide (2). Adenocarcinoma is the most common histologic type (3). Asian and Caucasian patients with lung cancer have been compared in several studies, with conflicting findings regarding survival. Several cancer registry studies have shown that Asian American patients have better overall survival compared to Caucasian American patients, in both early-stage (4,5) and advanced (5) non-small cell lung cancer (NSCLC). However, one study of never smoking women showed that Asian American patients had worse survival compared to Caucasian American patients (6).
None of these prior studies included proportion of ground-glass opacity as a factor in their analyses, and proportion of ground-glass has been associated with both race and survival (7,8). Among patients with lung cancer, Asians are more likely than Caucasians to be never smokers and to develop sub-solid tumors that harbor EGFR mutations (9). Sub-solid tumors, with varying proportion of ground-glass opacity, are being found more frequently, either incidentally on CT scans obtained for other reasons or on low-dose screening CT scans (10). Patients with these sub-solid lung adenocarcinomas have better overall survival than those with pure solid tumors (7,11,12). Many groups have studied how to monitor these sub-solid lesions (13,14), and many are advocating for sublobar resection of these tumors given their lower likelihood of being associated with lymph node metastases and their better prognosis (15).
A reliable study of differences in survival between Asian and Caucasian patients with lung adenocarcinoma must include the proportion of ground-glass opacity as a confounding variable, and this variable is unavailable in nearly all databases and cancer registries. We therefore set out to use our more granular institutional data, which includes information on proportion of ground-glass opacity, to determine whether Asians and Caucasians with sub-solid nodules indeed have different biology, and whether that biology results in differences in survival.
We hypothesized that Asian patients would have better overall and recurrence-free survival compared to Caucasian patients, which might make them better candidates for sublobar resection. We based this hypothesis on prior data that EGFR mutations are predominant in Asian patients with sub-solid tumors and that KRAS mutations are predominant in Caucasian patients with sub-solid tumors, and our belief that this difference would confer a survival advantage for Asian patients. Our institutional review board approved this retrospective study under protocol 21285 and waived the requirement for informed consent.
Methods
Patients
We identified our study cohort from patients who underwent surgical resection for lung cancer of any stage at Stanford University Medical Center from April 2002 to June 2015. A prospective database including all thoracic surgery patients was started in 2008. Patients before 2008 were identified retrospectively.
We excluded patients who were not Asian or Caucasian. Race was self-reported, and Asian race was defined to include patients having origins in the Far East, Southeast Asia, or the Indian subcontinent. We excluded patients who had undergone previous surgery for lung cancer, as well as patients with tumor histology other than adenocarcinoma and tumors that were not sub-solid. Sub-solid was defined as ≥10% ground-glass opacity on preoperative CT scan or ≥10% lepidic or in-situ component on surgical pathology. Percentage of ground-glass opacity was estimated by retrospective review by either a radiologist or thoracic surgeon. Percentage of adenocarcinoma subtypes has been routinely quantified by our pathologists since 2012, and tumors resected prior to 2012 were retrospectively re-reviewed for this purpose.
Clinical data were obtained through review of electronic medical records. Staging was reclassified using the American Joint Committee on Cancer eighth edition (16). Overall and recurrence-free survival were calculated from the date of surgery. Information on patients with recurrences were reviewed in detail, and new lung nodules considered to be separate primary tumors were not categorized as recurrences. The Stanford University institutional review board approved the study.
Statistical analysis
Patient baseline characteristics and outcomes were summarized and compared by race. Continuous variables were described using mean with standard deviation, or median with interquartile range (IQR) (25th and 75th percentiles). They were compared using the Student’s t test for mean differences and Wilcoxon Rank Sum test for median or rank differences. Categorical variables were compared using the chi-square test. Kaplan-Meier curves were created to visually depict the overall and recurrence-free survival by race and mutation status, and were compared by the log-rank test. Univariable and multivariable Cox proportional hazards models were performed in time-to-event survival analysis. Race and proportion of ground-glass opacity were included a priori in multivariable models. Age, sex, smoking status, stage, grade, mutation status, surgical type, and adjuvant therapy were included in the multivariable model if the P value of unadjusted effect was less than 0.3 in the univariable analysis and the P value of the adjusted effect kept significant or nearly significant (P value <0.15) in the multivariable model after adjustment of multiple covariates. For all statistical analyses, a two-sided α-level of 0.05 was considered statistically significant. Analyses were performed using SAS version 9.4 (SAS Institute Inc. NC, USA).
