Effect of a passive exhalation port on tracheostomy ventilation in amyotrophic lateral sclerosis patients: a randomized controlled trial
Introduction
Amyotrophic lateral sclerosis (ALS; also called motor neuron disease) is a devastating disease leading to progressive weakness affecting the limbs and the bulbar muscles. The most frequent cause of death in patients with ALS is ventilatory failure secondary to respiratory muscle weakness (1). Although non-invasive ventilation (NIV) combined with assisted coughing techniques can be an effective strategy to manage respiratory complications that develop during advanced stages of the disease (2), it cannot be suggested for patients with severe bulbar involvement who are unable to adequately protect their airways or those who require round-the-clock ventilatory assistance (3). In those cases, long-term tracheostomy ventilation (LT-TV) can be provided by a portable ventilator through an open single-limb respiratory circuit with an active non-rebreathing exhalation valve (e.g., a diaphragm or a balloon valve driven by ventilator pressure). A single-limb circuit with an active expiratory valve, in fact, combines the practicality of a single tube while ensuring carbon dioxide (CO2) removal (4). An active exhalation valve could nevertheless inadvertently generate a high expiratory resistance increasing the intrinsic positive end-expiratory pressure (PEEP) and effort during expiration. It could also raise the peak airway pressure to a level that could cause discomfort or even barotraumas if the inspiratory phase of ventilator is excessively long or the patient coughs (5).
A single-limb respiratory circuit with a Whisper Swivel connector that vents expiratory gas into the atmosphere was designed in the early ‘90s to administer NIV ventilatory support via a bilevel positive airway pressure system (6). A number of “vented” or intentional leak respiratory systems are now available for NIV treatment that ensure CO2 elimination by means of a mask with an integrated leak, an exhalation port connector or a “plateau valve” positioned between the circuit and the mask (7). These new breathing systems aim to improve gas flow during expiration by reducing expiratory resistance and the work of breathing.
Portable ventilators utilizing an intentional leak system and connecting the circuit to a tracheostomy tube have recently been made available. Although representing an attractive alternative to a conventional exhalation valve, “vented” systems for tracheostomized patients have not gained popularity in view of safety issues, in particular, in connection to the problem of rebreathing with CO2 retention (8).
In view of these considerations and the dearth of clinical studies, we set out to investigate the effect of a passive exhalation port on gas exchange in tracheostomized ALS patients. It was our hypothesis that the effect of a passive exhalation port on CO2 retention would be similar to that of a standard exhalation valve. Given the importance of patient comfort and care-giver satisfaction in neuromuscular patients receiving long-term ventilatory assistance (9), the impact of a passive exhalation port on the patients’ respiratory symptoms and on caregiver burden was also evaluated.
Methods
A pragmatic, single-center, two-armed parallel-group randomised controlled trial (RCT) was carried out at the Respiratory Pathophysiology Division of the University-City Hospital of Padova to evaluate the clinical outcome of ten tracheostomized ALS patients receiving ventilation via a passive exhalation port circuit (group A). The outcomes of these patients were compared with those of ten tracheostomized ALS subjects who were ventilated using a single-limb respiratory circuit with an active exhalation valve (group B). The participants were recruited between 1st January 2014 and 31st December 2015.
Ethics statement
The study design was approved by the Institutional Review Board of Padova City Hospital (No. 2145p; date of approval: December 6, 2012), and the study was carried out in accordance with the principles expressed in the Declaration of Helsinki. The participants who were considered eligible were provided with oral/written information about the study and gave their informed consent.
Subjects
All the consecutive ALS patients receiving home NIV that were referred to the division’s out-patient clinic with indications for elective tracheostomy were considered for study eligibility. The diagnosis of ALS was based on standard clinical, enzymatic, electromyographic, and biopsy criteria. Indications for elective tracheostomy were based on the need for round-the-clock (>20 h) ventilatory support associated to an inability to protect the airway despite the use of manually and/or mechanically assisted coughing (10).
The study’s exclusion criteria included:
- ALS-related dementia and/or other forms of dementia;
- Significant concomitant progressive lung diseases, including lung cancer, chronic obstructive pulmonary disease and interstitial lung disease;
- Unavailability of a non-professional caregiver network at the patient’s home;
- A respiratory infection, a cough, increased sputum production, or a febrile illness during the month preceding the study.
