|
Original Article
Results of chest wall resection and reconstruction in 162 patients with benign and malignant chest wall disease
Manoucheher Aghajanzadeh1, Ali Alavy1, Mehrdad Taskindost2, Zahra Pourrasouly2, Gilda Aghajanzadeh2, Sara massahnia3
1Department of thoracic surgery & Pulmonology, Respiratory Diseases & TB Research Center of Guilan University of Medical Science (GUMS), Razi Hospital, Rasht, Iran; 2Department of general surgery, Respiratory Diseases & TB Research Center of Guilan University of Medical Science (GUMS), Razi Hospital, Rasht, Iran; 3Respiratory Diseases & TB Research Center of Guilan University of Medical Science (GUMS), Razi Hospital, Rasht, Iran
Corresponding to: Manoucheher Aghajanzadeh, MD, Department of thoracic surgery, Respiratory Diseases & TB Research Center of Guilan University of Medical Science (GUMS), Razi Hospital, Rasht 999067, sardar gangle ave, Iran. Tel: +98-1315550028; Fax: +98-1315530169. Email: maghajanzadeh2003@yahoo.com.
|
|
Abstract
Background: Chest wall resection is a complicated treatment modality with significant morbidity. The purpose of
this study is to report our experience with chest wall resections and reconstructions.
Methods: The records of all patients undergoing chest wall resection and reconstruction were reviewed. Diagnostic
procedures, surgical indications, the location and size of the chest wall defect, performance of lung resection, the
type of prosthesis, and postoperative complications were recorded.
Results: From 1997 to 2008, 162 patients underwent chest wall resection.113 (70%) of patients were male. Age of
patients was 14 to 69 years. The most common indications for surgery were primary chest wall tumors. The most
common localized chest wall mass has been seen in the anterior chest wall. Sternal resection was required in 22
patients, Lung resection in 15 patients, Rigid prosthetic reconstruction has been used in 20 patients and nonrigid prolene
mesh and Marlex mesh in 40 patients. Mean intensive care unit stay was 8 days. In-hospital mortality was 3.7
% (six patients).
Conclusion: Chest wall resection and reconstruction with Bone cement sandwich with mesh can be performed as a
safe and effective surgical procedure for major chest wall defects and respiratory failure is lower in prosthetic reconstruction
patients than previously reported ( 6).
Key words
chest wall tumor; chest wall resection; chest wall disease
J Thorac Dis 2010;2:81-85. DOI: 10.3978/j.issn.2072-1439.2010.02.02.005
|
|
Introduction
Early attempts for chest wall resection were limited
by availability of suitable materials for reconstruction.
Initial materials used for chest reconstruction included
autogenous tissue such as fascia lata grafts, rib grafts, or
large cutaneous grafts ( 1, 2). Since the first known chest
wall resection in the18th century, improvements in surgical
technique and anesthesia, critical care units, antibiotics,
and the development and refinements in reconstraction
techniques have allowed extensive chest wall resection
to be performed with acceptable morbidity and mortality
( 3). After radical en bloc chest wall resection, skeletal reconstraction when appropriate and adequate skin
coverage to preserve the reconstraction are the essential
elements for successful management of the chest wall
defects. In cases of chest wall tumors wide excision and
reconstraction is very important because distant metastasis
and local recurrence in incomplete resection is high
leading to poor survival. Since 1980s, the use of prosthetic
materials including polytetrafluoroethylene, polypropylene
mesh, and polypropylene mesh–methylmethacrylate
composites combined with the use of myocutaneous flaps
has enabled successful reconstruction of even the largest
chest wall defects ( 3). Although primary closure of muscle
and skin after chest wall resection is attainable in most
cases, many patients commonly require more sophisticated
reconstructive soft-tissue and skin coverage. A variety
