Analyses of trends in prevalence of congenital heart defects and folic acid supplementation
Introduction
Congenital heart defects (CHDs) are of public health concern given that they occur in approximately 1% of all live births (1,2), are the leading cause of infant deaths due to birth defects (3,4), and are one of the most common reasons for use of health services among children with chronic conditions, accounting for $5.6 billion in hospitalization costs alone in 2009–15.1% of all pediatric hospitalization costs (5). These costs extend beyond childhood as children with CHD are now surviving well into later decades in life (6). Most CHDs are thought to be multifactorial in origin, involving genetic and environmental factors (7-10). Despite notable contributions of epidemiologic research in the discovery of the etiology of these defects, the number of modifiable risk factors identified has been limited: including maternal pre-pregnancy diabetes, exposure to certain medications or maternal infections (e.g., rubella), uncontrolled maternal phenylketonuria, and possibly maternal pre-pregnancy obesity, smoking, and consanguinity (8,11). There is a need to identify modifiable risk factors to provide the evidence basis for prevention interventions.
One potential modifiable risk factor for CHD that has been the subject of several epidemiologic studies in recent years is intake of folic acid during the periconceptional period (12-23). However, the results of these studies have been inconsistent. Most recently, Liu and colleagues in a study published in Circulation (24) conducted an ecologic analysis of CHD prevalence before and after the initiation of a public policy in Canada mandating folic acid fortification of food (FAFF). In this article, the authors describe trends in prevalence rates of CHD (overall), and CHD subtypes, among all live births and stillbirths/late-pregnancy terminations (>20 weeks gestation) before and after FAFF was mandated in Canada in 1998.
Liu et al. (24) studied approximately 6 million births between 1990 and 2011 in 14 geographic areas in Canada in an effort to quantify the effect of FAFF on non-chromosomal subtypes of CHD. They evaluated six subtypes of non-chromosomal CHD and found an overall reduction in prevalence of non-chromosomal CHDs of 11% (95% CI: 2−18) after FAFF. Evaluation of individual subtypes of CHD showed reductions in prevalence after FAFF of 27% (95% CI: 15−38) for conotruncal defects, 23% (95% CI: 4−39) for coarctation of the aorta, 15% (95% CI: 4−25) for ventricular septal defects (VSDs), and 18% (95% CI: 5−31) for atrial septal defects (ASDs). Findings from this type of ecologic analysis need to be interpreted with several methodological considerations in mind.
Methodological considerations
One fundamental assumption of analysis of trends in prevalence of CHD is that ascertainment and classification of CHDs in the population under study remain constant during the study period. CHDs can occur as simple (i.e., isolated/nonsyndromic) defects, while others (at least 30%) occur as complex defects (i.e., genetic, syndromic, or associations with other noncardiac defects). Some of the more complex types of CHDs may have a higher likelihood of stillbirth and/or prenatal diagnosis and elective termination (e.g., CHDs associated with chromosomal anomaly). Also, advances in prenatal echocardiography in recent decades have both allowed for earlier fetal echocardiographic screening as well as improved sensitivity and accuracy of prenatal detection and diagnosis of severe forms of CHDs (e.g., hypoplastic left heart syndrome) that may result in elective termination. If trends in prenatal diagnosis and elective termination (prior to 20 weeks gestation) of pregnancies complicated by CHD increased during the study period, then it may be difficult to assess the extent to which the observed decreasing trend in birth prevalence of CHDs is directly related to early diagnosis and elective termination versus concomitant effects of FAFF.
In the study by Liu and colleagues (24), terminations resulting from congenital anomalies were not identifiable in the dataset until 1997 (the year prior to FAFF). Therefore, terminations due to the presence of CHD were not included in the calculation of the prevalence rates for CHD in the cohort prior to FAFF for years 1990-1996 (7 of the 8 years of this cohort). If those terminations had been included, the difference in CHD prevalence detected between the two time periods/FAFF groups might have actually been much lower than shown by the study results.