The subjects recruited were randomized in a 1:1 ratio to the two study arms using blinded sealed envelopes.
Baseline assessment
The baseline characteristics considered (outlined in Table 1), included demographic data and the patients’ symptoms. Previous use of domiciliary NIV and forced vital capacity (FVC) obtained from pulmonary function testing done within about 6 months from admission were also recorded. Arterial blood gas (ABG) values obtained during spontaneous breathing were evaluated.

Full table
The following scales were examined:
- The Borg dyspnea scale (11) and the visual analogue scale (VAS; score of 1 to 10) were used to assess dyspnea;
- The Epworth sleepiness scale (ESS) was used to assess subjective evaluation of daytime sleepiness (12);
- The EuroQol-VAS (EQ-VAS) was used to assess QoL (13).
The patient’s primary caregiver’s burden was assessed using the 15-item relative stress scale (RSS) (14), an instrument that stratifies the level of stress and distress of care-givers (15).
Intervention
The ALS patients eligible for the study were admitted to the respiratory intensive care unit (RICU) of our Division and underwent conventional surgical tracheostomy and invasive mechanical ventilation (IMV) administered via a tracheostomy tube. A SERVO-i Ventilator (Maquet, Solna, Sweden) was utilized for IMV during the first 24 hours after tracheostomy. Beginning on the second day, mechanical ventilation was delivered by means of a portable ventilator set on a dual control ventilation mode. Although the benefits of hybrid modes of ventilation are still being debated (16), we decided to utilize this ventilation modality since target tidal volume (target VT)-PSV/PCV modes are increasingly popular and available on most home ventilators. The group A patients received pressure control (PC) ventilation with the additional option average volume assured pressure support (AVAPS) with a Trilogy 100 ventilator (Philips Respironics, Murrysville, USA). The ventilator settings were adjusted by trained specialists as follows—expiratory positive airway pressure (EPAP): 4 cmH2O; inspiratory positive airway pressure (IPAP) maximal pressure: 25 cmH2O; IPAP minimal pressure: 10 cmH2O; target VT: 6–8 mL/kg ideal body weight (IBW); respiratory rate (RR): 15 b/min; inspiratory time (IT): 1.5 s. IPAP and target VT settings were then readjusted based on ABG measurements in the attempt to maintain arterial oxygen saturation (SaO2) over 90% and PaCO2 between 35 and 45 mmHg. A Whisper Swivel II connector was positioned between the distal end of the circuit and a cuffed tracheostomy tube; a heat-moisture-exchanger (HME) filter was inserted at the end of the tracheostomy tube (Figure 1).
The group B patients received pressure assisted controlled ventilation (PACV) with the additional option target VT through an Airox Legendair ventilator (Covidien, Gosport, UK) which was chosen because it was commonly used for home ventilation by the pulmonologists of our Division. The ventilator settings were adjusted as follows—EPAP: 4 mbar; IPAP: 20 mbar; IPAP maximal pressure: 25 mbar; target VT: 6–8 mL/kg IBW; RR: 15 b/min; IT: 33%. The IPAP and target VT setting were then readjusted based on ABG measurements in the attempt to maintain SaO2 over 90% and PaCO2 between 35 and 45 mmHg. An active pressure-piloted exhalation valve during the inspiratory phase was located between the distal end of the circuit and a cuffed tracheostomy tube; a HME filter was inserted at the end of the tracheostomy tube. None of the patients in either group required additional oxygen.
ABG was regularly measured at 8-hour intervals from admission to discharge by means of a blood gas analyzer (Rapid Point 405 SIEMENS AG, Munich, Germany). The patients were discharged from the RICU when they were clinically stable and showed a PaCO2 level that was consistently between 35 and 45 mmHg (“target” PaCO2) for at least 48 consecutive hours.
Outcome assessment
Thirty days after the tracheostomy, the patients were re-evaluated by a trained pulmonologist at our out-patient clinic; the data collected during that assessment included the following:
- The Borg dyspnea scale and the VAS dyspnea scores;
- The ESS score;
- The EQ-VAS score;
- The ABG results.
