Statin-ezetimibe versus statin lipid-lowering therapy in patients with acute coronary syndromes undergoing percutaneous coronary intervention
Introduction
Hypercholesterolemia is firmly believed as an important risk factor of atherosclerotic cardiovascular disease (ASCVD) and it is a major concern to the medical community as the incidence of ASCVD is increasing gradually. The cholesterol treatment trialist (CTT) collaborators reported that the incidence of major adverse cardiovascular events can be decreased by approximately 22% per 1.0 mmol/L low density lipoprotein cholesterol (LDL-C) reduction regardless of the baseline level of LDL-C (1). Statins are recommended by many guidelines as the first-line therapy for ASCVD and are used extensively in clinical practice (2-5).
Numerous studies have shown that statins can significantly reduce LDL-C levels and slow down process of coronary plaque formation, thus ameliorating the risk of cardiovascular events (6-9). According to 2013 American College of Cardiology (ACC) guideline, intensive statin therapy has been recommended to patients with ASCVD for lowering blood cholesterol (5). However, the safety concerns have been associated with high-dose statin therapy, especially in Asian patients.
The IMPROVE-IT study found that the risk of cardiovascular events further decreased significantly when LDL-C levels were reduced from 1.8 mmol/L down to 1.4 mmol/L for patients with ACS (10,11). The most recent FOURIER trail reported that inhibition of proprotein convertase subtilisin/kexin type 9 (PCSK9) combined with statin therapy can decrease LDL-C to 0.78 mmol /L (59% reduction compared with placebo group) and reduce the relative risk of vascular events by 15% (12). These results suggest that risks of cardiovascular events (MACEs) may be reduced directly by lowering LDL-C levels regardless of the type of lipid-lowering drug.
When moderate-intensity statin therapy is inefficient in lowering LDL-C level in some patients, two strategies could therefore be considered: doubling the dosage of the statin or adding another lipid-lowering drug, such as ezetimibe, to statin therapy. Studies comparing these two strategies were rare, especially in Asian patients. This study aimed to explore the efficacy and safety of statins/ezetimibe combination therapy as compared with statin therapy alone in treating patients with ACS who underwent successful PCI.
Methods
Study population
This was a retrospective observational study. The participants in the study were enrolled from the patients who had been hospitalized at department of cardiology in the first hospital of China Medical University between May 2016 and July 2016. The patients with ACS who underwent PCI and did not take lipid-lowering drugs within 1 month were eligible for inclusion. Two hundred and two patients were enrolled and treated with statin/ezetimibe or statin alone. Patients were excluded if they were pregnant or lactating. Patients with severe liver and kidney dysfunctions (ALT >3 times upper limit of normal, eGFR <50 mL/min), malignant tumors, severe autoimmune diseases or hypothyroidism were also excluded from the study.
Study method
Patients received standard medical treatment for acute coronary syndrome (ACS) according to their own conditions. They were divided into three groups according to the lipid-lowering strategies. Patients in group A received moderate-intensity statin treatment, patients in group B received ezetimibe combined with moderate-intensity statins (ezetimibe-statin combination) treatment; and patients in group C were treated with intensive statins. According to 2013 ACC/AHA guideline (5), moderate-intensity statin therapy was defined as administrations of rosuvastatin at dosage 5–10 mg daily, atorvastatin of 10–20 mg daily, pravastatin 40 mg daily or fluvastatin 80 mg daily, and high-intensity statin therapy was defined as administrations of atorvastatin at dosage of 40–80 mg or rosuvastatin 20 mg per day. Serum cholesterol level and hepatic/muscle enzymes were measured and recorded at admission, 1 and 3 months after therapy. The patients were followed up for 6 months and clinical outcomes were recorded.
End points
The extents or percentages of cholesterol lowering were compared among the three groups of patients. The percentage of patients attaining the LDL-C goal was also compared among the groups. The LDL-C goal is defined as LDL-C <70 mg/dL (1.8mmol/L) or a reduction of more than 50% if the pre-treatment level is within a range of 1.8 and 3.5 mmol/L (4). MACEs, defined as all-cause death, non-fatal myocardial infarction (MI), target vessel revascularization (PCI or bypass surgery) and stroke, were analyzed and compared at 3 months after PCI therapy.
Statistical analysis
In this study, categorical data were analyzed by the chi-square test and results were presented as absolute value and percentage. Continuous variables were analyzed by analysis of variance (ANOVA) and results were described as mean ± standard deviation. Differences of lipid in magnitude and percentage of change between two groups were assessed by LSD-t test. The difference was considered as statistically significant if P value was less than 0.05.