Results
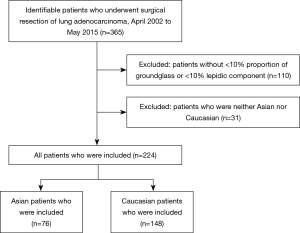
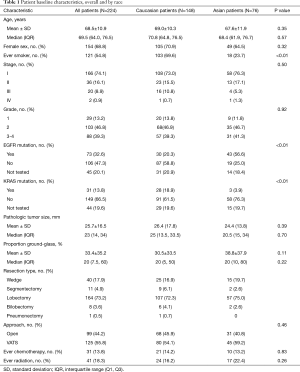
The cohort included 365 patients who underwent surgical resection of lung adenocarcinoma from April 2002 to May 2015 (Figure 1). Of these, 224 patients had sub-solid lesions that met our inclusion criteria, including 76 (33.9%) Asian and 148 (66.1%) Caucasian patients (Table 1). Both Asian and Caucasian patients were more likely to be women (64.5% and 70.9%). Asian patients were much more likely to be never smokers than Caucasian patients (76.3% vs. 29.0%, P<0.01). Asian patients were more likely to have tumors with an EGFR mutation (69.4% vs. 25.6% of those tested, P<0.01), while Caucasian patients were more likely to have tumors with a KRAS mutation (23.5% vs. 4.9% of those tested, P<0.01). Asian patients had a trend towards higher mean proportion of ground-glass opacity on preoperative CT scan (38.8% vs. 30.5%, P=0.11).
Full table
Median follow up time was 48.4 months (IQR, 32.3–72.6) (Table 2). Fifty patients had a recurrence, including 19 (25.0%) Asian patients and 31 (20.9%) Caucasian patients. Both Asian and Caucasian patients usually recurred distantly (83.3% vs. 93.5% of patients who recurred, P=0.36). Fifty patients died, including 15 (19.7%) Asian patients and 35 (23.7%) Caucasian patients.
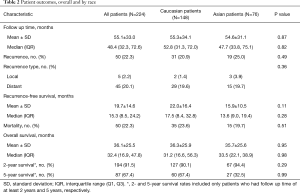
Full table
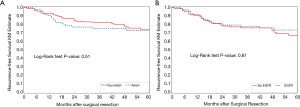
For recurrence-free survival, Kaplan-Meier curves showed no significant difference by race (Figure 2A) or EGFR mutation status (Figure 2B). Univariable analysis showed that female sex, smaller proportion of ground-glass opacity, higher grade, and higher stage were all associated with recurrence (Table 3). On multivariable analysis, female sex, smaller proportion of ground-glass opacity, and higher stage were still associated with recurrence. Higher proportion of ground-glass opacity was associated with a lower risk of recurrence (HR 0.76; 95% CI, 0.61–0.96; P=0.02 per 20% increase in proportion of ground-glass opacity).
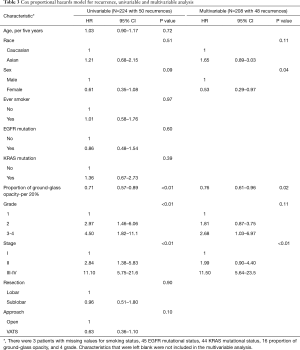
Full table
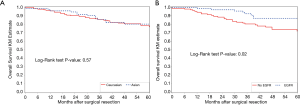
For overall survival, Kaplan-Meier curves again showed no significant difference by race (Figure 3A), but there was improved survival among patients with an EGFR mutation (P=0.02) (Figure 3B). Univariable analysis showed that smaller proportion of ground-glass opacity, higher grade, higher stage, sublobar resection, and open surgical approach were all associated with higher mortality (Table 4). However, only higher stage was significantly associated with worse overall survival on multivariate analysis. EGFR mutational status was not significant, although there remained a trend towards improved survival for patients with EGFR mutations (HR 0.48; 95% CI, 0.19–1.24; P=0.13).
