Effectiveness of catheter ablation of atrial fibrillation: are we at the dawn of a new era?
It is known that atrial fibrillation (AF), the most common supraventricular cardiac arrhythmia (1), is associated with increased mortality and morbidity, with cerebral thromboembolism representing the most serious and threatening complication. Therefore, together with the adoption of either rate control—decreasing ventricular response by atrioventricular nodal-blocking drugs—or rhythm control strategy—restoring sinus rhythm by anti-arrhythmic drugs (AADs) and/or atrial fibrillation transcatheter ablation (AFTCA), oral anticoagulation therapy (OAT) is mandatory for all patients with high thromboembolic risk (CHA2DS2-VASc ≥3 for females and ≥2 for males) (2).
Recently, Saliba et al. (3) published the results of a large retrospective, cohort study comparing the occurrence of stroke/transient ischemic attack (TIA) and mortality in AF patients treated by primary catheter ablation (ablation cohort) compared to the remaining patients (non-ablation cohort), retrieving data from the Israel population-based electronic medical records (EMRs) database. From an initial sample of 43,041 adult AF patients (1,015 in the ablation cohort; 42,026 in the non-ablation cohort), a propensity score-matched sample of 4,741 patients (969 in the ablation group; 3,772 in the non-ablation group) was analysed. The cumulative incidence of stroke/TIA was significantly lower in the ablation group (P=0.002), with an incidence rate of 2.1 compared to 3.26 per 100 person-years in the non-ablation group. The corresponding CHA2DS2-VASc adjusted HR was 0.58 (95% CI, 0.43–0.72) for patients in the ablation group with respect to non-ablation group. Similarly, a reduction in the cumulative incidence of mortality was found (P<0.0001), with a CHA2DS2-VASc adjusted HR of 0.57 (95% CI, 0.47–0.66) in the ablation group compared to non-ablation group.
These results are intriguing and surely highlight the potential benefits deriving from AFTCA on a real-life setting. In particular, dealing with AFTCA effectiveness in decreasing stroke/TIA and mortality with respect to medically managed AF patients (on AADs or rate control drugs), this study supports AFTCA as first line approach in AF management. Even considering the limitations imposed by the observational nature of the study, the message conveyed is that AFTCA, compared to rate control or rhythm control by AADs, can decrease by 40% the risk of mortality and stroke/TIA. Translating this concept in terms of number needed to treat (NNT), we could affirm that in our clinical practice, by treating less than three AF patients by AFTCA (NNT 2.7) we avoid one cerebrovascular accident or a death. In some way it is even possible that this study, due to study design, has underestimated the impact of AFTCA on stroke/TIA and mortality. In fact, in the study of Saliba et al. the acute and long-term procedural success rate of AFTCA was not available. For this reason, patients with unsuccessful AFTCA have been anyway considered in the ablation group, trimming down the measure of AFTCA-related reduction in cerebrovascular accidents and mortality. In addition, the included population is characterized by a high thromboembolic risk, as documented by the post propensity match mean CHA2DS2-VASc score of 3.6 in both ablation and non-ablation group. It is plausible that, in AF patients with a lower (CHA2DS2-VASc =0/1) thromboembolic risk, in whom AF is more commonly the solely thromboembolic risk factor, AFTCA impact on the reduction of stroke/TIA and mortality would result even greater.
“Rate or rhythm control for atrial fibrillation?”. This is the vexed clinical dilemma that afflicts cardiologists when facing the most common cardiac arrhythmia. Until about ten years ago, “rate vs rhythm control” was purely a matter of “nodal-blocking drugs vs AADs”. In this pre-AFTCA era, two ground-breaking studies tried to solve the dilemma: the RACE (4) and the AFFIRM study (5). In both studies, rhythm control strategy with AADs did not offer any survival advantage over rate control strategy. On the contrary, rhythm control strategy was characterized by more complications and hospitalisations, due to the intrinsic toxicities of AADs. However, successive observational studies suggested that rhythm-control therapy with AADs could be superior to rate control, with the benefit emerging only on the long run, both concerning mortality (6) than cerebrovascular events (7). In a study by Ionescu-Ittu et al. (6), the mortality rate in a rhythm control group decreased steadily with respect to the rate control group only after the first 5 years of follow up (during which no differences between medical rhythm control and rate control therapy were found). A likely explanation of this phenomenon is that during the first years of medical rhythm control therapy the potential toxicities of the AADs could balance the intrinsic survival advantage granted by rhythm control itself, resulting in no net survival advantages of the rhythm over rate control.