Results
Patient characteristics
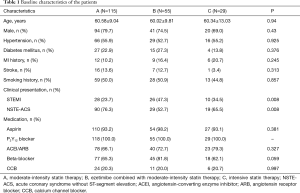
In this study, 118 patients were assigned to moderate-intensity statin group, 55 to ezetimibe combined with moderate-intensity statins (ezetimibe/statin) group, and 29 to high-intensity statin group. The baseline characteristics of participants in three groups were shown in Table 1. The average age of the patients was 60.4 years and 47 (23.3%) were women. The proportions of STEMI among the three groups was significantly different (P=0.008), and the ezetimibe/statin combination group had higher proportion of STEMI compared with moderate-intensity statin and high-intensity statin group. No other obvious difference was found in baseline characteristics of the patients among the three groups.
Full table
Lipid data
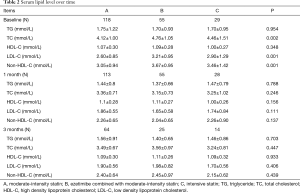
At admission, the mean concentration of serum TC was 4.34 mmol/L, the mean serum LDL-C level was 2.81 mmol/L and the average level of serum non-HDL-C was 3.28 mmol/L in overall patients. There were significant differences in TC, LDL-C and Non-HDL-C levels among the three groups of patients (P<0.05). The mean levels of serum LDL-C, TC and non-HDL-C in ezetimibe/statin combination group were higher than those in moderate-intensity statins group (P<0.001) (Table 2). No significant difference was found in the mean TG and HDL-C levels among the three groups.
Full table
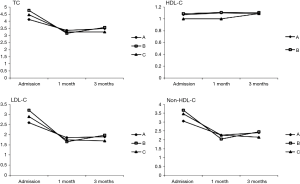
At 1 month follow-up, the TC, LDL-C and non-HDL-C levels were significantly decreased in overall patients as shown in Figure 1. The mean concentration of TC was 3.29 mmol/L, the mean level of LDL-C was 1.78 mmol/L, and the average level of non-HDL-C was 2.20 mmol/L. The reduction percentage of serum TC was significantly larger in ezetimibe-statin combination treated patients than in those of moderate-intensity statin group (31.70% vs. 15.24%, Table 3). The lowering percentage of LDL-C was 30.71% in overall patients. Compared with moderate-intensity statin group, the lowering percentage of LDL-C was higher in ezetimibe-statin combination group and intensive statins group (P<0.05) (Figure 2). The lowering percentage of LDL-C was 45% in ezetimibe-statin combination group and 35.3% in intensive statin group (P=0.117). The lowering percentage of non-HDL-C was significantly higher in ezetimibe-statin combination treated patients than in those of two other groups (Table 3).
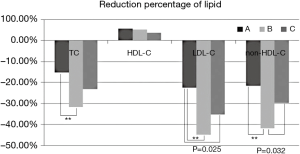
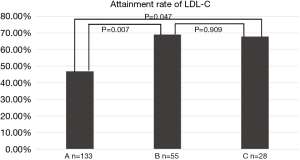
As shown in Figure 3, greater proportions of patients achieved goal of lowering LDL-C in ezetimibe-statin combination group (69.1%) and intensive statin group (67.9%) than in moderate-intensity statin group (46.9%) at 1 month of follow-up (P<0.05).
Clinical outcomes
During 6 months’ follow-up, only three MACE cases occurred in overall patients. There was one onset stroke and one coronary artery bypass grafting in moderate-intensity statin group and one onset stroke in ezetimibe-statin combination group.
Safety
At 1 month follow-up, no significant difference was detected about serum ALT level among the three groups (Table 4). Only three patients had elevation in serum ALT level that exceeded 3 times the upper limits of the normal range in ezetimibe-statin combination group during the follow-up. There were 16 patients whose serum ALT elevated over the normal upper limit. Eight (7.1%) were in moderate-intensity statin group, six (11.3%) in ezetimibe-statin combination group and two (7.7%) in intensive statins group (P=0.585). The concentration of serum ALT in patients of ezetimibe-statin combination group was higher than that of the other two groups, but no significant difference among the three groups (P=0.24).

