Successful management of a giant dissecting aortic aneurysm in a patient with Behcet’s disease
Introduction
Behcet’s disease is a systemic vasculitis that is characterized by recurrent oral ulceration, genital ulcerations, skin lesions, vascular diseases and ocular lesions. This disease is commonly seen in Asian and Mediterranean countries and typically affects young adults. Compared to other vasculitis, Behcet’s disease can involve both arteries and veins of all sizes (1). Thoracic aortic aneurysm with subsequent dissection is a very rare complication that is associated with a high surgical risk and a poor prognosis (2). We report the case of a 20-year-old Chinese patient with Behcet’s disease who presented with a giant dissecting thoracic aortic aneurysm and was successfully managed using a complex surgical technique.
Case presentation
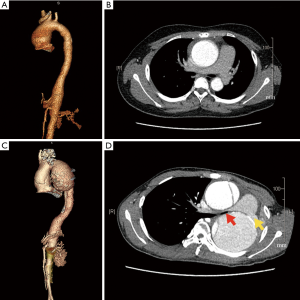
A 20-year-old male was referred to our center with a 1-week history of recurrent back pain. The patient’s physical examination was significant for systolic hypertension (170/80 mmHg) and a grade III/VI diastolic decrescendo murmur that was best heard at the left sternal border. During the first office visit four years prior, the patient presented with recurrent oral aphthae, disseminated folliculitis and a positive pathergy test, and was diagnosed with Behcet’s disease by a rheumatologist. Computed tomography angiography (CTA) at that time showed a dilated ascending aorta with a maximal diameter of 50 mm (Figure 1A,B). The patient denied surgical treatment for the ascending aorta dilation or the receipt of medical therapy to control his vasculitis, and had been lost to follow-up. The current CTA showed an extensive, dissecting thoracic aortic aneurysm (Figure 1C,D): the ascending aorta was 58 mm in diameter, and the descending aorta was 74 mm in diameter. The location of the primary entry tear was at the aortic arch level. The patient’s left pulmonary artery (Figure 1D, yellow arrow) and left main bronchus (Figure 1D, red arrow) were severely compressed by the aneurysm, leading to total atelectasis of the left lung. Transthoracic echocardiography (TTE) revealed a severely dilated left ventricle with a left ventricle end-diastolic diameter (LVEDD) of 69 mm, and severe aortic regurgitation with an aortic annulus diameter of 32 mm. The patient’s left ventricular ejection fraction (LVEF) was 41%.
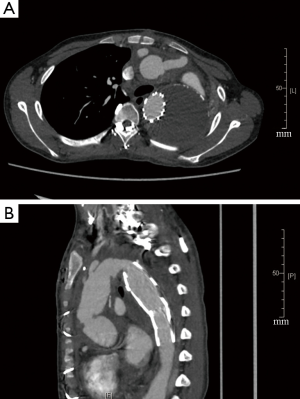
This patient was diagnosed with (I) Stanford type A aortic dissection; (II) ascending aortic aneurysm; (III) descending aortic aneurysm; (IV) severe aortic regurgitation; (V) Behcet’s disease. The patient underwent emergent surgery of aortic root and arch replacement using a frozen elephant trunk technique. The right femoral and right axillary arteries were cannulated first. The right atrium was cannulated after a standard median sternotomy was performed to initiate cardiopulmonary bypass. Anterograde cerebral perfusion was used during circulatory arrest. Intraoperatively, the patient’s aortic wall was found to be markedly thick which was consistent with chronic inflammatory reactions. Both the diseased aortic valve and the dilated ascending aorta were resected and replaced with a 23-mm mechanical valve (St. Jude Medical, Inc., Saint Paul, Minnesota, USA). A 26-mm aortic stent (Cronus, MicroPort, Shanghai, China) was deployed into the descending thoracic aorta after the dissected arch was resected. A 28/10/8/8/10*10 mm four-branched Gelweave graft (Vascutek Ltd., Inchinnan, Scotland, UK) was then anastomosed separately to the valve and to the stent in the descending aorta, using a 3–0 running polypropylene suture. Afterwards, the left common carotid artery, left subclavian artery and brachiocephalic artery were anastomosed to the four branches graft successively with a 5-0 running polypropylene suture. To ensure distal perfusion, an aortofemoral bypass was established with the perfusion branch of the graft via the subcutaneous tunnel. The patient’s postoperative course was uneventful. He was discharged on cyclophosphamide, prednisolone, warfarin, furosemide and spironolactone. Also, he was referred to a rheumatologist for follow-up medication adjustment. At the three-month follow-up, the patient was fully functional. The follow-up CTA revealed a false lumen thrombosis and no signs of leakage or pseudoaneurysm formation (Figure 2A,B). The TTE showed significantly a decreased LVEDD (55 mm) and an improved LVEF (55%). The patient reported that he was doing well and had no specific discomfort during the 10-month follow-up appointment.
