The value of delayed phase enhanced imaging in malignant pleural mesothelioma
Introduction
Magnetic resonance imaging (MRI) is frequently employed in the management of malignant pleural mesothelioma (MPM), most often to evaluate areas of suspected local invasion not fully characterized on computed tomography (CT) or fluorodeoxyglucose (FDG)-positron emission tomography (PET)/CT which are both limited in evaluation of these anatomic areas (1-4). Accurate characterization of local extent of MPM invasion is important since multifocal or full thickness invasion of the chest wall, diaphragm and pericardium is typically considered a contraindication to surgery. The excellent spatial resolution and tissue contrast of MRI affords an advantage over CT for this application (1,2). Yet an analysis of a large international database examining outcomes in patients with MPM undergoing pleurectomy revealed that up to 80% of MPM patients with clinical stage I and II and 22.8% of clinical stage III were upstaged (5) at surgery. Furthermore, MRI has not yet shown a definitive preoperative advantage for surgical treatment (2). By optimizing the tumor enhancement of MPM on both of these modalities, it may be possible to improve the diagnostic accuracy of MRI in preoperative staging.
Review of the radiologic literature reveals that published MRI protocols for MPM do not specify time delays following intravenous (IV) contrast administration (6-8). Furthermore, to our knowledge there is no published data exploring the kinetics of MPM enhancement beyond 2 min following IV contrast administration for the purpose of optimizing MPM enhancement. In this retrospective study, we examine the kinetics of MPM tumor enhancement on IV contrast enhanced MRI examinations to determine if optimal MPM enhancement may occur at a longer time delay than currently routinely acquired.
Methods
Institutional review board approval was obtained (IRB #822808) prior to the conduct of this study.
Patient population
A retrospective study was conducted of all adult patients with MPM who had undergone IV contrast-enhanced MRI at our institution between the years of 2000–2016. If the patients had undergone multiple MRI scans, only the initial exam was used for data analysis. Patients with exams that were non-diagnostic, unenhanced, non-measurable disease (less than 1 cm radial thickness of tumor) (9), incomplete MRI protocols (non-standard sequences or examples terminated prior to completion of the protocol) or studies without post-IV contrast images beyond 2 min were excluded from the study. In patients with more than one MRI examination, the exam with the longest post-IV contrast time delay was chosen.
MRI imaging
All of the magnetic resonance (MR) examinations were performed on either the 1.5T (MAGNETOM Avanto, MAGNETOM Espree, MAGNETOM Essenza) or 3T (MAGNETOM Skyra, MAGNETOM Verio) MRI scanners (SIEMENS, Munich, Germany). All studies included pre-contrast and post-contrast fat-saturated axial T1 gradient echo (GRE) sequences with 5 mm slice thickness, base resolution of 256, phase resolution of 75%, bandwidth of 610 (Hz/Px), and flip angle of 10. IV contrast was administered by hand injection using 0.7 mmol/kg of IV gadobenate dimeglumine (Multihance, Bracco Diagnostic Inc., Monroe Township, NJ, USA).
Quantitation of tumor enhancement
Using the clinical Centricity PACS (GE Healthcare, Little Chalfont, Buckinghamshire, UK) workstation, measurements of tumor signal were obtained at the same location on pre-contrast and all subsequent post-contrast phases in which the echo time (TE) and repetition time (TR) were fixed. The exact timing of image acquisition, as indicated on the images, relative to contrast injection was recorded. In three patients, the exact time of IV contrast bolus administration was not documented and an assumption of 30 sec post-IV contrast administration was made for the initial post-contrast image, as this is the standard imaging protocol at our institution. Time-enhancement curves were then generated for each lesion in order to assess peak tumor enhancement.
Conspicuity grading
In order to determine whether the quantitative maximal tumor enhancement corresponded with a measurable perceived difference by the radiologist, a qualitative comparison was made of tumor conspicuity for each imaging time point in the MRI exam. For each image used for quantitative measurement, a de-identified high-resolution image was downloaded for qualitative conspicuity grading. Six board-certified attending thoracic radiologists, each blinded to the image phase of enhancement, were presented sets of two side-by-side MRI images of a single tumor location obtained at different phases of IV contrast enhancement for a single MRI examination. The radiologists were asked to independently determine which of the two images had more conspicuous tumor relative to the surrounding anatomic structures. Every possible combination of phase of enhancement was provided for review at the selected tumor location. This was repeated for each patient examination in the study. Chi-squared statistical analysis was then performed to determine whether there was a correlation between differences in MRI phase of enhancement (signal intensity) and subjective assessment of tumor conspicuity.