Full table
Only one patient had CK increased over the normal upper limit in moderate-intensity statin group, and no significant difference was found about mean serum CK among the three groups (Table 4). Five patients in moderate-intensity statin group and two in ezetimibe-statin combination group had muscle-related adverse events.
No significant difference was detected on the average fasting blood glucose among the three groups (P=0.223). Fasting blood glucose in six patients exceeded 6.1 mmol/L, three patients in moderate-intensity statin group, two in ezetimibe-statin combination group and one in intensive statin group. There was no onset diabetes in overall patients.
No significant difference was presented in serum creatinine (Table 4) or Cys-C levels among the three groups (P=0.65 and P=0.181).
Discussion
In this study, we demonstrated that moderate statin/ezetimibe combination therapy could contribute to greater reduction of non-HDL-C than intensive statin therapy. We also found that ezetimibe-statin combination therapy had similar effect on LDL-C and TC reductions as intensive statin therapy. These two therapies were more effective on cholesterol reduction than moderate-intensity statin therapy. No significant differences of side effects were seen in the three groups of patients.
Statins can improve the risk of MACE in ASCVD patients (13,14) and high-intensity statin therapy might be more effective as compared with standard-dose statin therapy (15,16). However, many studies have shown that intensive statin therapy could lead to a higher incidence of side effects or adverse events, especially in Chinese population (3,17). Regimens with higher statin dosage or combination of another lipid lowering drug might be effective for patients who aren’t responsive well to standard statin treatment (18). The 2016 ESC/EAS Guidelines for dyslipidemia recommend that all ACS patients without contraindications or intolerance history should start or continue to use high-intensity statin as soon as possible regardless of initial LDL-C levels (Recommendation Class I) (4). However, Chinese guidelines recommend moderate-intensity statins as a general treatment regimen for dyslipidemia (3). For patients unable to reach LDL-C goal or intolerant with moderate-intensity statins therapy, ezetimibe combined with low/moderate-intensity statins may be a better alternative choice (3). It is unknown whether ezetimibe combined with statin can lead to better clinical outcomes than intensive statin therapy.
Many studies have shown that LDL-C levels are closely related to cardiovascular events (1,19,20). This study explored the efficacy of different lipid-lowering therapies in reducing lipid levels, particularly LDL-C and non-HDL-C levels. We found that the TC, LDL-C and non-HDL-C levels in the ezetimibe-statin treated and intensive statins patients were higher than in those of moderate-intensity statin group at the time of admission. The baseline lipid profile difference could account for the different lipid lowering strategies prescribed by physicians in three groups of patients. Ji et al. found in their study that the LDL-C reduction was similar between ezetimibe-statin combination group and intensive statin group for patients suffered acute myocardial infarction (9), which was consistent with this study. In our study, the proportion of patients reaching LDL-C goal was higher in ezetimibe-statin combination group than moderate-intensity statin group, and this result was similar to other published studies (21). In addition, we found that the extent of LDL-C reduction and the proportion of patients reaching LDL-C goal were similar in ezetimibe-statin combination group and intensive statins group.
LDL-C is believed to be the main risk factor for the formation of atherosclerotic plaque and is therefore identified as the primary target of cholesterol management. However, recent studies found that very low density lipoproteins (VLDLs) also have atherogenic potentials similar to LDL (22). Therefore, non-HDL-C, including VLDL-C, LDL-C and other ApoB containing lipoproteins, is the major form of atherogenic lipoproteins. Existing data has demonstrated that non-HDL-C has a stronger association with risk of ASCVD events, as compared with LDL-C (23). In addition, LDL-C is calculated by a formula (LDL-C = TC − HDL-C − ⅕TG) in most institutions, and therefore, the calculated LDL-C level could be under-evaluated if TG level is higher than normal. In contrast£¨non-HDL-C can be measured accurately even under non-fasting conditions. For these reasons, international atherosclerosis society (IAS) has recommended non-HDL-C as an alternate target to LDL-C in the management of dyslipidemia (24). Moreover, the IAS panel predicted that non-HDL-C will replace LDL-C as the primary target of hypercholesterolemia treatment in future guidelines (24). In the present study, we found that there was a greater reduction of non-HDL-C in ezetimibe-statin combination group than in intensive statin group (P=0.032). This result suggested that ezetimibe-statin combination therapy might be more effective than intensive statin in lowering ApoB-containing lipoproteins and decreasing cardiovascular events for ASCVD patients.
