Resectable primary pleural myxoid liposarcoma with a pedicle: report of a rare case and literature review
Introduction
Liposarcoma is a malignant tumor of mesenchymal origin and constitutes approximately 15% to 20% of all malignant mesenchymal tumors (1). Liposarcoma usually occurs in the lower extremities or retroperitoneum (2). An intrathoracic origin is uncommon, and most liposarcomas in this region arise in the mediastinum, constituting 2.7% of all cases (3,4). Primary liposarcoma originating from the pleura is extremely rare; among the reported cases, very few were subclassified as the myxoid type (5-9). Here, we report a primary pleural myxoid liposarcoma occupying most of the left thoracic cavity. Due to the size of the tumor, complete resection seemed impossible; however, it was attached to the pleura with a thin pedicle and was resected completely. We present the radiological findings of our case with a review of the literature.
Case presentation
A 43-year-old woman was referred to the hospital with a mass in the left thoracic cavity incidentally detected at medical check-up. The interval since previous computed tomography (CT) examination, which was negative, was 4 years. She was a non-smoker and asymptomatic, and weight loss was not observed. There was no significant finding in her medical history. Physical examination revealed resonant percussion in the left lung and decreased breath sounds with dullness on percussion in the right lung. Bronchoscopy revealed extrinsic compression of the left lower bronchus and no evidence of endobronchial tumor growth. Bronchial brush cytology was negative. The upper and lower extremities showed no abnormality. Laboratory tests were within normal limits. Echocardiography showed mitral regurgitation, and the heart was compressed to the right.
All CT examinations of the brain, chest and abdomen were performed using multislice CT (Definition, Siemens, Germany, dual-source, 64-slice). The scan parameters included a tube voltage of 120 kV, an effective tube current of 100 mA, and a slice thickness 5 mm. Unenhanced CT was followed by contrast-enhanced scan using a non-ionic contrast agent. Unenhanced CT on admission revealed a bulky mass in the left thoracic cavity and a contralateral shift of the mediastinum (Figure 1A). The mass was heterogeneous with slightly low attenuation, consisting of soft tissue areas interspersed with amorphous fat deposits. Linear peripheral calcification was noted. There was no significant enhancement of the soft tissue component after injection of contrast media (Figure 1B).
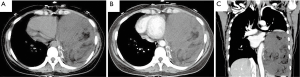
Reformatted coronal views helped clarify the tumor’s relationship to the surrounding structure (Figure 1C). The mass occupied the lower part of the left thoracic cavity and abutted the mediastinum with inversion of the ipsilateral diaphragmatic dome. The adjacent left lower lobe lung tissue was compressed and collapsed, and the left upper lobe remained inflated. No lung metastasis or nodal involvement was detected. The bones demonstrated no abnormality. Both cranial and abdominal CT showed no metastasis. Preoperatively, CT led to diagnosis of possible liposarcoma, teratoma, or hamartoma based on the fat component. Whether the lesion had invaded surrounding structures was unclear before surgery.
Left anterior lateral thoracotomy through the fourth intercostal space was performed. The mass was well-circumscribed with a pedicle originating from the left anterior pleura close to the left diaphragm, and the tumor was fully resected en masse. The tumor did not invaded surrounding vital structures or caused any pleural effusion or adhesion. The left lung expanded completely after removal of the tumor. The patient tolerated the operation well and was discharged on postoperative day 21.
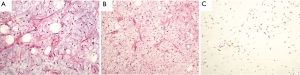
The tumor measured 21 cm × 17 cm × 9 cm, and it was well-circumscribed, characterized by a smooth surface on gross examination. The cut section revealed that the tumor contained yellowish mucoid material and was soft in consistency, partially calcified. Microscopic examination showed spindle lipoblasts, the hallmark of liposarcoma, in varying stages of differentiation, with delicate branching capillary networks set in a predominant myxoid matrix (Figure 2A). No atypical nuclei and scant mitotic figures were identified (Figure 2B). Immunohistochemistry showed that tumor cells contained nuclear S100 (Figure 2C), and the proliferative activity of the tumor cells, examined by Ki-67 staining, was about 2%.
