Transarterial embolization of intralobar pulmonary sequestration in a young adult with hemoptysis
Introduction
Pulmonary sequestration is a congenital malformation characterized by the presence of dysplastic pulmonary tissue that does not communicate with the tracheobronchial tree, and has an aberrant systemic arterial supply. It occurs in 0.15–1.8% of the general population (1). Pulmonary sequestrations are classified as intra- or extra-lobar based on their location with respect to the parietal pleura, the absence or presence of a distinct pleural covering, and on variable pulmonary versus systemic venous drainage, respectively (1-3). Recent evidence supports transcatheter embolization as an effective treatment option for symptomatic pulmonary sequestration (4-7), as it is a minimally invasive technique associated with lower morbidity than surgical resection. We present a case of a young adult with recurrent hemoptysis secondary to intrapulmonary sequestration, successfully treated with a combination of polyvinyl alcohol (PVA) particles, gelfoam and platinum coil embolization.
Case presentation
An 18-year-old male smoker, with a 4-year pack history, presented to the emergency department with chest tightness and acute hemoptysis measuring approximately 240 mL in volume, on a 4-year history of recurrent, self-limiting episodes of minimal volume hemoptysis. There was no recent history of fever, productive cough or trauma. Past medical history was negative for pneumonia, or any pediatric illness. The patient was hemodynamically stable, and physical examination and routine laboratory investigations were unremarkable.
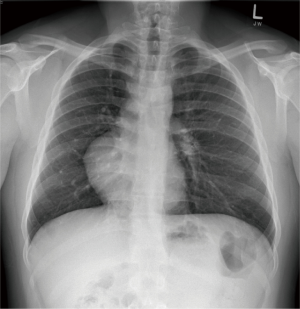
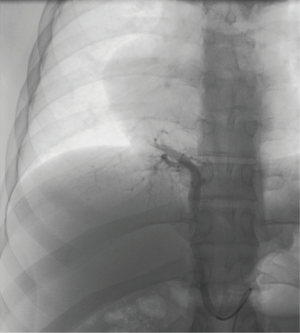
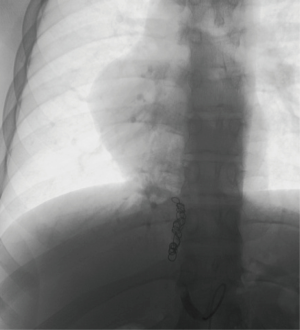
A chest radiograph suggested dextrocardia, without any significant pulmonary parenchymal or pleural abnormality (Figure 1). Subsequent CT thorax demonstrated right lower lobe medial basal segment ground glass attenuation, and a single, infra-diaphragmatic, aberrant feeding artery originating from the abdominal aorta between the celiac axis and the superior mesenteric artery (Figure 2). Conventional pulmonary venous drainage to the medial basal segment of the right lower lobe was noted. Dextrocardia secondary to partial absence of the pericardium, and persistence of the left superior vena cava (SVC) with the left SVC draining into the coronary sinus were also noted (Figure 2). The right common femoral artery was accessed for digital subtraction angiography, which confirmed a single feeding artery originating from the abdominal aorta coursing above the diaphragm toward the right lower lobe, supplying the abnormal lung tissue (Figure 3). Arteriovenous shunting was present and venous drainage was via the right inferior pulmonary vein. Given the large volume of acute hemoptysis, the feeding artery was supra-selectively catheterized and embolized with 700–1,000 micron PVA particles and gelfoam, followed by 8–12 mm platinum coil embolization. Post embolization angiogram demonstrated successful occlusion of the feeding artery (Figure 4). A 6-French Angioseal closure device was deployed in the right common femoral artery. No immediate complications occurred, and the patient was discharged home later that day.
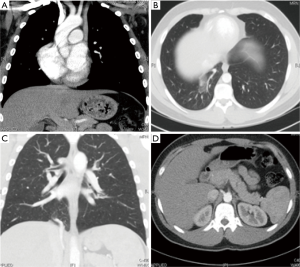
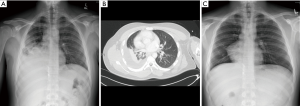
One-week post-embolization, the patient presented to hospital with right sided pleuritic chest pain. Chest radiograph and CT thorax (Figure 5A,B) demonstrated right basal airspace opacity consistent with infarcted sequestration, and associated volume loss and a right pleural effusion. Since the patient was systemically well, they were discharged home with pain medication, and their symptoms resolved. Two-week follow-up chest radiograph demonstrated near complete resolution of the previous imaging findings (Figure 5C). The patient was well and asymptomatic without any history of hemoptysis, dyspnea, exercise intolerance or pneumonia at his recent 3.5-year clinical follow-up visit.