Lots of studies have shown that intensive statin therapy may cause elevated indicators of liver functions, statin-associated muscle symptoms, onset diabetes mellitus and other adverse events (10,25-28). Approximately 5% to 10% patients receiving statin therapy may have adverse events, the most common of which is statin-associated myopathy(28). Previous trials reported that about sixty-two percent of patients discontinued statin therapy due to statin-related adverse events (29). No significant difference was found in muscle enzymes among the three groups of patients in our study. During the duration of follow-up, only one patient had CK elevation over the normal upper limit and five patients had muscle-related discomfort in the moderate-intensity statins group, two patients had muscle-related discomfort in ezetimibe-statin combination group and no patient had muscle-related discomfort in the intensive statins group. This finding was not consistent with the previous studies (30). This difference may be attributed to different patient sample, lost visits during follow-up and differences in patients’ compliance in different studies. Moreover, no significant difference was found in liver function test, serum creatinine and blood glucose levels among the three groups of patients.
In conclusion, the present study demonstrated that ezetimibe-statin combination therapy was more effective on lowering non-HDL-C level than intensive statin therapy. The results indicated that statin/ezetimibe combination therapy could be considered as an alternative to high-intensity statin therapy for Chinese ASCVD patients.
Acknowledgements
Funding: This study was supported by Liaoning provincial Science and Technology Plan (Project ID: 2015225027).
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: Due to the retrospective nature of this study, the need for ethics approval and informed consent were waived.
References
- Collaboration CTT. Efficacy and safety of more intensive lowering of LDL cholesterol: a meta-analysis of data from 170 000 participants in 26 randomised trials. Lancet 2010;376:1670-81. [Crossref] [PubMed]
- Chinese Society of Cardiology Task Force on Interventional Cardiology, et al. Chinese Guidelines for Percutaneous Coronary Intervention. Zhonghua Xin Xue Guan Bing Za Zhi 2016;44:1-20. [PubMed]
- Joint Committee for Developing Chinese guidelines on Prevention and Treatment of Dyslipidemia in Adults. Guidelines for prevention and treatment of dyslipidemia in Chinese adults (revised 2016). Chin Circul J 2016;31:937-53.
- Catapano AL, Graham I, De Backer G, et al. 2016 ESC/EAS Guidelines for the Management of Dyslipidaemias. Eur Heart J 2016;37:2999-3058. [Crossref] [PubMed]
- Stone NJ, Robinson JG, Lichtenstein AH, et al. 2013 ACC/AHA Guideline on the Treatment of Blood Cholesterol to Reduce Atherosclerotic Cardiovascular Risk in Adults. J Am Coll Cardiol 2014;63:2889-934. [Crossref] [PubMed]
- Tsujita K, Sugiyama S, Sumida H, et al. Impact of Dual Lipid-Lowering Strategy With Ezetimibe and Atorvastatin on Coronary Plaque Regression in Patients With Percutaneous Coronary Intervention: The Multicenter Randomized Controlled PRECISE-IVUS Trial. J Am Coll Cardiol 2015;66:495-507. [Crossref] [PubMed]
- Nicholls SJ, Brandrup-Wognsen G, Palmer M, et al. Meta-analysis of comparative efficacy of increasing dose of Atorvastatin versus Rosuvastatin versus Simvastatin on lowering levels of atherogenic lipids (from VOYAGER). Am J Cardiol 2010;105:69-76. [Crossref] [PubMed]
- Catapano AL, Farnier M, Foody JM, et al. Combination therapy in dyslipidemia: where are we now? Atherosclerosis 2014;237:319-35. [Crossref] [PubMed]
- Ji MS, Jeong MH. Clinical outcome of statin plus ezetimibe versus high-intensity statin therapy in patients with acute myocardial infarction propensity-score matching analysis. Int J Cardiol 2016;225:50-9. [Crossref] [PubMed]
- Cannon CP, Blazing MA, Giugliano RP, et al. Ezetimibe Added to Statin Therapy after Acute Coronary Syndromes. N Engl J Med 2015;372:2387-97. [Crossref] [PubMed]