The RSS score was calculated for the patient’s primary caregiver.
Data on ventilator breakdown and/or technical problems, including leaks and disconnections, were also collected.
Study end-points
The study’s primary end-point to assess the effect of the passive exhalation port system on gas exchanges was CO2 retention, defined as PaCO2 level >45 mmHg at the 30-day assessment.
The study’s secondary end-points were:
- The time to “target” PaCO2 during the patient’s RICU stay;
- The level of dyspnea, measured using the Borg dyspnea scale and the VAS dyspnea scores;
- Daytime sleepiness, measured using the ESS;
- The patients’ QoL, measured using the EQ-VAS;
- The primary caregiver’s burden, measured using the RSS;
- The number of respiratory-related hospitalizations and deaths during the 3 months following discharge from RICU.
Data on hospital admissions and mortality were extracted from patients’ clinical records.
Sample size and statistical analysis
This is the first study to our knowledge that specifically aimed to investigate the effect of a passive exhalation port on tracheostomized ALS patients. The results of other studies were not therefore available to test an a priori hypothesis on the expected incidence or magnitude of complications associated with the use of this instrument or to estimate an appropriate sample size. Based on our Division’s clinical records, we decided to recruit 20 consecutive patients.
Given the study design, it was impossible to carry out a blinded study although the personnel involved in data collection and analysis were not informed about which patients were assigned to the study groups.
Standard descriptive statistics were used to compare the baseline demographic and clinical characteristics. The qualitative variables were described as frequencies and percentages. The quantitative variables were described as means, standard deviations (SDs) or medians and ranges, as appropriate. The independent unpaired Student t test was used to compare normally distributed continuous variables. Nonparametric data were compared using the Mann-Whitney U test. Categorical variables were compared using the chi-square test or Fisher’s exact test, when required. The calculations were carried out using MedCalc Statistical Software (Ostend, Belgium). A bilateral P value <0.05 was considered statistically significant for all study tests.
Results
The 24 ALS patients attending our Division’s out-patient clinic during the period specified who had indications for elective tracheostomy were assessed for study eligibility. Four/24 were found to be ineligible for the study: 2 who were without a caregiver network at home and 2 who were diagnosed with ALS-related dementia. Twenty/24 patients were thus considered eligible for the current study.
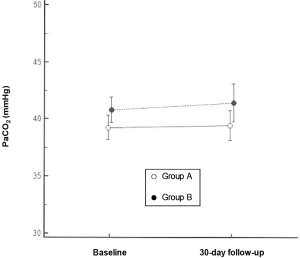
There were no significant differences between the two study arms as far as any baseline parameters were concerned (Table 1). At the 30-day assessment, one in each of the two groups showed significant CO2 retention. The PaCO2 values for the patients in group A were not significantly different from those in group B (39.455±3.942 vs. 41.437±4.751 mmHg; P=0.3621) or the baseline value (“target” PaCO2) (39.455±3.942 vs. 38.622±2.885; P=0.5914) (Figure 2). The time needed to achieve the “target” PaCO2 during their stay at RICU was similar in the two groups {3.5 [2–6] vs. 2 [2–7] days; P=0.3270}.
Scores on the Borg dyspnea scale {2 [1–3] vs. 1 [1–3]; P=0.2891}, the VAS dyspnea {20 [10–85] vs. 20 [0–50]; P=0.8571}, the ESS {8 [4–10] vs. 5.5 [0–12]; P=0.1443}, and the EQ-VAS {55 [50–80] vs. 50 [30–80]; P=0.4593} were not significantly different in the two groups. The differences in the RSS score were not statistically significant {49 [30–65] vs. 52 [25–64]; P=0.8650} (Table 2).

Full table
No ventilator breakdown and/or technical problem was reported.
The number of hospitalizations and of deaths did not significantly differ in the two groups (2 vs. 1, Fisher exact P value =1.000, and 2 vs. 2, respectively) during the first 3 months following discharge from RICU.