Discussion
Seventy-five percent of pulmonary sequestrations are intralobar (8). They typically present in older children or young adults with recurrent pulmonary infection, and rarely, hemoptysis (1,8). While hemoptysis is usually minor, and associated with concurrent pneumonia (2), it can be massive and life-threatening. It is thought to be due to increased capillary pressure secondary to elevated systemic pressures in the feeding arteries (6). In patients with suspected pulmonary sequestration, CT thorax/abdomen should be performed for confirming the diagnosis and for pre-intervention planning (9).
Our patient was incidentally found to have partial absence of the pericardium, cardiac dextroposition and a duplicated SVC. Dextrocardia and duplicated SVC were previously described in association with intralobar and extralobar sequestration patients, respectively (10). While congenital cardiopulmonary, diaphragmatic, vertebral, and gastrointestinal anomalies are more commonly associated with extralobar sequestration, they can occur in up to 17% of intralobar cases (10-12), and evaluation for these can be done at the time of the CT.
Historically, sequestrations have been surgically resected in order to prevent recurrent pulmonary infections, and to protect the normal surrounding lung parenchyma (6,13,14). Evidence of spontaneous regression of pulmonary sequestrations (15), and the morbidity associated with surgical resection raises debate about the optimal management in asymptomatic patients. Spontaneous regression is thought to occur as a consequence of progressive fibrosis of the dysplastic lung tissue and feeding vessel culminating in vascular thrombosis and infarction of the abnormal lung (16). Embolization mimics this mechanism by occluding the arterial supply, leading to sequestration infarction, and is an alternative treatment method to conventional surgical resection. Moreover, the sequestration’s vascular supply is often friable due to inflammation from repeated infection, and embolization has been demonstrated to reduce the risk of intraoperative hemorrhage (17). Embolization was selected for our patient as it was the most non-invasive treatment method, and it would have reduced the risk of intra-operative hemorrhage in the event of treatment failure requiring surgical management. While the majority embolizations for sequestrations have been performed in the pediatric population (4,5,16-21), there is emerging evidence to support a potential role for this therapy in the young adult population. Successful embolization for definitive management of intralobar pulmonary sequestration has been described in a 31-year-old man with recurrent hemoptysis (6), in a 25-year-old man with hemoptysis (7), and a 23-year-old with recurrent pulmonary infections and hemoptysis (22). Sequestration embolization with PVA has been used alone (5,18), and in combination with coils (6). Other embolic agents such as coils have been used alone (4,7,17,20,21), and in combination with gelfoam (16,19,20) or an Amplatzer occlusion device (22). In a retrospective review with 72-month follow-up, no significant difference in complications or clinical outcomes was found in sequestration patients who were treated with embolization compared to surgical resection for symptoms of volume overload secondary to left-to-right shunting (23). Our case of intralobar sequestration with evidence of arteriovenous shunting successfully treated with embolization with long-term clinical follow-up adds to the limited body of literature on this topic.
While general complications of arterial embolization following endovascular treatment of pulmonary sequestration have been reported, including femoral arterial access site thrombosis and transient limb ischemia (7,19), non-target embolization of the pulmonary arteries and aorta (6,7), transient hypertension, pain, and fever (20), there is limited literature specific to the long-term follow-up of pulmonary sequestration embolization. In a case series with 24 to 70-month follow-up of five neonates who underwent transcatheter sequestration embolization via the umbilical artery, four of the neonates had complete treatment response, one had partial regression, and no complications occurred (16). In a case of pediatric bilateral sequestration successfully treated with coil embolization and gelfoam, no complication occurred over the 1-year follow-up period (21). While the requirement for repeat embolization in order to achieve cure was reported in two of six patients in pediatric case series (4) and in a 23-year-old (22), our patient was successfully treated with one embolization. The concern of radiation exposure with transcatheter embolization is valid; however, it is more relevant in a pediatric population. Furthermore, multiple techniques can be used to reduce dose including collimating, using last image hold, pulsed fluoroscopy, and avoiding magnification mode (24). Transcatheter arterial embolization is a promising minimally invasive technique that may be considered as an alternative to surgery to treat symptomatic pulmonary sequestration presenting with hemoptysis and/or arteriovenous shunting. Further research is required in order to determine the long-term clinical outcomes and complications from embolization in this patient population.
Acknowledgements
None.
Footnote
Conflicts of Interest: Amol Mujoomdar has received a speaker honorarium from Medtronic and BTG. The other authors have no conflicts of interest to declare.
Informed Consent: Written informed consent was obtained from the patient for publication of this manuscript and any accompanying images.