Because the surgical margins were clear, no further treatments seemed necessary. The patient did not receive any adjuvant therapy postoperatively and was closely monitored thereafter. No sign of recurrence was detected by CT at the operation site, and the patient has remained disease free for the eight-month following the operation.
Discussion
The World Health Organization (WHO) classified liposarcomas into well-differentiated, myxoid, pleomorphic and dedifferentiated subtypes, according to complex histological components (10). Myxoid liposarcoma, the most common subtype (11,12), is usually asymptomatic, incidentally identified and may have grown to a giant size by the time of diagnosis. It is most frequently deeply located in the lower extremities and occurs in adults, usually in the fourth and fifth decade with no particular gender difference (12-15).
Because of its extreme rarity, the English literature contains little information about primary pleural myxoid liposarcoma; only nine cases have been reported, including ours (Table 1). There is a male predominance (male: female ratio =6:3), with a mean age of 53 years. Four patients were asymptomatic. Available data indicate that tumors range in size from 3.5 to 24 cm. Seven of nine patients underwent surgical resection followed by chemotherapy or radiotherapy in four patients. One patient died of intractable left ventricular failure before the operation, one patient was lost to follow up, three patients died between seven and nine months after treatment, and four patients were still alive during the follow-up from five months to four years.
A pedicle was found connecting the mass to the pleura intraoperatively, not by CT, in two cases, including the present case. Pedicles are found in approximately 42% of solitary fibrous tumors of the pleura, another rare type of pleural neoplasm (16). On imaging, a pedicle can be detected as a dependent movement in the intrathoracic position of the mass with a change in the patient position (17). This indirect finding may also be useful to identify pleural myxoid liposarcoma. The pedicle is a thin structure, so it is difficult to visualize the pedicle directly. The mass of this report occupied the lower part of the left thoracic cavity, compressed and shifted surrounding structures, so there was no extra space for it to move, even with changes in respiration or position. Pedicled tumors can be excised completely, obtaining negative histologic margins, with pedunculated tumors having the best prognosis (18). Similar results could potentially be duplicated for pleural liposarcoma. In our case, despite its huge size, the mass still presented a well-defined smooth margin; it may bs associated with the origin of pleura. Although this finding is not common, the presence of a distinct pedicle may indicate potential resectability of the mass. Due to the rarity of myxoid liposarcoma, a larger number of long-term follow-up cases will be needed.
Liposarcoma remains the most variable mesenchymal tumor, with a wide spectrum of imaging appearances (15). The CT findings of liposarcoma vary from a predominantly fat-containing mass to a solid mass depending on the histologic subtype, tumor components, and degree of vascularity. Myxoid liposarcoma is characterized by an abundant myxoid matrix, which causes an attenuation even less than that of muscle. CT showed inhomogeneous density owing to varying proportions of the admixture of lipoblasts and mucinous stroma in different regions of the mass. In our case, the lack of enhancement (a net value of 3 HU) likely resulted from the predominance of myxoid material inside the tumor, a sparse capillary network and an insufficient interval between contrast administration and scan. The variable densities of myxoid liposarcoma could be attributed to its histologic composition, namely lipoblasts in various stages of differentiation embedded in a myxoid matrix. The calcifications, more commonly seen in the myxoid type, occurred in areas of tissue degeneration and were dystrophic in nature. In this case, the linear peripheral calcification was found. Thus, these findings, such as fat containing, evidence of calcification, inhomogeneous density and the lack of enhancement supported the diagnosis of liposarcoma.
We reported a huge pleural myxoid liposarcoma with a pedicle that was resected with a negative margin. Primary pleural myxoid liposarcoma is a rare tumor that is usually asymptomatic, incidentally identified and often grows to a large size by the time of diagnosis. A pedicle of the tumor may indicate potential resectability of mass, so the existence of a pedicle should be examined to inform the preoperative strategy.