Discussion
Because Behcet’s disease is a vasculitis that can involve all sizes of vessels in both the arterial and venous systems, screening for vascular system problems, including obtaining a detailed history, a careful physical examination, lab tests (compete blood count, erythrocyte sedimentation rate, and C-reactive protein test), and vascular imaging (CTA, vascular ultrasound, and magnetic resonance imaging), is essential to the work-up of Behcet’s disease. Ideally, the screening should be performed as soon as the diagnosis is made (or suspected) and should be repeated during the follow-up.
Arterial involvement is seen in 2.5% to 7% of all patients with Behcet’s disease, while thoracic aortic involvement is even more rare (1,3). Limited surgical experience is available regarding the management of thoracic aortic aneurysm or dissection in patients with Behcet’s disease. Our patient underwent a single-stage aortic root and total arch replacement with a frozen elephant trunk technique through a standard median sternotomy. This procedure effectively addressed his giant dissecting aneurysm, restored the blood supply to the abdominal aorta and lower extremities, and prevented the possible surgical difficulties introduce by a second-stage operation. The clamshell thoracotomy would have been an alternative approach for the management of this giant dissecting aneurysm case. Compared to a median sternotomy, the clamshell can provide better exposure to the entire thoracic aorta and has been demonstrated to be a safe and effective procedure for patients with extensive thoracic aortic disease (4). However, the clamshell approach is associated with relatively high respiratory insufficiency (4), making us hesitant to use it in this patient, who had ipsilateral lung atelectasis. Lateral thoracotomy was another option as it could easily expose the downstream descending aorta and might have been suitable if we had decided to replace the descending aorta with a Dacron graft. However, considering our patient was complicated by aortic root and ascending aorta dilation, we believe our surgical plan was more suitable for this patient.
Both anastomotic pseudoaneurysm and aneurysm recurrence are two fearful postoperative complications in patients with Behcet’s disease (5,6), and may be associated with a chronic inflammatory reaction. Cyclophosphamide and corticosteroids can effectively reduce aneurysm recurrence (7). Standard drug therapy, follow-up with a rheumatologist and regular cardiovascular imaging examinations are important for our patient’s long-term medical management.
Acknowledgements
Funding: This work was supported by the Fudan University Zhongshan Hospital Youth Research Funding (No. 2016ZSQN24 to Yi Lin and No. 2016ZSQN27 to Zhiqi Zhang).
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Informed Consent: Written informed consent was obtained from the patient for publication of this manuscript and any accompanying images.
References
- Tascilar K, Melikoglu M, Ugurlu S, et al. Vascular involvement in Behçet's syndrome: a retrospective analysis of associations and the time course. Rheumatology (Oxford) 2014;53:2018-22. [Crossref] [PubMed]
- Bardakci H, Kervan U, Boysan E, et al. Aortic arch aneurysm, pseudocoarctation, and coronary artery disease in a patient with Behçet's syndrome. Tex Heart Inst J 2007;34:363-5. [PubMed]
- Fei Y, Li X, Lin S, et al. Major vascular involvement in Behçet's disease: a retrospective study of 796 patients. Clin Rheumatol 2013;32:845-52. [Crossref] [PubMed]
- Doss M, Woehleke T, Wood JP, et al. The clamshell approach for the treatment of extensive thoracic aortic disease. J Thorac Cardiovasc Surg 2003;126:814-7. [Crossref] [PubMed]
- Hosaka A, Miyata T, Shigematsu H, et al. Long-term outcome after surgical treatment of arterial lesions in Behçet disease. J Vasc Surg 2005;42:116-21. [Crossref] [PubMed]
- Tuzun H, Seyahi E, Arslan C, et al. Management and prognosis of nonpulmonary large arterial disease in patients with Behçet disease. J Vasc Surg 2012;55:157-63. [Crossref] [PubMed]
- Liu Q, Ye W, Liu C, et al. Outcomes of vascular intervention and use of perioperative medications for nonpulmonary aneurysms in Behçet disease. Surgery 2016;159:1422-9. [Crossref] [PubMed]