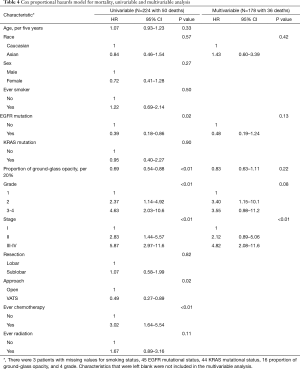
Full table
Discussion
Our findings confirm, in a granular, single-institution series that the biology of sub-solid lung adenocarcinoma is very different between Asians and Caucasians. Among patients who underwent surgical resection of sub-solid lung adenocarcinoma at our institution, Asian patients were much more likely to have EGFR mutations, whereas Caucasian patients were more likely to have KRAS mutations. Asian patients were more likely to be never smokers and had a trend toward tumors with a higher proportion of ground-glass opacity. However, contrary to our hypothesis, race was not associated with recurrence-free or overall survival.
Several previous studies have shown that Asian patients are more likely to be never smokers (9,17) and to express EGFR mutations (18,19), while Caucasian patients are more likely to express KRAS mutations (20). Sub-solid tumors have been extensively studied in Asia (13,14), as well as at our institution, where our population includes a large proportion of Asian- American patients, as well as never and light smokers (7,21-23).
However, it has been unclear whether overall survival differs between Asian and Caucasian patients with NSCLC. Most cancer registry studies have shown that Asians have better overall survival, when all histologic types are included. Ou et al. studied 19,702 patients with stage I NSCLC in the California Cancer Registry from 1989 to 2003 (4). Zell et al. studied 27,435 cases of stage IIIB and IV NSCLC in the United States Surveillance, Epidemiology, and End Results (SEER) database from 1999 to 2003 (5). In both of these studies, multivariable analysis showed that Asian patients had improved overall survival compared to Caucasian patients.
Ahn et al. compared 4,622 Korean patients with NSCLC (all histologic types) in the Samsung Medical Center Cancer Registry in Seoul, South Korea and 8,846 Caucasian American patients with NSCLC in the CSPOC/SANDIOCC databases in California, United States (24). Multivariable analysis showed that Korean ethnicity was significantly associated with improved overall survival. Younger age, female sex, and bronchioloalveolar carcinoma were also favorable factors.
Only one registry study included smoking status in their analysis. Ou et al. studied 20,140 patients with NSCLC and known smoking status from 1991 to 2005 in the Cancer Surveillance Program of Orange County and the San Diego/Imperial Organization for Cancer Control (CSPOC/SANDIOCC), which collected data from those three southern California counties before the California Cancer Registry took over collection in 2008 (25). Multivariable analysis showed that Asian ethnicity was independently associated with improved overall survival compared to all other ethnicities. Younger age, female sex, and bronchioloalveolar carcinoma were all independently associated with improved overall survival. Smoking status was not significant.
There was one study in northern California that showed conflicting results, that Asian Americans had worse overall survival compared to Caucasian Americans. Gomez et al. studied female never smokers with NSCLC in the San Francisco Bay Area Lung Cancer Study (SFBALCS) (6). At the time, population estimates showed that almost 90% of Asian and Pacific Islander (API) women and 56% of non-Hispanic white women were never smokers. Among the screened patients with NSCLC, about 70% of API women and 10% of non-Hispanic White women were never smokers. Multivariable analysis showed that among never smokers with NSCLC, US-born API women had higher overall mortality compared to non-Hispanic white women (HR 1.69; 95% CI, 1.01–2.75).
Proportion of ground-glass opacity is an important predictor of survival that is not included in any of these databases. At our institution, we previously studied patients with cN0 lung adenocarcinoma who underwent surgical resection and found that patients with even a minor (1–25%) ground-glass component had improved overall survival compared to those with pure solid lesions (7). Hattori et al. showed that patients with clinical stage IA NSCLC with sub-solid tumors [consolidation tumor ratio (CTR), 0.50–0.99] had improved five-year overall survival compared to those with pure-solid tumors (CTR, 1.0) (12). Finally, the current lung adenocarcinoma classification system includes a predominate lepidic component as an important prognostic feature (26).
In this study, which focused more narrowly on sub-solid tumors and included proportion of ground-glass opacity in the analysis, there was no difference in survival between Asian and Caucasian American patients. An advantage of our Northern California population, which includes many people of Asian origin and also many non-smokers, is that we were able to include a sizeable number of both Asian and Caucasian patients with sub-solid tumors in our cohort. With single-institution, granular data, we were able to include mutation status and proportion of ground-glass opacity in our analysis—all known to affect survival but unavailable in most database studies.
