Operative or non-operative treatment of congenital tracheal stenosis: is there something new?
In the 2015, 8th issue of the International Journal of Clinical and Experimental Medicine, HONG et al. (1) focus on the non-operative treatment of congenital tracheal stenosis (CTS) associated with left pulmonary artery sling (PAS). The authors support no tracheal intervention in most of the patients with this rare malformation (the “ring-sling” complex) due to the severe complications and bad outcome observed in their short experience (14 cases in 5 years). Slide tracheoplasty or tracheal stenting were the procedures used in 7 patients of the series with only one reported survivor (14.2%). On the other side, 7 patients with PAS and CTS underwent re-implantation of the left pulmonary artery (LPA) with no tracheal intervention, and a good outcome was achieved in every case (100%). According to their data, there was no significant difference in the narrowest diameter of the stenotic trachea between survivors without tracheal procedures and those needing tracheal intervention (1). Regarding the length of the stenotic tracheal segment, it was significantly longer in patients requiring tracheal interventions (surgery or stenting) compared to those in whom these procedures were not done. In order to select the most appropriate candidates for CTS non-operative treatment, the authors propose an indicator based on a particular ratio: narrowest tracheal diameter/stenosis length to total tracheal length (diameter/length %). According to their study, those patients with a high ratio, above 10–11, would be good candidates for non-operative treatment while those clearly below this index would eventually require a tracheal intervention.
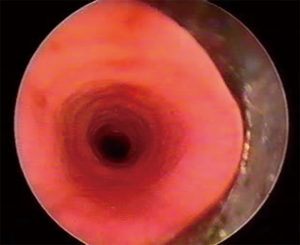
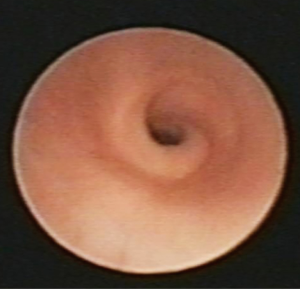
CTS is a very rare disease estimated to affect one in 64,500 live births. Between 60% and 70% of children born with CTS have other associated malformations, particularly of the cardiovascular type (2). One of the most constant associated anomaly is the left PAS which is present in approximately 50% of cases. Complete tracheal rings are almost inherent to CTS except for other very rare tracheal anomalies like complete tracheal cartilaginous sleeve or corkscrew trachea (3,4) (Figures 1,2). Albeit the aberrant course of the LPA, between the trachea and the esophagus, in its way to the left lung, CTS is generally considered an intrinsic anomaly thus extrinsic vascular compression is probably not responsible for the decreased tracheal lumen. In this setting, re-implantation of the LPA without addressing the tracheal anomaly will probably be inadequate in most of the cases (2,5). Selection of candidates for tracheal reconstruction is a crucial issue. Several aspects may be considered for this purpose: narrowest diameter, length of the stenosis, clinical symptoms and associated malformations, may all play a role. In 2003 (3), we proposed a clinical classification that has proved to be useful in the therapeutic decision-making. In fact, the overall experience in managing CTS has demonstrated that a strong correlation exists between the morphologic type and the functional or clinical category (6,7). There seems to be general agreement that patients with moderate or severe respiratory symptoms developing in the neonatal period or later on, near the end of the first year of life when physical activity increases, should be operated. Associated cardiovascular anomalies, most frequently left PAS, should be repaired during the same procedure (2,7,8). Of course, tracheal diameter and other bronchoscopic findings such as: length of the stenotic segment, presence of a tracheal bronchus, or diverse anatomical variants, may play a role too in the management strategy of CTS. Some children may show minimal clinical symptoms due to a mild tracheal stenosis with no associated malformations (group I A) (3). In this setting, diagnosis can be delayed or made incidentally when the child undergoes a surgical procedure for another condition. This selected group of patients is probably a minority and can be followed in an expectant way, eventually not requiring surgical reconstruction. Nevertheless, most of the patients fall into clinical or functional groups II and III (3) and should be first stabilized, then a complete diagnostic work-up done and finally operated as soon as possible. All this clinical care pathway should ideally be performed in a referral center with a multidisciplinary approach (5).
Surgical procedures for CTS repair fall into three categories: (I) tracheal reconstruction with autologous tracheal tissue (tracheal resection, slide tracheoplasty, and free tracheal autograft); (II) tracheoplasty with nontracheal autologous tissue (costal cartilage or pericardial patch); and (III) tissue-engineered tracheal replacement (5). Surgical treatment of CTS has evolved considerably for the last two decades and slide tracheoplasty, on cardiopulmonary bypass, has emerged as the “gold standard” technique (2,5,9,10). Increasing experience with this procedure has dramatically improved the outcomes of CTS, in contrast to what Hong et al. (1) describe in their article. This surgical technique is effective, versatile and its indications are expanding from long segment CTS to short segment CTS, tracheal stenosis with bronchial extension, and even other tracheal diseases such as recurrent tracheoesophageal fistula (11,12). Moreover, its main advantage lays on the fact that reconstruction is performed using native tracheal tissue so granulation tissue formation is minimal compared to other surgical techniques using cartilage or pericardial grafts (5). Tracheal growth after slide tracheoplasty has been clinically demonstrated (13).