of techniques including pedicled muscle transposition,
free muscle flaps, and omental flaps have been used to
provide adequate wound coverage, it allows quick healing,
early rehabilitation, and better cosmesis ( 4). Respiratory
complications continue to be the most frequent and are
reported in 20% to 24% of the patients ( 3). The Purpose of
this study is to retrospectively review our experience with chest wall resections and reconstruction and determine the
postoperative complications
|
|
Patients and methods
It is a retrospective study performed on the available
charts of 162 consecutive patients who underwent chest
wall resection and reconstruction at Guilan University of
Medical Sience Razi Hospital between1997 and 2008. All
patients with more than two rib resections were included
in the present series. Mean age was 40 years (range 20 to
70); 114 (70%) were men and 48 (30%) women. Prior to
chest wall resections, all patients underwent pulmonary
function tests. All patients received conventional chest
roentgenography, which occasionally detects a defect or
mass. For patients with a mass, a computed tomography
(CT) scan or MRI of the chest was done to evaluate the
extentension of the lesion and a tissue diagnosis
utilizing
fine or core needle aspiration or incisional biopsy was
attempted. In patients with suspected distant metastases,
CAT Scans were used. Patients’ charts were retrospectively
reviewed for age, sex, anatomic defect during the surgical
resection, the number of ribs or the portion of sternum
resected, and the surgical reconstruction technique. The
in-hospital outcomes as morbidity and mortality, length of
hospital stay (in the intensive care unit, and postoperative,
hospital stay) was reviewed. For the infected chest wall
requiring resection Vicry mesh was used in addition to
muscle flap coverage. In Some instances where chest wall
resection was done for radionecrosis only muscle flaps
were used.
|
|
Results
During 11 years duration, 162 patients underwent chest
wall resection .113 (70%) of patients were male. Age of
patients was 14 to 69 years. (Median age of patients was
40 years). The most common indications for surgery were
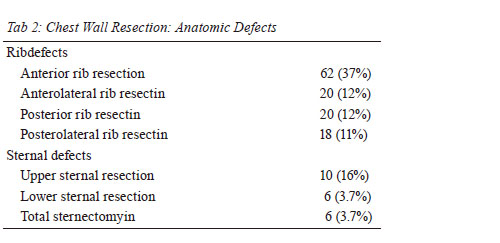
primary chest wall tumors (120patients, 74%) ( Table 1).
Ten patients (6%) underwent concomitant lung surgery and
chest wall resection. The median number of ribs resected
was 3 (range, 2 to 6). The anterior and lateral ribs were the
most commonly resected ribs (62 patients,
37%; A total of
22 patients underwent sternal resection; the most common
was an Upper sternal resection in 10 patients. Six patients
(3.7%) required total sternectomy for chondrosarcoma
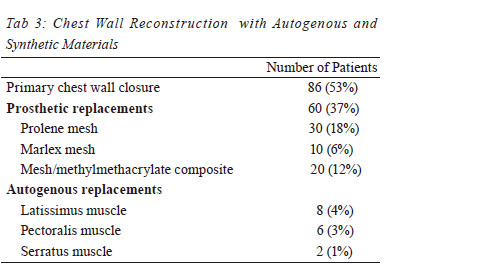
( Table 2). Immediate closure and repair was performed
in all of the patients. Primary repair of the soft tissue and
skin was performed in 86 patients (53%) and synthetic
materials were used for chest wall integrity reconstruction
in 76 patients (Prolene mesh, Marlex mesh, Bone cement
sandwich with mesh). Sixtheen patients (8%) underwent pedicled muscle flap transposition. The three most common
muscle groups utilized were latissimus flap, pectoralis flap
and Latismos dorsi ( Table 3). Bone and soft tissue sarcoma
was the most commonly foung histological chest was
tumor. The overall length of stay in the intensive care unit
in rigid reconstraction patients was 5 to 16 days (median 8
days) and for non-rigid patients was 3 to 8 (median 5 days)
and the postoperative in hospital length of stay in rigid
reconstraction patients was 16 days (range 6 to 42). And for
non-rigid patients was 6 to 10 (median 7 days). Six of 162 patients (3.7%) died during their hospital stay
due to multiplesystem organ failure, including 2 of the 18
patients who underwent rigid prosthetic reconstruction
and 4 of the patients undergoing concomitant lung surgery
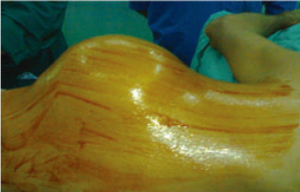
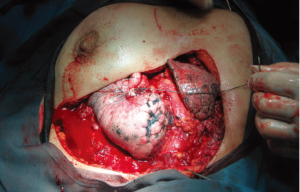
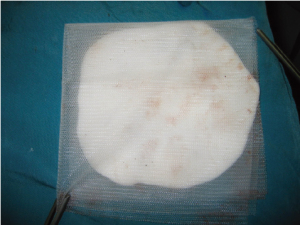