The study by Liu and colleagues (24) examined the trends in prevalence of all CHD types as a group as well as for different CHD subgroups. These different CHD subgroups included conotruncal defects (common truncus, transposition of the great vessels, Tetralogy of Fallot), severe nonconotruncal defects (including endocardial cushion defects, common ventricle, and hypoplastic left heart syndrome), coarctation of the aorta, VSDs, ASDs, and “other heart and circulatory system abnormalities” (all ICD codes for congenital heart defects not included in the first 5 categories). Separate analysis of trends in prevalence for each one of these CHD subgroups comprised of anatomically similar specific CHD phenotypes allowed for a more robust assessment of trends in prevalence pre and post FAFF and to corroborate expected changes in prevalence for CHD subgroups previously reported to show associations with folic acid intake, namely conotruncal defects and VSDs (13-15,17,20,21,23). Of interest is that the decreasing trends in prevalence for these two subgroups of CHD continued well beyond the implementation of the FAFF policy, suggesting that other factors besides FAFF may have contributed in part to the decrease in prevalence during this period.
The sudden drop in prevalence of 16/10,000 to 10/10,000 between 2000 and 2002 for the subgroup of other specific CHDs was not addressed by Liu et al. Such sudden changes in prevalence may occur as a result of changes in surveillance methods such as increased detection and/or reporting of cases, or improved classification of cases and non-cases.
In studies of temporal trends in prevalence of CHD in relation to changes in FAFF at the population level, it is important to consider temporal changes in prevalence of other maternal risk factors known to be associated with CHD births. In their study (24), Liu et al. took into account an increasing prevalence of diagnosed pregestational diabetes, increasing average maternal age, and the increase in termination rate resulting from a fetal diagnosis of CHD. However, the prevalence of other potential confounders such as prepregnancy obesity and cigarette smoking that were not taken into account may have declined during the study period and could have contributed to the observed decline in prevalence in relation to FAFF implementation and beyond.
Another limitation of the study by Liu et al. (24) is that there was no information provided on trends in blood folate levels for women of childbearing age in the study population, leaving the reader to assume that the decreased prevalence of CHD observed was due to an increase in maternal folate levels due to the population level change of the FAFF. However, there was no evidence provided of trends in prevalence of serum folate levels in the population of women of childbearing age to justify this assumption.
Challenges in drawing causal inferences from ecologic analyses of environmental factors and CHDs
As pointed out in the study by Liu and colleagues (24), there are several studies in the literature supporting a preventative role for folic acid in the development of CHDs (12-22,24). These studies were reviewed and the findings demonstrated a similar trend in relation to the inverse association of outflow tract anomalies (most consistently conotruncal defects) and ventricular septal defects with the supplementation of either isolated folic acid (by dietary fortification or pharmaceutical supplementation) or multivitamin supplementation. More precisely, there was a reduction in risk of these subtypes of cardiac defects seen in multiple studies, despite mode of supplementation with folic acid (presumed mechanism of multivitamin supplementation). The findings for outflow tract anomalies and ventricular septal defects are summarized in (Table 1). Czeizel and colleagues (15,16) reported a 43% reduction in the prevalence of CHD in their series of 2 studies which included a randomized controlled trial (RCT) in Hungary followed by a cohort controlled trial which was initiated after the RCT was stopped. In an additional study in 2015, Czeizel reported that medical documentation of maternal high-dose folic acid supplementation during the critical period of cardiac development was associated with a significant reduction in prevalence of VSDs (OR 0.57, 95% CI: 0.45–0.73), d-transposition of the great vessels (OR 0.47, 95% CI: 0.26−0.86), Tetralogy of Fallot (0.53, 95% CI: 0.17–0.94), and ASDs (OR 0.63, 95% CI: 0.4–0.98) (17).
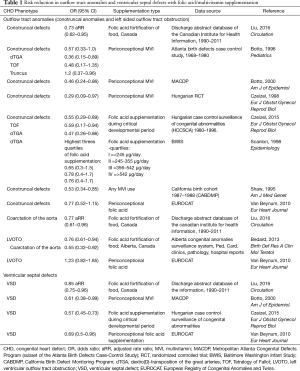
Full table
Alternatively, Bedard et al., (13) in a population in Alberta, Canada, reported no significant decline overall in isolated CHDs after FAFF but found that left ventricular outflow tract obstruction and coarctation of the aorta showed a significant decrease in prevalence. Of interest is that they did find that isolated ASDs and ASD with associated VSD both demonstrated an increased prevalence after FAFF (13).