In the last decade, instead, AFTCA, introduced in clinical practice at the end of the past century, reached a wide diffusion and a recognised role as an effective non-medical rhythm control approach. The rise of this procedure reignited the never faded debate on AF management, in particular since AFTCA is devoid of the intrinsic toxicities of AADs and is superior over AADs in maintaining sinus rhythm (8,9). To date, only few observational studies have assessed the effect of AFTCA on long-term outcomes such as stroke/TIA and mortality (10-13). All these studies indicated a reduced burden of mortality and cerebrovascular events in AF patients treated with AFTCA, but, in addition to the observational design (due to which certain conclusions may not be taken), they are characterized by limited generalizability. In this context, the study from Saliba et al. (3), by providing real clinical practice data with strong generalizability, adds significant data. In any case, to prove a cause-effect relationship in favour of AFTCA in terms of mortality and stroke/TIA we need to wait for large randomized clinical trials (RCTs) data. In the next few years, an ongoing RCT will probably provide the medical community with the elements to solve this clinical dilemma: the “Catheter Ablation vs Anti-arrhythmic Drug Therapy for Atrial Fibrillation Trial (CABANA)” (14). This study was designed to prove that AFTCA is superior to both rate and rhythm control drug therapy in AF patients by decreasing the incidence of the composite endpoint of total mortality, disabling stroke, serious bleeding, or cardiac arrest. Should the results support the superiority of the former, AFTCA will probably become the first line treatment of AF. In the meantime, the late-breaking results of a completed RCT, the “Catheter Ablation vs. Standard Conventional Treatment in Patients With LV Dysfunction and AF (CASTLE-AF)” (15), were recently presented at the European Society of Cardiology (ESC) 2017 Congress. In this trial, confirming previous non-randomized evidences (16), AFTCA, in patients with AF and concomitant systolic heart failure (HF), provides significant benefit with respect to conventional drug treatment (rhythm and rate control drugs) in terms of mortality (HR, 0.53; 95% CI: 0.32–0.86) and hospitalisation for HF (HR, 0.56; 95% CI: 0.37–0.83).
Hence, waiting for evidence on the long-term impact of AFTCA in the general AF population, as clear as for the subpopulation of AF patients with concomitant HF, the following issues weigh consideration.
OAT discontinuation after AFTCA
Proven that the benefit of OAT for thromboembolic risk reduction is considered to outweigh the measurable risk of bleeding in most AF patients, it is still debated whether OAT should be continued after an apparently successful AFTCA. Due to lack of systematic evidence, current guidelines recommend that decision of maintaining OAT after catheter ablation should follow general anticoagulation recommendations, regardless of the presumed rhythm outcome (2). Anyway, growing observational data hint that in several patients the risk of bleeding could outweigh the risk of thromboembolic events, thus favouring OAT discontinuation after a successful AFTCA. A recent study from Karasoy et al. (17) demonstrated that OAT discontinuation was not associated with significantly increased thromboembolic risk neither in low- nor in high-risk patients, while OAT maintenance was associated with higher incidence rates of serious bleeding compared with OAT discontinuation. An Italian multicentre observational study confirmed these findings (18): 1,500 patients were retrospectively divided into three groups: group A—AFTCA maintaining antivitamin K (AVK); group B—AFTCA discontinuing AVK; group C—rate control and AVK. Thromboembolic events did not differ between the groups (5/500, 1% group A; 7/500, 1.4% group B; 11/500, 2.2% group C; P=0.45), while haemorrhagic events were greater in group A (9/500, 1.8%) and C (12/500, 2.4%) than in group B (no events; P=0.003). Thus, in this experience, OAT maintenance with antivitamin K (AVK) following AFTCA eventually conferred a haemorrhagic risk greater to the thromboembolic protective effect. RCTs are mandatory to support these findings, possibly paving the way for a standardised approach first to detect AF following the procedure, and, consequently, decide which AF patient could stop OAT in order to prevent the extra-risk of bleedings. In this sense, the work from Saliba et al. (3) lacks details regarding OAT prosecution after AFTCA: in particular, it would have been interesting, in such a large study, to assess among the patients in the ablation group the distribution of cerebral ischemic events stratified by OAT recommendation.