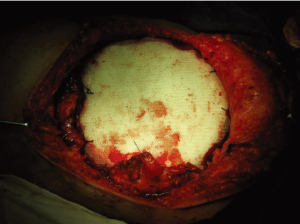
and chest wall resection. We have shown a successful
repair of a patient with lung cancer and chest wall invasion
requiring en bloc removal of the entire forequarter, the
third to eight ribs, and the involved chest wall. The defect was repaired with a double fold of prolen mesh and Bone
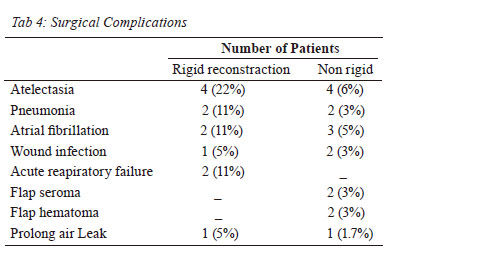
cement sandwich (Fig. 1, 2, 3, 4). Twenty-Eight of 76
patients (36 %) had complications during their hospital
stay ( Table 4). The most common complications were
atelectasis (8 patients, 10%), Atrial fibrillation (5 patients,
6 %) and Pneumonia (4 patients, 5 %). Factors associated with postoperative complications were type of prosthesis,
the location of the lesion, the need for sternal resection,
medical comorbidities, and lung resection and size of the
chest wall defect.

|
|
Discussion
In the treatment of patients requiring chest wall
resection, three tenets of surgical resection should be
main Tained ( 5). First, a sufficient amount of tissue must
be resected to dispose of all devitalized tissue. Second,
in segments of large chest wall resections a replacement
must be found to restore the rigid chest wall to prevent
physiologic flail. Third, healthy soft – tissue coverage is
essential to seal the pleural space, to protect the viscera
and great vessels, and to prevent infection. Although there
is controversy as to which chest wall lesions should be
reconstructed but, generally, lesions less than 5 cm in size
in any location, and those up to 10 cm in size posteriorly do not need reconstruction for functional reasons ( 3). Posterior
defects in proximity to the tip of the scapula, larger lesions
likely to produce paradoxical chest wall motion and most
anterior defects require reconstruction ( 6). A basic tenet prior to the initiation of chest wall
reconstruction is an appropriate and thorough chest wall
resection that leaves healthy, viable margins to which
materials and tissues used in a reconstruction may be
anchored securely ( 4). For patients in whom combined pulmonary and chest
wall resection may be required we agree with Pairolero,
that if the mediastinal lymph nodes are not positive, an
en bloc resection is warranted as the 5-year mortality is
more associated with the extent of the pulmonary cancer
than with the extent of chest wall resection ( 7). In contrast
Magdeleinat and colleagues do not consider N2 disease
a contraindication to en bloc resection and have recently
reported an actuarial 5-year survival after complete en
bloc resection of lung cancer invading the chest wall
( 8). Chape and colleagues found that only histologic
differentiation (well versus poorly differentiated) and the
depth of chest wall invasion (parietal pleura versus other)
were independent predictors of long-term survival in
multivariate analyses ( 9). Persisting or recurring chest wall involvement with
breast carcinoma after local excision and radiation therapy
may require chest wall resection to achieve local control
( 10, 11). Chest wall recurrences were found in 1% to 2% of
stage I and in 10% to 12% of stage II breast carcinomas
surgically treated with an extremely variable disease-free
interval ( 11). In current series we have 4 cases of nonhealing
radiation-induced ulcers who underwent chest wall
resection and reconstruction. With modern surgical technique a wide range of
reconstructive options are at the surgeon's disposal and
hence it is imperative that the appropriate procedure be
selected in a given patient. For small defects (less than 5
cm) or those located posteriorly under the scapula above the
fourth rib (after resection of Pancoast tumors) the skeletal
component can be ignored and the defect is uausally closed
with only soft tissue. For patients undergoing large chest
wall defects or pulmonary collapse, stabilization of the
chest wall defect may be indicated. LeRoux and Shama
have set forth the ideal characteristics of a prosthetic
material ( 12): A. rigidity to abolish paradoxical chest