However, our study does also have several limitations. It is retrospective and includes only a single institution. All Asian patients were grouped together, even though this group is ethnically diverse and cancer incidence and mortality may vary among subgroups (27,28). The majority of patients underwent anatomic lung resection, but we do not know the selection criteria for sublobar or wedge resection. In addition, mutational testing became routine only a few years ago, so not all tumors were tested (29).
Conclusions
Several previous studies that suggested that Asian Americans with NSCLC have improved overall survival compared to Caucasian Americans did not include smoking status, proportion of ground-glass opacity, or mutation status in their analysis. Our study showed that among patients with sub-solid lung adenocarcinoma, Asian patients were more likely to be never smokers and have the EGFR mutation, and Caucasian patients were more likely to have the KRAS mutation. There was a trend towards Asian patients having a higher proportion of ground-glass opacity. However, on multivariable analysis, there was no significant difference in overall or recurrence-free survival.
Acknowledgments
Funding: None.
Footnote
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/jtd.2020.04.37). JBS reports grants and personal fees from Becton-Dickinson, outside the submitted work; In addition, JBS has a patent CRISPR/Cas-mediated Genome Editing to Treat EGFR-mutant Lung Cancer issued. NSL reports other from Auspex, outside the submitted work and serves as an unpaid editorial board member of Journal of Thoracic Disease from Sep 2019 to Aug 2021. MFB serves as an unpaid editorial board member of Journal of Thoracic Disease from Sep 2018 to Aug 2020. The other authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. Our institutional review board approved this retrospective study under protocol 21285 and waived the requirement for informed consent.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Cronin KA, Lake AJ, Scott S, et al. Annual Report to the Nation on the Status of Cancer, part I: National cancer statistics. Cancer 2018;124:2785-800. [Crossref] [PubMed]
- Bray F, Ferlay J, Soerjomataram I, et al. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 2018;68:394-424. [Crossref] [PubMed]
- Alberg AJ, Brock MV, Ford JG, et al. Epidemiology of lung cancer: Diagnosis and management of lung cancer, 3rd ed: American College of Chest Physicians evidence-based clinical practice guidelines. Chest 2013;143:e1S-e29S.
- Ou SH, Zell JA, Ziogas A, et al. Prognostic factors for survival of stage I nonsmall cell lung cancer patients: a population-based analysis of 19,702 stage I patients in the California Cancer Registry from 1989 to 2003. Cancer 2007;110:1532-41. [Crossref] [PubMed]
- Zell JA, Ou SH, Ziogas A, et al. Survival improvements for advanced stage nonbronchioloalveolar carcinoma-type nonsmall cell lung cancer cases with ipsilateral intrapulmonary nodules. Cancer 2008;112:136-43. [Crossref] [PubMed]
- Gomez SL, Chang ET, Shema SJ, et al. Survival following non-small cell lung cancer among Asian/Pacific Islander, Latina, and Non-Hispanic white women who have never smoked. Cancer Epidemiol Biomarkers Prev 2011;20:545-54. [Crossref] [PubMed]
- Berry MF, Gao R, Kunder CA, et al. Presence of Even a Small Ground-Glass Component in Lung Adenocarcinoma Predicts Better Survival. Clin Lung Cancer 2018;19:e47-e51. [Crossref] [PubMed]
- Moon Y, Lee KY, Park JK. The prognosis of invasive adenocarcinoma presenting as ground-glass opacity on chest computed tomography after sublobar resection. J Thorac Dis 2017;9:3782-92. [Crossref] [PubMed]
- Zhou F, Zhou C. Lung cancer in never smokers-the East Asian experience. Transl Lung Cancer Res 2018;7:450-63. [Crossref] [PubMed]
- Migliore M, Fornito M, Palazzolo M, et al. Ground glass opacities management in the lung cancer screening era. Ann Transl Med 2018;6:90. [Crossref] [PubMed]
- Suzuki K, Koike T, Asakawa T, et al. A prospective radiological study of thin-section computed tomography to predict pathological noninvasiveness in peripheral clinical IA lung cancer (Japan Clinical Oncology Group 0201). J Thorac Oncol 2011;6:751-6. [Crossref] [PubMed]
- Hattori A, Matsunaga T, Takamochi K, et al. Importance of Ground Glass Opacity Component in Clinical Stage IA Radiologic Invasive Lung Cancer. Ann Thorac Surg 2017;104:313-20. [Crossref] [PubMed]
- Kakinuma R, Noguchi M, Ashizawa K, et al. Natural History of Pulmonary Subsolid Nodules: A Prospective Multicenter Study. J Thorac Oncol 2016;11:1012-28. [Crossref] [PubMed]
- Kobayashi Y, Sakao Y, Deshpande GA, et al. The association between baseline clinical-radiological characteristics and growth of pulmonary nodules with ground-glass opacity. Lung Cancer 2014;83:61-6. [Crossref] [PubMed]
- Katsumata S, Aokage K, Nakasone S, et al. Radiologic Criteria in Predicting Pathologic Less Invasive Lung Cancer According to TNM 8th Edition. Clin Lung Cancer 2019;20:e163-70.