- Blazing MA, Giugliano RP, Cannon CP, et al. Evaluating cardiovascular event reduction with ezetimibe as an adjunct to simvastatin in 18,144 patients after acute coronary syndromes: final baseline characteristics of the IMPROVE-IT study population. Am Heart J 2014;168:205-12. [Crossref] [PubMed]
- Sabatine MS, Giugliano RP, Keech AC, et al. Evolocumab and Clinical Outcomes in Patients with Cardiovascular Disease. N Engl J Med 2017;376:1713-22. [Crossref] [PubMed]
- Cannon CP, Braunwald E, McCabe CH, et al. Intensive versus Moderate Lipid Lowering with Statins after Acute Coronary Syndromes. N Engl J Med 2004;350:1495-504. [Crossref] [PubMed]
- Pedersen TR, Faergeman O, Kastelein JJP, et al. High-Dose Atorvastatin vs Usual-Dose Simvastatin for Secondary Prevention After Myocardial Infarction The IDEAL Study: A Randomized Controlled Trial. JAMA 2005;294:2437-45. [Crossref] [PubMed]
- Pauriah M, Elder DH, Ogston S, et al. High-potency statin and ezetimibe use and mortality in survivors of an acute myocardial infarction: a population-based study. Heart 2014;100:867-72. [Crossref] [PubMed]
- Lipinski M, de Vries FM, Kolthof J, et al. Efficacy of Standard and Intensive Statin Treatment for the Secondary Prevention of Cardiovascular and Cerebrovascular Events in Diabetes Patients: A Meta-Analysis. PLoS ONE 2014;9:e111247. [Crossref] [PubMed]
- Silva M, Matthews ML, Jarvis C, et al. Meta-analysis of drug-induced adverse events associated with intensive-dose statin therapy. Clin Ther 2007;29:253-60. [Crossref] [PubMed]
- Dujovne CA, Williams CD, Ito MK. What combination therapy with a statin, if any, would you recommend? Curr Atheroscler Rep 2011;13:12-22. [Crossref] [PubMed]
- Jarcho JA, Keaney JF. Proof That Lower Is Better — LDL Cholesterol and IMPROVE-IT. N Engl J Med 2015;372:2448-50. [Crossref] [PubMed]
- McCormack T, Dent R, Blagden M. Very low LDL-C levels may safely provide additional clinical cardiovascular benefit: the evidence to date. Int J Clin Pract 2016;70:886-97. [Crossref] [PubMed]
- Ambegaonkar BM, Tipping D, Polis AB, et al. Achieving goal lipid levels with ezetimibe plus statin add-on or switch therapy compared with doubling the statin dose. A pooled analysis. Atherosclerosis 2014;237:829-37. [Crossref] [PubMed]
- Varbo A, Benn M, Tybjaerg-Hansen A, et al. Remnant cholesterol as a causal risk factor for ischemic heart disease. J Am Coll Cardiol 2013;61:427-36. [Crossref] [PubMed]
- Boekholdt SM, Arsenault BJ, Mora S, et al. Association of LDL cholesterol, non-HDL cholesterol, and apolipoprotein B levels with risk of cardiovascular events among patients treated with statins: a meta-analysis. JAMA 2012;307:1302-9. [Crossref] [PubMed]
- Expert Dyslipidemia Panel of the International Atherosclerosis Society Panel m. An International Atherosclerosis Society Position Paper: global recommendations for the management of dyslipidemia--full report. J Clin Lipidol 2014;8:29-60. [Crossref] [PubMed]
- Barkas F, Elisaf M, Liberopoulos E, et al. Statin therapy with or without ezetimibe and the progression to diabetes. J Clin Lipidol 2016;10:306-13. [Crossref] [PubMed]
- Pandor A, Ara RM, Tumur I, et al. Ezetimibe monotherapy for cholesterol lowering in 2,722 people: systematic review and meta-analysis of randomized controlled trials. J Intern Med 2009;265:568-80. [Crossref] [PubMed]
- Thompson PD, Panza G, Zaleski A, et al. Statin-Associated Side Effects. JACC 2016;67:2395-410. [Crossref] [PubMed]
- Ahmad Z. Statin intolerance. Am J Cardiol 2014;113:1765-71. [Crossref] [PubMed]
- Cohen JD, Brinton EA, Ito MK, et al. Understanding Statin Use in America and Gaps in Patient Education (USAGE): an internet-based survey of 10,138 current and former statin users. J Clin Lipidol 2012;6:208-15. [Crossref] [PubMed]
- Armitage J, Bowman L, Wallendszus K, et al. Intensive lowering of LDL cholesterol with 80 mg versus 20 mg simvastatin daily in 12 064 survivors of myocardial infarction: a double-blind randomised trial. Lancet 2010;376:1658-69. [Crossref] [PubMed]

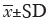 )
) 