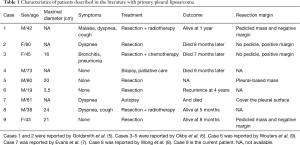
Full table
Acknowledgements
Funding: This work was supported by key research and development program of Shandong province (2015GSF118135).
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Informed Consent: Written informed consent was obtained from the patient’s husband for publication of this manuscript and any accompanying images.
References
- Tepetes K, Christodoulidis G, Spyridakis ME, et al. Liposarcoma of the stomach: A rare case report. World J Gastroenterol 2007;13:4154-5. [Crossref] [PubMed]
- Ribeiro P, Lemos J, Vaz A, et al. Something inside the heart: a myxoid liposarcoma with cardiac involvement. Rev Port Cardiol 2011;30:341-6. [PubMed]
- Kara M, Ozkan M, Dizbay Sak S, et al. Successful removal of a giant recurrent mediastinal liposarcoma involving both hemithoraces. Eur J Cardiothorac Surg 2001;20:647-9. [Crossref] [PubMed]
- Uchikov A, Poriazova E, Zaprianov Z, et al. Low-grade pulmonary myxoid liposarcoma. Interact Cardiovasc Thorac Surg 2005;4:402-3. [Crossref] [PubMed]
- Goldsmith P, Papagiannopoulos K. Pleural myxoid liposarcoma: features of 2 cases and associated literature review. J Cardiothorac Surg 2007;2:48. [Crossref] [PubMed]
- Okby NT, Travis WD. Liposarcoma of the pleural cavity: clinical and pathologic features of 4 cases with a review of the literature. Arch Pathol Lab Med 2000;124:699-703. [PubMed]
- Evans AR, Wolstenholme RJ, Shettar SP, et al. Primary pleural liposarcoma. Thorax 1985;40:554-5. [Crossref] [PubMed]
- Wong WW, Pluth JR, Grado GL, et al. Liposarcoma of the pleura. Mayo Clin Proc 1994;69:882-5. [Crossref] [PubMed]
- Wouters EF, Greve LH, Visser R, et al. Liposarcoma of the pleura. Neth J Surg 1983;35:192-3. [PubMed]
- Christopher DM, Unni KK, Mertens F. WHO classification of tumors. Pathology and genetics: tumors of soft tissue and bone. IARC Press: Lyon, France, 2002:19-34.
- Shin NY, Kim MJ, Chung JJ, et al. The Differential Imaging Features of Fat-Containing Tumors in the Peritoneal Cavity and Retroperitoneum: the Radiologic-Pathologic Correlation. Korean J Radiol 2010;11:333-45. [Crossref] [PubMed]
- Benjaminov O, Gutman H, Nyabanda R, et al. Myxoid Liposarcoma: An Unusual Presentation. AJR 2007;188:817-21. [Crossref] [PubMed]
- Kim TW, Reyes CV. Myxoid liposarcoma mimicking fluid density. J Surg Oncol 1985;30:80-2. [Crossref] [PubMed]
- Raghavan R, Raghuram P, Parekh PV, et al. Posterior mediastinal liposarcoma simulating a lung mass: an unusual case report. Cancer Imaging 2007;7:141-4. [Crossref] [PubMed]
- Weiss SW, Goldblum JR. Liposarcoma. In: Enzinger and Weiss’s soft tissue tumors, 4th edn. St Louis: CV Mosby, 2001:641-93.
- England DM, Hochholzer L, McCarthy MJ. Localized benign and malignant fibrous tumors of the pleura. A clinicopathologic review of 223 cases. Am J Surg Pathol 1989;13:640-58. [Crossref] [PubMed]
- Klein JS. Pleura, chest wall, diaphragm, and miscellaneous chest disorders. In: Brant WE, Helms C, editors. Fundamentals of Diagnostic Radiology. 3rd ed. Philadelphia: Lippincott Williams and Wilkins; 2007:527-62.
- Karabulut N, Goodman LR. Pedunculated solitary fibrous tumor of the interlobar fissure: a wandering chest mass. AJR 1999;173:476-7. [Crossref] [PubMed]