References
- Corbett HJ, Humphrey GM. Pulmonary sequestration. Paediatr Respir Rev 2004;5:59-68. [Crossref] [PubMed]
- Frazier AA, Rosado de Christenson ML, Stocker JT, et al. Intralobar sequestration: radiologic pathologic correlation. Radiographics 1997;17:725-45. [Crossref] [PubMed]
- Yucel O, Gurkok S, Gozubuyuk A, et al. Diagnosis and surgical treatment of pulmonary sequestration. Thorac Cardiovasc Surg 2008;56:154-57. [Crossref] [PubMed]
- Chien KJ, Huang TC, Lin CC, et al. Early and late outcomes of coil embolization of pulmonary sequestration in children. Circ J 2009;73:938-42. [Crossref] [PubMed]
- Curros F, Chigot V, Emond S, et al. Role of embolization in the treatment of bronchopulmonary sequestration. Pediatr Radiol 2000;30:769-73. [Crossref] [PubMed]
- Madhusudhan KS, Das CJ, Dutta R, et al. Endovascular embolization of pulmonary sequestration in an adult. J Vasc Interv Radiol 2009;20:1640-2. [Crossref] [PubMed]
- Ganeshan A., Freedman J, Hoey ET, et al. Transcatheter coil embolization: a novel definitive treatment option for intralobar pulmonary sequestration. Heart Lung Circ 2010;19:561-5. [Crossref] [PubMed]
- Petersen G, Martin U, Singhal A, et al. Intralobar sequestration in the middle-aged and elderly adult: Recognition and radiographic evaluation. J Thorac Cardiovasc Surg 2003;126:2086-90. [Crossref] [PubMed]
- Felker RE, Tonkin IL. Imaging of Pulmonary Sequestration – Review Article. AJR Am J Roentgenol 1990;154:241-9. [Crossref] [PubMed]
- Van Raemdonck D, De Boeck K, Devlieger H, et al. Pulmonary sequestration: a comparison between pediatric and adult patients. Eur J Cardiothorac Surg 2001;19:388-95. [Crossref] [PubMed]
- Stocker JT, Drake RM, Madewell JE. Cystic and congenital lung disease in the newborn. Perspect Pediatr Pathol 1978;4:93-154. [PubMed]
- Kravitz RM. Congenital malformations of the lung. Pediatr Clin North Am 1994;41:453-72. [Crossref] [PubMed]
- Bratu I, Flageolo H, Chen MF, et al. The multiple facets of pulmonary sequestration. J Pediatr Surg 2001;36:784-90. [Crossref] [PubMed]
- Pescarus R, Rakovich G, Oulette D, et al. A patient with an aberrant pulmonary blood supply and recurrent infection. Can Respir J 2010;17:e94-5. [Crossref] [PubMed]
- García-Peña P, Lucaya J, Hendry GM, et al. Spontaneous involution of pulmonary sequestration in children: a report of two cases and review of the literature. Pediatr Radiol 1998;28:266-70. [Crossref] [PubMed]
- Lee KH, Sung KB, Yoon HK, et al. Transcatheter arterial embolization of pulmonary sequestration in neonates: long-term follow-up results. J Vasc Interv Radiol 2003;14:363-7. [Crossref] [PubMed]
- Tokel K, Boyvat F, Varan B. Coil embolization of pulmonary sequestration in two infants; a safe alternative to surgery. AJR Am J Roentgenol 2000;175:993-5. [Crossref] [PubMed]
- Ojha V, Samui PP, Dakshit D. Role of endovascular embolization in improving the quality of life in a patient suffering from complicated intralobar pulmonary sequestration - A case report. Respir Med Case Rep 2015;16:24-8. [PubMed]
- Park ST, Yoon CH, Sung KB, et al. Pulmonary sequestration in a newborn infant: treatment with arterial embolization. J Vasc Interv Radiol 1998;9:648-50. [Crossref] [PubMed]
- Lee BS, Kim JT, Kim EA, et al. Neonatal pulmonary sequestration: clinical experience with transumbilical arterial embolization. Pediatr Pulmonol 2008;43:404-13. [Crossref] [PubMed]
- Ahn SJ, Kim EY, Kim JH, et al. Successful endovascular treatment of bilateral intralobar pulmonary sequestration with a bridging isthmus in a child. Pediatr Pulmonol 2014;49:E126-9. [Crossref] [PubMed]
- Marine LM, Valdes FE, Mertens RM, et al. Endovascular treatment of symptomatic pulmonary sequestration. Ann Vasc Surg 2011;25:696.e11-15. [Crossref] [PubMed]
- Brown SC, De Laat M, Proesmans M, et al. Treatment strategies for pulmonary sequestration in childhood: resection, embolization, observation? Acta Cardiol 2012;67:629. [PubMed]
- Miller DL, Balter S, Schueler BA, et al. Clinical radiation management for fluoroscopically guided interventional procedures. Radiology 2010;257:321-32. [Crossref] [PubMed]