Leirgul et al. (18) evaluated 517,748 non-chromosomal pregnancies in Norway and found that maternal report of regular intake of multivitamin and folic acid had no association with severe CHDs. However, these investigators did find and unexpected 20% increase in the prevalence of VSDs in those reporting regular use of folic acid in pregnancy, and an overall trend during the 10-year study period of increased folic acid intake from 1999 to 2009 and a simultaneous overall decline in CHD over the same period. There was no significant change in prevalence for severe defects over the study period (18).
When considering the findings of Liu and colleagues (24) in comparison with the above mentioned studies, Liu et al. (24) reported a smaller overall decrease in prevalence of CHD than Czeizel (17). However, both groups demonstrated a reduction in both conotruncal defects as well as atrial and ventricular septal defects when subtypes of CHD were evaluated. Bedard (13) showed a decrease in left sided obstructive lesions (left ventricular outflow tract obstruction and coarctation of the aorta). He detected no significant overall decline in CHD, unlike the first two groups. He also showed an increase in the prevalence of isolated ASDs as well as ASDs with associated VSDs. Similarly, Leirgul and colleagues (18) reported an overall decline in all forms of CHD but no association with severe CHD, and surprisingly found a 20% increase in prevalence of VSDs.
Feng and colleagues (25) recently published a comprehensive meta-analysis of the literature on maternal folic acid supplementation the risk of CHDs. This meta-analysis included 18 studies, consisting of 1 randomized controlled trial, 1 cohort study, and 16 case-control studies. They found an overall significantly decreased risk of CHD with maternal folic acid supplementation (OR 0.72, 95% CI: 0.63−0.82). When CHD subtypes were considered, they found similar risk reduction for conotruncal defects (RR 0.64, 95% CI: 0.54−0.76), septal defects-isolated VSD or ASD (RR 0.64, 95% CI: 0.49−0.83), and atrioventricular septal defects (RR 0.60, 95% CI: 0.44−0.83).
The finding of decreased prevalence of conotruncal defects with maternal folic acid supplementation in several studies is encouraging, but not consistent across all studies. The difference in findings related to septal defects across the studies could be due to a number of factors including differences in clinical methods and rates of detection of septal defects as well as timing of evaluation and differences in defining ASD versus patent foramen ovale (persistent fetal structure). When considering differences across studies for each subtype, the variability of classification methods of CHD used in each case may also explain, in part, the resulting discrepancies in findings.
Importance of dietary intake of folic acid and additional supplementation in women of childbearing age
Despite these methodological considerations, the results of the study by Liu and colleagues are important as they are suggestive that folic acid levels may have an impact on the reduction in prevalence of CHD. The current FAFF policy in Canada and in the US and recommendations for increased dietary intake of folic acid and additional pharmacological supplementation for all women of childbearing age to prevent neural tube defects may also contribute to the prevention of some CHDs (e.g., conotruncal defects and VSDs). The development of the cardiac system, also being completed early in gestation, stands to potentially benefit from such prevention intervention efforts, particularly among pregnancies to women with increased susceptibility for CHDs among their offspring (e.g., women with cardiometabolic disorders). Further studies are needed to investigate the role of folic acid in the development of CHD and the potential biologic mechanisms of the presumed impact on the development of the fetal cardiovascular system.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
- Hoffman JI, Kaplan S. The incidence of congenital heart disease. J Am Coll Cardiol 2002;39:1890-900. [Crossref] [PubMed]
- van der Linde D, Konings EE, Slager MA, et al. Birth prevalence of congenital heart disease worldwide: a systematic review and meta-analysis. J Am Coll Cardiol 2011;58:2241-7. [Crossref] [PubMed]
- Petrini J, Damus K, Johnston RB Jr. An overview of infant mortality and birth defects in the United States. Teratology 1997;56:8-10. [Crossref] [PubMed]