Statistical analysis
For quantitative analysis of tumor enhancement, time-enhancement curves were generated. For enhancement curves demonstrating a point of peak enhancement and subsequent gradual decrease in signal intensity, theses curves were best modeled using a polynomial curve. For enhancement curves demonstrating a continued rise to a plateau, these curves were best modeled using a hyperbolic curve. A simulation of the predicted values was run out to 10 min. Calculations were made for estimated time points of tumor enhancement above 80%, 85%, and 90% of projected peak values over the evaluated time period and reported in frequencies. Chi-squared analysis was performed to determine the association between differences in tumor signal intensity and subjective conspicuity. An analysis was performed for lesions with differences in signal intensity of 0–10% and lesions with differences in signal intensity of >10%.
Results
Patient characteristics
Of the 42 patients that had MRIs for the evaluation of MPM in the specified time frame, a total of 12 patients fulfilled the criteria for our retrospective study. One patient was female and 11 patients were male. The average age of patients was 62, ranging from 53 to 74 years old. Of these patients, 11 had epithelioid histologic subtype and 1 patient had sarcomatoid histologic subtype.
Delayed peak signal intensity of mesothelioma
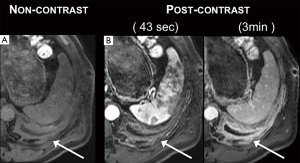
The enhancement curves of four patients demonstrated a point of peak enhancement with subsequent gradual decrease in signal intensity (Figure 1). The remaining eight patients continued to rise to a plateau that had not been reached (Figures 1,2) within the imaged time frame. Using best fit models and simulated projections of tumor enhancement over time, a time range between 150 and 300 sec was determined to be the most optimal time point for achieving near peak enhancement (Figure 3). Between 150 and 300 sec, 80% of patients are projected to have reached >80%, >85%, and >90% of peak projected signal intensity respectively.
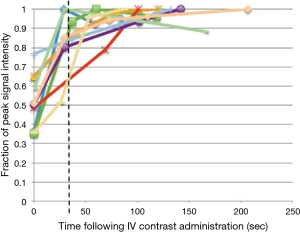
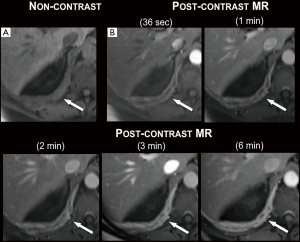
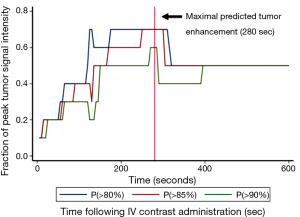
Correlation between increased signal intensity and increased conspicuity of the tumor
Quantitative increases in pleural tumor signal intensity on serial time delay MRI images corresponded to the perception of greater tumor conspicuity when viewed by thoracic radiologists (Figures 4,5). When sets of MR images of different phases of enhancement were compared side by side, tumors with 0–10% greater signal intensity, relative to the counterpart image, were qualitatively considered more conspicuous 55 out of 79 times. By comparison, the tumor of lesser signal intensity in the set was considered more conspicuous 24 out of 79 times. When the MR image set compared tumors with measured differences in signal intensity of >10%, thoracic radiologists found the tumor with the higher signal intensity more conspicuous 86 out of 87 times. By comparison, the tumor with lesser signal intensity more considered more conspicuous 1 out of 87 times. For both of the subsets analyzed, the correlation between higher tumor signal intensity and improved tumor conspicuity was statistically significant (P<0.01), although the impact was greater for lesions with a >10% difference in signal intensity.
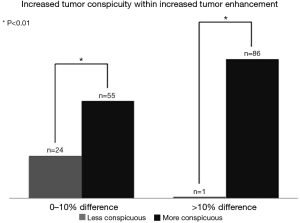
Discussion
In this study, we demonstrate evidence that the optimal time delay for maximal MPM tumor enhancement is likely between 150 and 300 sec following IV contrast administration on MRI. To our knowledge, there are no studies of MPM enhancement on MRI at time delays beyond 2 min. In our experience, MRI exams performed for MPM often do not include delayed phase imaging beyond 2 min following the administration of IV contrast with protocols. Our data suggests that MR imaging of MPM should routinely include imaging between 150–300 sec following IV contrast administration.