Timing of AFTCA
Another relevant and yet unanswered issue regards the proper time to perform AFTCA. Waiting for ongoing RCTs which will surely shed light on this issue, an observational study, although presenting the bias of having been performed in our “highly motivated AFTCA rhythm control centre”, clearly indicates that the long-term progression from non-permanent (paroxysmal and persistent) to permanent AF is reduced if AFTCA is performed early (paroxysmal phase) in the natural history of the disease (19): in fact, after a median follow-up of 64 months, AF progression to a permanent form was documented in 2.7%, 10.0% and 14.6% of patients with respectively baseline paroxysmal, persistent and long-standing persistent AF. Also after very long term follow up (10 years) this trend is confirmed with a reduced recurrence rate in patients with baseline paroxysmal over persistent AF (20). Given that long arrhythmia duration is known to cause consequent structural remodelling of the left atrial substrate, increased tissue fibrosis and intra-atrial conduction disturbances perpetuating the arrhythmia, this finding is quite intuitive, however it is not, to date, reflected in clear clinical recommendations. As earlier AFTCA is performed the better it would be for AF patients in respect to relevant long-term outcomes, such as death and stroke; this statement should not remain just an intriguing hypothesis.
AF and dementia
In the past few years, it has emerged that AF, even in absence of clinically relevant ictus or TIAs, relates to an increased risk of dementia/cognitive decline (21). Different mechanisms have been proposed to explain this independent association, such as silent cerebral ischemia (SCI), cerebral microbleeds (CMBs) and an altered cerebral blood flow during AF. Our group previously demonstrated that patients with paroxysmal and persistent AF have a higher prevalence and number of areas of SCI—small hyperintense T2-weighted cerebral MR lesions—than healthy individuals and this related to worse cognitive performance than subjects in sinus rhythm (22), possibly due to cardiac microembolisms. An increase burden of CMBs can also be involved in the genesis of the AF-related cognitive impairment, in particular in those patients on OAT (23). Again, understanding whether and when OAT could be stopped in AF patients with sinus rhythm restoration would help in preventing this extra brain damage. Finally, AF per se causes an altered cerebral blood flow. We recently demonstrated in a computational study that AF induces transient hypoperfusions and hypertensive events in the cerebral circulation (24,25), suggesting that the chronic occurrence of such critical hemodynamic events explains the genesis of a subgroup of non-microembolic SCI and non-OAT-related CMBs. In this sense, considering that AF rhythm status is independently associated with an increased risk of cognitive impairment/dementia, a procedure such as AFTCA, characterized by the lowest number of AF recurrences in the long-term especially when performed early in the history of the disease, may surely prevent, compared to a rate control strategy, the proportion of brain damage directly related to the AF-induced cerebral hemodynamics.
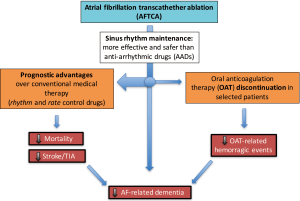
In conclusion, in addition to the superiority in maintaining sinus rhythm when compared to AADs, growing clinical evidence points towards long-term protective effects of AFTCA in terms of mortality and cerebral thromboembolic events (Figure 1). Should the previous several observational findings be confirmed by the ongoing RCTs, AFTCA will become the mainstay first-step therapy for AF.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
- Staerk L, Sherer JA, Ko D, et al. Atrial Fibrillation. Circ Res 2017;120:1501-17. [Crossref] [PubMed]
- Kirchhof P, Benussi S, Kotecha D, et al. 2016 ESC Guidelines for the management of atrial fibrillation developed in collaboration with EACTS. Europace 2016;18:1609-78. [Crossref] [PubMed]
- Saliba W, Schliamser JE, Lavi I, et al. Catheter ablation of atrial fibrillation is associated with reduced risk of stroke and mortality: A propensity score–matched analysis. Heart Rhythm 2017;14:635-42. [Crossref] [PubMed]
- Wyse DG, Waldo AL, DiMarco JP, et al. A comparison of rate control and rhythm control in patients with atrial fibrillation. N Engl J Med 2002;347:1825-33. [Crossref] [PubMed]
- Investigators A. Relationships between sinus rhythm, treatment, and survival in the Atrial Fibrillation Follow-Up Investigation of Rhythm Management (AFFIRM) Study. Circulation 2004;109:1509-13. [Crossref] [PubMed]
