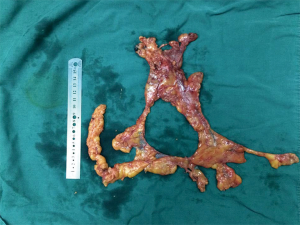
Video-assisted thoracoscopic completion thymectomy based on mediastinal pleura guidance
Introduction
Thymectomy is considered a curative treatment for resectable thymoma patients (1). Thymectomy is a primary reason for high remission rates because it completely removes the thymoma, thymus and mediastinal lipid. Although a variety of other surgical approaches have been widely applied, such alternatives have many drawbacks. There is no ideal clinical surgical approach that can be used to entirely remove the thymus, thymoma and bilateral mediastinum lipid with minimal invasion. We introduced the right thorax three-port video-assisted thoracoscopic surgery (VATS) thymectomy based on mediastinal pleura guidance; we used single-lumen endotracheal tube (SLET) anesthesia and CO2 artificial pneumothorax to achieve the en bloc resection of the thymoma, thymus, and bilateral mediastinal lipid and pleura. This new approach is applicable for the treatment of Masaoka stages I and II thymomas with myasthenia gravis.
Operative technique
The “mediastinal pleura guidance” technique is that by grasping and pulling mediastinal pleura and lipid upward altogether, the mediastinal pleura and the lipid attached to it are wholly resected along the bilateral mediastinal pleura. It can achieve en block resection of the thymoma, thymus and bilateral mediastinum lipid, and avoid lipid broken and ectopic thymus residual (Figure 1).

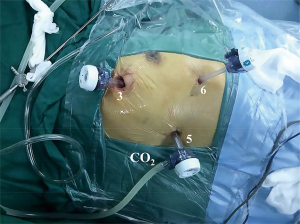
A combination of midazolam (0.03 mg/kg) and rocuronium (0.6 mg/kg) for general anesthesia was used to facilitate intubation. The patients were intubated with a SLET soon after they lost consciousness. The tidal volume was adjusted to 8–10 mL/kg. In the VATS thymectomy, a thoracoscopic thymectomy was performed with the patients placed in a right lateral supine position; three 1-cm skin incisions were made on the right anterior axillary line in the third and sixth intercostal spaces and on the right posterior axillary line in the fifth intercostal space. A 30° oblique view rigid thoracoscope was inserted into the fifth port for viewing. Additionally, the third and sixth ports were used for the operation with an ultrasonic scalpel and grasper. CO2 artificial pneumothorax was used, and the pressure was maintained between 8–12 mmHg (average 10 mmHg). The use of single-phase ventilation cannulas was necessary to effectively prevent CO2 leaks during the operation (Figure 2).
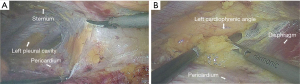
It was the essential points of operation that sweeping all thymoma, thymus, and mediastinal lipid based on the guidance of grasping mediastinal pleura. The dissection was initiated at the right mediastinal pleura at the junction of the superior vena cava and the pericardium. Then, the right mediastinal pleura, as well as the lipid that was attached to it, was mobilized cephalad along the superior vena cava and medially along the right phrenic nerve using an endoscopic ultrasonic scalpel. The dissection was continued along the innominate veins until both of the upper poles of the thymus and the peripheral lipid were completely resected. Meanwhile, the thymic veins were cut off using the ultrasonic scalpel. Then, the residual right mediastinal pleura and the attached lipid were completely swept. The left pleural space was opened at the left cardiodiaphragmatic angle of the retrosternal space. The lipid above the pericardium and the left mediastinal pleura were swept from caudal to cephalad until the mediastinal lipid and thymus were swept en bloc (Figure 3A,B). The resected specimen was then placed in an endoscopic pouch and taken out through one incision (Figure 4). In cases where the mass was too large, we appropriately enlarged one port or carefully extracted the mass piece by piece using the endoscopic pouch to avoid potential intrathoracic spreading. Finally, one drainage tube was placed in the incision of the fifth intercostal space.