- Petrini J, Damus K, Russell R, et al. Contribution of birth defects to infant mortality in the United States. Teratology 2002;66 Suppl 1:S3-6. [Crossref] [PubMed]
- Association AH. Congenital Cardiovascular Defects; Statistical Fact Sheet; 2016 Update. 2016. Available online: https://www.heart.org/idc/groups/heart-public/@wcm/@sop/@smd/documents/downloadable/ucm_483967.pdf
- Marelli AJ, Mackie AS, Ionescu-Ittu R, et al. Congenital heart disease in the general population: changing prevalence and age distribution. Circulation 2007;115:163-72. [Crossref] [PubMed]
- Fung A, Manlhiot C, Naik S, et al. Impact of prenatal risk factors on congenital heart disease in the current era. J Am Heart Assoc 2013;2:e000064. [Crossref] [PubMed]
- Jenkins KJ, Correa A, Feinstein JA, et al. Noninherited risk factors and congenital cardiovascular defects: current knowledge: a scientific statement from the American Heart Association Council on Cardiovascular Disease in the Young: endorsed by the American Academy of Pediatrics. Circulation 2007;115:2995-3014. [Crossref] [PubMed]
- Vecoli C, Pulignani S, Foffa I, et al. Congenital heart disease: the crossroads of genetics, epigenetics and environment. Curr Genomics 2014;15:390-9. [Crossref] [PubMed]
- Zhu H, Kartiko S, Finnell RH. Importance of gene-environment interactions in the etiology of selected birth defects. Clin Genet 2009;75:409-23. [Crossref] [PubMed]
- Shieh JT, Bittles AH, Hudgins L. Consanguinity and the risk of congenital heart disease. Am J Med Genet A 2012;158A:1236-41. [Crossref] [PubMed]
- Bailey LB, Berry RJ. Folic acid supplementation and the occurrence of congenital heart defects, orofacial clefts, multiple births, and miscarriage. Am J Clin Nutr 2005;81:1213S-7S. [PubMed]
- Bedard T, Lowry RB, Sibbald B, et al. Folic acid fortification and the birth prevalence of congenital heart defect cases in Alberta, Canada. Birth Defects Res A Clin Mol Teratol 2013;97:564-70. [Crossref] [PubMed]
- Botto LD, Mulinare J, Erickson JD. Occurrence of congenital heart defects in relation to maternal mulitivitamin use. Am J Epidemiol 2000;151:878-84. [Crossref] [PubMed]
- Czeizel AE. Periconceptional folic acid containing multivitamin supplementation. Eur J Obstet Gynecol Reprod Biol 1998;78:151-61. [Crossref] [PubMed]
- Czeizel AE, Dudas I. Prevention of the first occurrence of neural-tube defects by periconceptional vitamin supplementation. N Engl J Med 1992;327:1832-5. [Crossref] [PubMed]
- Czeizel AE, Vereczkey A, Szabo I. Folic acid in pregnant women associated with reduced prevalence of severe congenital heart defects in their children: a national population-based case-control study. Eur J Obstet Gynecol Reprod Biol 2015;193:34-9. [Crossref] [PubMed]
- Leirgul E, Gildestad T, Nilsen RM, et al. Periconceptional Folic Acid Supplementation and Infant Risk of Congenital Heart Defects in Norway 1999-2009. Paediatr Perinat Epidemiol 2015;29:391-400. [Crossref] [PubMed]
- Scanlon KS, Ferencz C, Loffredo CA, et al. Preconceptional folate intake and malformations of the cardiac outflow tract. Baltimore-Washington Infant Study Group. Epidemiology 1998;9:95-8. [Crossref] [PubMed]
- Shaw GM, O'Malley CD, Wasserman CR, et al. Maternal periconceptional use of multivitamins and reduced risk for conotruncal heart defects and limb deficiencies among offspring. Am J Med Genet 1995;59:536-45. [Crossref] [PubMed]
- van Beynum IM, Kapusta L, Bakker MK, et al. Protective effect of periconceptional folic acid supplements on the risk of congenital heart defects: a registry-based case-control study in the northern Netherlands. Eur Heart J 2010;31:464-71. [Crossref] [PubMed]
- Werler MM, Hayes C, Louik C, et al. Multivitamin supplementation and risk of birth defects. Am J Epidemiol 1999;150:675-82. [Crossref] [PubMed]
- Botto LD, Khoury MJ, Mulinare J, et al. Periconceptional multivitamin use and the occurrence of conotruncal heart defects: results from a population-based, case-control study. Pediatrics 1996;98:911-7. [PubMed]
- Liu S, Joseph KS, Luo W, et al. Effect of Folic Acid Food Fortification in Canada on Congenital Heart Disease Subtypes. Circulation 2016;134:647-55. [Crossref] [PubMed]
- Feng Y, Wang S, Chen R. Maternal folic acid supplementation and the risk of congenital heart defects in offspring: a meta-analysis of epidemiological observational studies. Sci Rep 2015;5:8506. [Crossref] [PubMed]