motion, B. inertness allow in-growth of fibrous tissue and
decrease the likelihood of infection, c. malleability so that
it can be fashioned to the appropriate shape at the time of
operation, D. radiolucency to allow radiographic follow-up
of the underlying problem ( Fig. 5). Historically, bone, cartilage, metal sheets, superstructures
with autogenous rib graft, fascia lata, Teflon, and numerous other substances were used with minimal
success ( 13). In cases where structural integrity is necessary for
preventing chest wall collapse, methyl methacrylate
sandwich, silicone, Tef lon, or acrylic materials have
been utilized ( 5). The use of a rigid prosthesis using a
PPM-methylmethacrylate “sandwich” was developed by
some of the authors and has since been widely adopted
( 1, 3, 14, 15). It is our practice to use this rigid prosthesis for
those defects likely to produce a flail segment, including
large anterior or lateral lesions, or the area of the sternum
in which some protection of the underlying organs is
essential. This technique provides rigid repair that can
be tailored to any size, shape, or contour of chest wall
defect. The present report represents a good experience
with the use of this method of reconstruction. While the
importance of rigidity in chest wall reconstruction is
still unclear, observations of chest wall trauma provide
the significance of paradoxic motion of the chest wall.
However, this respiratory uncoordinated motion is seen
in almost every major resection of the chest wall but it
is not associated with pulmonary insufficiency, which is
seen with its traumatic counterpart, flail chest. We have
commonly used Bone cement sandwich with prolene mesh
with excellent physiologic and aesthetic success. Reports
of transposition of the latissimus dorsi muscle for chest
wall coverage had been described in 1896 by Tansini
( 18). The numerous advances in chest reconstruction over
the years with the introduction of muscle and musculocutaneous
flaps have made them the mainstay in chest wall
reconstruction ( 19). The thoracic trunk is well suited for
vascularized coverage given the many local muscle flaps
(eg, latissimus dorsi, pectoralis major, rectus abdominis,
trapezius, or deltoid muscles) or greater omentum (used
alone or in combination as potions for wound coverage) ( 20). Respiratory complications including pneumonia, acute
respiratory distress syndrome, and atelectasis are by far the
most common and have been reported to be as high as 24%
( 4). Wound complications such as infection, dehiscence,
flap loss, and hematoma are reported to occur in 8% to
20% ( 3, 4, 14). Our experience demonstrates that respiratory
complications occur red in 18% of 76 patients and
included pneumonia, atelectasis, respiratory failure (adult
respiratory distress syndrome) and Prolong air leak. Tthis
frequency of complications is far less than in other series
( 3, 4). Our experience reiterates the fact that respiratory
complications will remain a significant problem. Lardinois
and coworkers recently reported their experience with 26
patients having chestwall resection and reconstruction with
mesh/methylmethacrylate (MMM). The authors report
that there was no 30-day mortality and nearly all patients
had a satisfactory cosmetic and functional outcome. ( 15).
The prostheses were imaged with cine-magnetic resonance
imaging and found to exhibit no paradoxic chest wall
motion in any patient ( 15). In our series factors associated
with postoperative complications and mortality were
sternal resection, concominant lung resction and patient
pulmonary capability, size of the chest wall resection as
others ( 15). In conclusion: The key to a successful outcome
in these complex cases is the coordinated effort by the
surgical teams. The frequency of respiratory complications
can be decreased by routine use of a rigid prosthesis for
reconstruction.
|
|
References
- Carbone M, Pastorino U. Surgical treatment of chest wall tumors.