Comments
Video-assisted thoracoscopic completion thymectomy based on mediastinal pleura guidance enables the en bloc resection of thymus and mediastinal fat without damaging the phrenic nerves. Over the last few decades, the best surgical approaches for thymectomy have been controversial. The major drawbacks of the unilateral VATS thymectomy are the difficulty in completely removing the thymus and the bilateral mediastinal pleura and lipid and the risk of causing irreversible damage to the phrenic nerve on the opposite side. Compared with the unilateral VATS thymectomy, the bilateral VATS thymectomy may require longer operation times and more incisions (3). In the subxiphoid thymectomy, it is difficult to remove the lipid attached to the left cardiophrenic angle, which may cause an inevitable oppression of the heart during the operation (4). Moreover, lengthened ultrasonic scalpels and graspers and other specific instruments are needed for this process. In an effort to solve these problems, we explored the right thorax three-port VATS approach based on mediastinal pleura guidance and achieved the en bloc resection of the thymoma, thymus and bilateral mediastinum lipid. In addition, by taking advantage of this approach, the bilateral phrenic nerves and vessels were readily and clearly exposed in a manner that avoided injuries and bleeding.
SLET combined with artificial pneumothorax completely exposed the anterior mediastinum during the operation. Double-lumen endotracheal tube (DLET) with one-lung ventilation is not suitable for the unilateral VATS thymectomy approach because the left lung ventilation with a general tidal volume would inevitably interfere with the resection of the left mediastinal lipid and pleura. In addition, DLET has many other disadvantages, including more associated complications and increased procedural time and cost compared to SLET (5). Compared with DLET, SLET has fewer complications and is easier to perform. Previous research has shown that SLET with two-lung low tidal volume ventilation and artificial pneumothorax may be preferable to DLET, simultaneously satisfying the demands for anterior mediastinum exposure and effective ventilation (6).
This approach may be a safe and practicable treatment for thymoma patients with myasthenia gravis. This method has been performed in 15 cases. All the cases in our study were cured, and no deaths occurred in the perioperative period. The intraoperative blood loss varied from 43 to 80 mL, with an average of 50.2 mL. And the duration of the surgery varied from 76 to 120 min, with a mean time of 94 min. Our preliminary observations showed that the method might have a potential tendency on reducing the blood loss and the operative times. No relapse, no phrenic nerve paralysis and no signs of myasthenia gravis were observed at the 6-month follow-up for all the postoperative patients. However, given the limitations with small sample sizes and lacking control group, further case-controlled study is required to confirm the efficacy of this approach.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Informed Consent: Written informed consent was obtained from the patient for publication of this manuscript and any accompanying images.
References
- Tomaszek S, Wigle DA, Keshavjee S, et al. Thymomas: review of current clinical practice. Ann Thorac Surg 2009;87:1973-80. [Crossref] [PubMed]
- Guo K, Tong L, Li X, et al. The procedure of VATS completion thymectomy based on mediastinal pleura guidance. Asvide 2017;4:227. Available online: http://www.asvide.com/articles/1357
- Liu Z, Yang J, Lin L, et al. Unilateral video-assisted thoracoscopic extended thymectomy offers long-term outcomes equivalent to that of the bilateral approach in the treatment of non-thymomatous myasthenia gravis. Interact Cardiovasc Thorac Surg 2015;21:610-5. [Crossref] [PubMed]
- Suda T, Hachimaru A, Tochii D, et al. Video-assisted thoracoscopic thymectomy versus subxiphoid single-port thymectomy: initial results†. Eur J Cardiothorac Surg 2016;49 Suppl 1:i54-8. [PubMed]
- Sancheti MS, Dewan BP, Pickens A, et al. Thoracoscopy without lung isolation utilizing single lumen endotracheal tube intubation and carbon dioxide insufflation. Ann Thorac Surg 2013;96:439-44. [Crossref] [PubMed]
- Kim H, Kim HK, Choi YH, et al. Thoracoscopic bleb resection using two-lung ventilation anesthesia with low tidal volume for primary spontaneous pneumothorax. Ann Thorac Surg 2009;87:880-5. [Crossref] [PubMed]