Although still controversial, there is increasing experience with airway stenting in children (14-17). Tracheobronchomalacia and airway stenosis are the most frequent indications for stenting when other therapeutic options have failed or there are no other surgical alternatives. In most cases, stents are used as a life-saving procedure in extremely sick children. In CTS stents are generally used as a temporary measure after surgical reconstruction or in the management of postsurgical complications (7,18). Although some authors describe initial treatment of CTS with metallic stents (19,20), this approach is not shared by most tracheal teams (5,7,10,11). Hong et al. (1) report the use of stents, probably metal mesh although it is not stated in their manuscript, as a first line intervention in three patients with CTS. This is probably not the best way to deal with symptomatic CTS as their results show (no survival).
In summary, CTS is an infrequent malformation that includes a wide variety of anatomical and clinical presentations. Although the tracheal diameter/length (%) ratio may be a useful indicator for the need of surgical reconstruction, the clinical situation of the child remains the most important issue in the therapeutic algorithm. Slide tracheoplasty is a highly successful technique and is recommended as the treatment of choice for symptomatic patients with CTS. Airway stenting may be useful in the management of postsurgical complications but cannot be considered a definite or first line treatment. Previously considered a fatal disease, CTS is currently a treatable malformation with a good outcome in experienced units. Nevertheless, management of patients with CTS is complex and an interdisciplinary team approach in referral centers is of paramount importance.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
- Hong X, Zhou G, Liu Y, et al. Management of pulmonary artery sling with tracheal stenosis: LPA re-implantation without tracheoplasty. Int J Clin Exp Med 2015;8:2741-7. [PubMed]
- Hewitt RJ, Butler CR, Maughan EF, et al. Congenital tracheobronchial stenosis. Semin Pediatr Surg 2016;25:144-9. [Crossref] [PubMed]
- Antón-Pacheco JL, Cano I, García A, et al. Patterns of management of congenital tracheal stenosis. J Pediatr Surg 2003;38:1452-8. [Crossref] [PubMed]
- Antón-Pacheco JL, López M, Moreno C, et al. Congenital tracheal stenosis caused by a new tracheal ring malformation. J Thorac Cardiovasc Surg 2011;141:e39-40. [Crossref] [PubMed]
- Antón-Pacheco JL, Cano I, Comas J, et al. Management of of tracheal stenosis in infancy. Eur J Cardiothorac Surg 2006;29:991-6. [Crossref] [PubMed]
- Herrera P, Caldarone C, Forte V, et al. The current state of congenital tracheal stenosis. Pediatr Surg Int 2007;23:1033-44. [Crossref] [PubMed]
- Hofferberth SC, Watters K, Rahbar R, et al. Management of Congenital Tracheal Stenosis. Pediatrics 2015;136:e660-9. [Crossref] [PubMed]
- Elliott M, Roebuck D, Noctor C, et al. The management of congenital tracheal stenosis. Int J Pediatr Otorhinolaryngol 2003;67:s183-92. [Crossref] [PubMed]
- Manning PB, Rutter MJ, Border WL. Slide tracheoplasty in infants and children: risk factors for prolonged postoperative ventilatory support. Ann Thorac Surg 2008;85:1187-91; discussion 1191-2. [Crossref] [PubMed]
- Butler CR, Speggiorin S, Rijnberg FM, et al. Outcomes of slide tracheoplasty in 101 children: a 17-year single-center experience. J Thorac Cardiovasc Surg 2014;147:1783-9. [Crossref] [PubMed]
- Manning PB, Rutter MJ, Lisec A, et al. One slide fits all: the versatility of slide tracheoplasty with cardiopulmonary bypass support for airway reconstruction in children. J Thorac Cardiovasc Surg 2011;141:155-61. [Crossref] [PubMed]
- Provenzano MJ, Rutter MJ, von Allmen D, et al. Slide tracheoplasty for the treatment of tracheoesophogeal fistulas. J Pediatr Surg 2014;49:910-4. [Crossref] [PubMed]
- Speggiorin S, Gilbert TW, Broadhead M, et al. Do tracheas grow after slide tracheoplasty? Ann Thorac Surg 2012;93:1083-6. [Crossref] [PubMed]
- Antón-Pacheco JL. Tracheobronchial stents in children. Semin Pediatr Surg 2016;25:179-85. [Crossref] [PubMed]
- Nicolai T. Airway stents in children. Pediatr Pulmonol 2008;43:330-44. [Crossref] [PubMed]
- Fayon M, Donato L, de Blic J, et al. French experience of silicone tracheobronchial stenting in children. Pediatr Pulmonol 2005;39:21-7. [Crossref] [PubMed]
- Serio P, Fainardi V, Leone R, et al. Tracheobronchial obstruction: follow-up study of 100 children treated with airway stenting. Eur J Cardiothorac Surg 2014;45:e100-9. [Crossref] [PubMed]
- Antón-Pacheco JL, Comas JV, Luna C, et al. Treatment strategies in the management of severe complications following slide tracheoplasty in children. Eur J Cardiothorac Surg 2014;46:280-5. [Crossref] [PubMed]
- Maeda K, Yasufuku M, Yamamoto T. A new approach to the treatment of congenital tracheal stenosis: Balloon tracheoplasty and expandable metallic stenting. J Pediatr Surg 2001;36:1646-9. [Crossref] [PubMed]
- Xu X, Li D, Zhao S, et al. Treatment of congenital tracheal stenosis by balloon-expandable metallic stents in paediatric intensive care unit. Interact Cardiovasc Thorac Surg 2012;14:548-50. [Crossref] [PubMed]