- Detterbeck FC, Boffa DJ, Kim AW, et al. The Eighth Edition Lung Cancer Stage Classification. Chest 2017;151:193-203.
- Zhou W, Christiani DC. East meets West: ethnic differences in epidemiology and clinical behaviors of lung cancer between East Asians and Caucasians. Chin J Cancer 2011;30:287-92. [Crossref] [PubMed]
- Paez JG, Janne PA, Lee JC, et al. EGFR mutations in lung cancer: correlation with clinical response to gefitinib therapy. Science 2004;304:1497-500. [Crossref] [PubMed]
- Jiang H. Overview of gefitinib in non-small cell lung cancer: an Asian perspective. Jpn J Clin Oncol 2009;39:137-50. [Crossref] [PubMed]
- Suda K, Tomizawa K, Mitsudomi T. Biological and clinical significance of KRAS mutations in lung cancer: an oncogenic driver that contrasts with EGFR mutation. Cancer Metastasis Rev 2010;29:49-60. [Crossref] [PubMed]
- Gu B, Burt BM, Merritt RE, et al. A dominant adenocarcinoma with multifocal ground glass lesions does not behave as advanced disease. Ann Thorac Surg 2013;96:411-8. [Crossref] [PubMed]
- Gao RW, Berry MF, Kunder CA, et al. Survival and risk factors for progression after resection of the dominant tumor in multifocal, lepidic-type pulmonary adenocarcinoma. J Thorac Cardiovasc Surg 2017;154:2092-9.e2. [Crossref] [PubMed]
- Burt BM, Leung AN, Yanagawa M, et al. Diameter of Solid Tumor Component Alone Should be Used to Establish T Stage in Lung Adenocarcinoma. Ann Surg Oncol 2015;22 Suppl 3:S1318-23. [Crossref] [PubMed]
- Ahn MJ, Lee J, Park YH, et al. Korean ethnicity as compared with white ethnicity is an independent favorable prognostic factor for overall survival in non-small cell lung cancer before and after the oral epidermal growth factor receptor tyrosine kinase inhibitor era. J Thorac Oncol 2010;5:1185-96. [Crossref] [PubMed]
- Ou SH, Ziogas A, Zell JA. Asian ethnicity is a favorable prognostic factor for overall survival in non-small cell lung cancer (NSCLC) and is independent of smoking status. J Thorac Oncol 2009;4:1083-93. [Crossref] [PubMed]
- Travis WD, Brambilla E, Noguchi M, et al. International Association for the Study of Lung Cancer/American Thoracic Society/European Respiratory Society: international multidisciplinary classification of lung adenocarcinoma: executive summary. Proc Am Thorac Soc 2011;8:381-5. [Crossref] [PubMed]
- Gomez SL, Noone AM, Lichtensztajn DY, et al. Cancer incidence trends among Asian American populations in the United States, 1990-2008. J Natl Cancer Inst 2013;105:1096-110. [Crossref] [PubMed]
- Kwong SL, Chen MS Jr, Snipes KP, et al. Asian subgroups and cancer incidence and mortality rates in California. Cancer 2005;104:2975-81. [Crossref] [PubMed]
- Lindeman NI, Cagle PT, Aisner DL, et al. Updated Molecular Testing Guideline for the Selection of Lung Cancer Patients for Treatment With Targeted Tyrosine Kinase Inhibitors: Guideline From the College of American Pathologists, the International Association for the Study of Lung Cancer, and the Association for Molecular Pathology. J Mol Diagn 2018;20:129-59. [Crossref] [PubMed]