- Ionescu-Ittu R, Abrahamowicz M, Jackevicius CA, et al. Comparative effectiveness of rhythm control vs rate control drug treatment effect on mortality in patients with atrial fibrillation. Arch Intern Med 2012;172:997-1004. [Crossref] [PubMed]
- Tsadok MA, Jackevicius CA, Essebag V, et al. Rhythm versus rate control therapy and subsequent stroke or transient ischemic attack in patients with atrial fibrillation. Circulation 2012;126:2680-7. [Crossref] [PubMed]
- Mont L, Bisbal F, Hernández-Madrid A, et al. Catheter ablation vs. antiarrhythmic drug treatment of persistent atrial fibrillation: a multicentre, randomized, controlled trial (SARA study). Eur Heart J 2014;35:501-7. [Crossref] [PubMed]
- Morillo CA, Verma A, Connolly SJ, et al. Radiofrequency ablation vs antiarrhythmic drugs as first-line treatment of paroxysmal atrial fibrillation (RAAFT-2): a randomized trial. JAMA 2014;311:692-700. [Crossref] [PubMed]
- Bunch TJ, Crandall BG, Weiss JP, et al. Patients treated with catheter ablation for atrial fibrillation have long-term rates of death, stroke, and dementia similar to patients without atrial fibrillation. J Cardiovasc Electrophysiol 2011;22:839-45. [Crossref] [PubMed]
- Lin YJ, Chao TF, Tsao HM, et al. Successful catheter ablation reduces the risk of cardiovascular events in atrial fibrillation patients with CHA2DS2-VASc risk score of 1 and higher. Europace 2013;15:676-84. [Crossref] [PubMed]
- Hunter RJ, McCready J, Diab I, et al. Maintenance of sinus rhythm with an ablation strategy in patients with atrial fibrillation is associated with a lower risk of stroke and death. Heart 2012;98:48-53. [Crossref] [PubMed]
- Reynolds MR, Gunnarsson CL, Hunter TD, et al. Health Outcomes With Catheter Ablation or Antiarrhythmic Drug Therapy in Atrial Fibrillation. Circ Cardiovasc Qual Outcomes 2012;5:171-81. [Crossref] [PubMed]
- ClinicalTrials.gov. Catheter Ablation vs Anti-arrhythmic Drug Therapy for Atrial Fibrillation Trial (CABANA). Available online: https://clinicaltrials.gov/ct2/show/NCT00911508
- Catheter ablation improves outcomes in patients with heart failure and atrial fibrillation (CASTLE-AF). Available online: https://www.escardio.org/The-ESC/Press-Office/Press-releases/catheter-ablation-improves-outcomes-in-patients-with-heart-failure-and-atrial-fibrillation
- Anselmino M, Matta M, D'ascenzo F, et al. Catheter ablation of atrial fibrillation in patients with left ventricular systolic dysfunction: a systematic review and meta-analysis. Circ Arrhythm Electrophysiol 2014;7:1011-8. [Crossref] [PubMed]
- Karasoy D, Gislason GH, Hansen J, et al. Oral anticoagulation therapy after radiofrequency ablation of atrial fibrillation and the risk of thromboembolism and serious bleeding: long-term follow-up in nationwide cohort of Denmark. European heart journal 2015;36:307-15. [Crossref] [PubMed]
- Gallo C, Battaglia A, Anselmino M, et al. Long-term events following atrial fibrillation rate control or transcatheter ablation: a multicenter observational study. J Cardiovasc Med (Hagerstown) 2016;17:187-93. [Crossref] [PubMed]
- Scaglione M, Gallo C, Battaglia A, et al. Long-term progression from paroxysmal to permanent atrial fibrillation following transcatheter ablation in a large single-center experience. Heart Rhythm 2014;11:777-82. [Crossref] [PubMed]
- Gaita F, Scaglione M, Battaglia A, et al. Very long-term outcome following transcatheter ablation of atrial fibrillation. Are results maintained after 10 years of follow up? Europace 2017. [Epub ahead of print]. [Crossref] [PubMed]
- Hui DS, Morley JE, Mikolajczak PC, et al. Atrial fibrillation: A major risk factor for cognitive decline. Am Heart J 2015;169:448-56. [Crossref] [PubMed]
- Gaita F, Corsinovi L, Anselmino M, et al. Prevalence of silent cerebral ischemia in paroxysmal and persistent atrial fibrillation and correlation with cognitive function. J Am Coll Cardiol 2013;62:1990-7. [Crossref] [PubMed]
- Bunch TJ, May HT, Bair TL, et al. Atrial Fibrillation Patients Treated With Long-Term Warfarin Anticoagulation Have Higher Rates of All Dementia Types Compared With Patients Receiving Long-Term Warfarin for Other Indications. J Am Heart Assoc 2016;5:e003932. [Crossref] [PubMed]
- Anselmino M, Scarsoglio S, Saglietto A, et al. Transient cerebral hypoperfusion and hypertensive events during atrial fibrillation: a plausible mechanism for cognitive impairment. Sci Rep 2016;6:28635. [Crossref] [PubMed]
- Scarsoglio S, Saglietto A, Anselmino M, et al. Alteration of cerebrovascular haemodynamic patterns due to atrial fibrillation: an in silico investigation. J R Soc Interface 2017;14:20170180. [Crossref] [PubMed]