In this study, we also demonstrate that by increasing tumor enhancement on MRI, there was significantly improved tumor conspicuity appreciated by the observing radiologist. This association was most pronounced when there was more than a 10% increase in tumor enhancement. This supports the hypothesis that improving tumor enhancement on MRI may positively impact clinical interpretation of exams being performed for MPM by increasing conspicuity of the tumor lesion relative to surrounding anatomic structures. Since artifacts from respiratory motion can be a limitation when interpreting imaging of pleural tumor, future studies may be considered with methods, such as respiratory gating, to limit this source of potential impairment of lesion conspicuity on MRI. In this retrospective review, we excluded tumor lesions under 1 cm in radial thickness both to limit the impact of respiratory motion on tumor conspicuity and to minimize the impact of volume averaging on tumor measurements. Future studies would be important in evaluating the potential impact of improved lesion conspicuity demonstrated with increased IV contrast time delays on detection of small lesions less than 1 cm in thickness.
Given that this study is of a small sample size and is retrospective in nature, results should be considered preliminary. Future studies are needed to more clearly define the kinetics of MPM and the potential impact of optimal tumor enhancement on MR staging accuracy including an assessment of the intra- and inter-variability in radiologic assessments of perceived tumor conspicuity. A larger sample size will also allow for assessment for potentially differential patterns of enhancement based on histology. In this study, 10 of the 11 patients were of the more common epithelioid subtype with one patient having sarcomatoid histology. Future studies, statistically powered to evaluate the enhancement patterns across MPM histological subtypes, will be necessary to validate the kinetics of MPM enhancement on MRI before adoption of this time delay can be recommended for clinical use.
In summary, these results suggest that IV contrast enhanced MRI imaging protocols of MPM should include a delayed phase time point between 150 and 300 sec for optimal tumor imaging. Future prospective studies are needed to validate the kinetics of MPM enhancement prior to adoption of this delayed phase imaging for clinical use and to assess the potential added value of this technique for staging of mesothelioma.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: Institutional review board approval was obtained (IRB #822808) prior to the conduct of this study.
References
- Patz EF Jr, Shaffer K, Piwnica-Worms DR, et al. Malignant pleural mesothelioma: value of CT and MR imaging in predicting resectability. AJR Am J Roentgenol 1992;159:961-6. [Crossref] [PubMed]
- Heelan RT, Rusch VW, Begg CB, et al. Staging of malignant pleural mesothelioma: comparison of CT and MR imaging. AJR Am J Roentgenol 1999;172:1039-47. [Crossref] [PubMed]
- Flores RM, Akhurst T, Gonen M, et al. Positron emission tomography defines metastatic disease but not locoregional disease in patients with malignant pleural mesothelioma. J Thorac Cardiovasc Surg 2003;126:11-6. [Crossref] [PubMed]
- Pilling J, Dartnell JA, Lang-Lazdunski L. Integrated positron emission tomography-computed tomography does not accurately stage intrathoracic disease of patients undergoing trimodality therapy for malignant pleural mesothelioma. Thorac Cardiovasc Surg 2010;58:215-9. [Crossref] [PubMed]
- Rusch VW, Giroux D, Kennedy C, et al. Initial analysis of the international association for the study of lung cancer mesothelioma database. J Thorac Oncol 2012;7:1631-9. [Crossref] [PubMed]
- Martini K, Meier A, Opitz I, et al. Diagnostic accuracy of sequential co-registered PET+MR in comparison to PET/CT in local thoracic staging of malignant pleural mesothelioma. Lung Cancer 2016;94:40-5. [Crossref] [PubMed]
- Gill RR, Umeoka S, Mamata H, et al. Diffusion-weighted MRI of malignant pleural mesothelioma: preliminary assessment of apparent diffusion coefficient in histologic subtypes. AJR Am J Roentgenol 2010;195:W125-30. [Crossref] [PubMed]
- Razek AA. Diffusion magnetic resonance imaging of chest tumors. Cancer Imaging 2012;12:452-63. [Crossref] [PubMed]
- Tsao AS, Garland L, Redman M, et al. A practical guide of the Southwest Oncology Group to measure malignant pleural mesothelioma tumors by RECIST and modified RECIST criteria. J Thorac Oncol 2011;6:598-601. [Crossref] [PubMed]