World J Surg 2001;25:218-30.[LinkOut]
- Arnold PG, Pairolero PC. Chest wall reconstructionan account of
500 consecutive patients. Plast Reconstr Surg 1996;98:804-8.[LinkOut]
- Deschamps C, Tirnaksiz BM, Darbandi R. Early and long-term
results of prosthetic chest wall reconstruction. J Thorac Cardiovasc
Surg 1999;117:588-92.[LinkOut]
- Mansour KA, Thourani VH, Losken A, Reeves JG, Miller JI Jr,
Carlson GW, et al. Chest wall resections and reconstruction: a
25-year experience. Ann Thorac Surg 2002;73:1720-5;discussion
1725-6.[LinkOut]
- Graeber GM, Langenfeld J. Chest wall resection and reconstruction. In: Franco KL, Putman JR, editors. Advanced therapy in thoracic
surgery. London: BC Decker;1998.p.175-85.
- Weyant MJ, Bains MS, Venkatraman E, Downey RJ, Park BJ, Flores
RM, et al. Results of Chest Wall Resection and Reconstruction With
and Without Rigid Prosthesis. Ann Thorac Surg 2006;81:279-285.[LinkOut]
- Pairolero PC. Extend resections for lung cancer. How far is too far?
Eur J Cardiothorac Surg 1999;16:S48-50.[LinkOut]
- Magdeleinat P, Alifano M, Benbrahem C. Surgical treatment of lung
cancer invading the chest wall: results and prognostic factors. Ann
Thorac Surg 2001;71:1094-9.[LinkOut]
- Chapelier A, Fadel E, Macchiarini P. Factors affecting long-term
survival after en-bloc resection of lung cancer invading the chest
wall. Eur J Cardiothorac Surg 2000;18:513-8.[LinkOut]
- Anderson BO, Burt ME. Chest wall neoplasms and their
management. Ann Thorac Surg 1994;58:1774-81.[LinkOut]
- Picciocchi A, Granone P, Cardillo G, Margaritora S, Benzoni
C, D'ugo D. Prosthetic reconstruction of the chest wall. Int Surg
1993;78:221-4.[LinkOut]
- LeRoux BT, Shama DM. Resection of tumors of the chest wall. Curr
Probl Surg 1983;20:345-86.[LinkOut]
- McCormack -PM. Use of prosthetic materials in chest-wall
reconstruction. Surg Clin North Am 1989;69:965-76.[LinkOut]
- McKenna Jr RJ, Mountain CF, McMurtrey MJ, Larson D, Stiles
QR. Current techniques for chest wall reconstructionexpanded
possibilities for treatment. Ann Thorac Surg 1988;46:508-12.[LinkOut]
- Lardinois D, Müller M, Furrer M, Banic A , Gugger M,
Krueger T, et al. Functional assessment of chest wall integrity
after methylmethacrylate reconstr uction. Ann Thorac Surg
2000;69:919-23.[LinkOut]
- McCormack P, Bains M, Martini N, Burt M, Kaiser LR. Methods
of skeletal reconstruction following resection of lung carcinomas
invading the chest wall. Surg Clin North Am 1987;67:979-86.[LinkOut]
- Tansini I. Nuovo processo per amputations della mammella per
cancro. Reform Med 1896;12:3-5.[LinkOut]
- Brown R, Fleming W, Jurkiewicz M. An island flap of the pectoralis
major muscle. Br J Plast Surg 1977;30:161-5.[LinkOut]
- Cohen M, Ramasastry SS. Reconstruction of complex chest wall
defects. Am J Surg 1996;172:35-40.[LinkOut]
- Hultman CS, Culbertson JH, Jones GE, Losken A, Kumar AV,
Carlson GW, et al. Thoracic reconstruction with the omentum:
indications, complica-tions, and results. Ann Plast Surg
2001;46:242-9.[LinkOut]
Cite this article as: Aghajanzadeh M, Alavy A, Taskindost M, Pourrasouly Z, Aghajanzadeh G, massahnia S. Results of chest wall resection and reconstruction in 162 patients with benign and malignant chest wall disease. J Thorac Dis 2010;2(2):81-85. doi: 10.3978/j.issn.2072-1439.2010.02.02.005
|