Discussion
One of the concerns facing clinicians who are evaluating the possibility of ventilating a patient using a passive exhalation port is the danger of rebreathing CO2. If, in fact, the patient’s expiratory flow exceeds the flow capacity of the leak port, the patient unavoidably exhales into the single-limb circuit and rebreathes CO2 on the subsequent inhalation. While the effect of using a passive exhalation port on gas exchange and symptoms has already been investigated in non-invasively ventilated patients (17-19), this is the first time, to our knowledge, that the effects of a Whisper Swivel connector have been prospectively evaluated in tracheostomized ALS patients.
The study results demonstrated that there are no substantial differences between the effects of a Trilogy ventilator connected to a Whisper Swivel passive exhalation port and those of a portable ventilator connected to an active exhalation valve in tracheostomized patients with ALS undergoing long-term ventilation at home. One out of 10 of the subjects in both groups, in fact, showed significant CO2 retention at the 30-day assessment. Interestingly, EPAP was set at 4 cmH2O, a relatively low level at which it has been demonstrated that rebreathed gas can amount up to 55% of each tidal volume in non-invasively ventilated individuals (19). We hypothesized that the Whisper Swivel connector was able to minimize rebreathing CO2 in the patients studied because the cuffed tracheostomy tube that was used ensured an “hermetically-sealed” condition that effectively maintained the EPAP level that had been fixed. Unlike tracheostomy ventilation, it is known that EPAP may fall below the set value in patients receiving NIV as a result of air leakage. Study results showed that although adding a HME to the patient’s circuit might have caused an increase in resistance to gas flow and the ventilator to display slightly higher pressures than those delivered to the patient, in fact, it did neither.
The study also demonstrated, interestingly, that utilization of a passive exhalation port did not have an adverse effect on primary clinical outcomes: the number of respiratory-related hospitalizations and of deaths were not significantly different in the two study arms at the 3-month assessment. Nor were significant differences noted in dyspnea {the Borg dyspnea scale: 2 [1–3] vs. 1 [1–3]; P=0.2891, the VAS dyspnea: 20 [10–85] vs. 20 [0–50]; P=0.8571} despite the fact that we might have expected a passive exhalation port to improve dyspnea, given its potential of reducing expiratory resistance, intrinsic PEEP, and the work of breathing. Even the patients’ daytime somnolence as measured by the ESS was not significantly different in the two groups {8 [4–10] vs. 5.5 [0–12]; P=0.1443}, a finding that led us to conclude that their quality of sleep was not influenced by the type of valve or port used. A nocturnal polysomnography would have made it possible to confirm this assumption.
Finally, the study uncovered that using a passive exhalation port with respect to a traditional active exhalation valve caused no significant increase in care-giver stress {RSS: 49 [30–65] vs. 52 [25–64]; P=0.8650}. The finding is important in view of the fact that the stress and anxiety experienced by caregivers assisting patients requiring long-term mechanical ventilation at home can lead to depressive symptoms and/or declining health (20).
The study has some limitations. First, the investigation was carried out on patients with a specific disease and its conclusions may not be applicable to patients with progressive respiratory failure caused by other etiologies. Focusing on a single disease made it possible nevertheless to exclude confounding factors associated to a more heterogeneous patient population. Second, the 30-day study period may have been too brief for differences in the end-points to emerge. On the other hand, a longer follow-up might have biased the results of the trial due to the high variability normally seen in ALS progression following tracheostomy (21). Third, the study was conducted on a small number of patients; clinical trials examining patients with rare diseases such as ALS often present this limitation (22). Fourth, both the participants and the treating clinicians were
Conclusions
Despite its limitations, the study provides useful information for clinicians caring for tracheostomized patients on long-term mechanical ventilation at home. Study results showed in fact that a standard Whisper Swivel connector:
- Can effectively minimize rebreathing CO2 in ALS patients receiving long-term ventilation via a tracheostomy;
- And its effects on patient symptoms and care-giver stress are similar to those of a traditional active exhalation valve.
Further studies will be able to clarify if a passive exhalation port can be used routinely to ventilate tracheostomized patients with different etiologies.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: The study design was approved by the Institutional Review Board of Padova City Hospital (No. 2145p). The participants who were considered eligible were provided with oral/written information about the study and gave their informed consent.
References
- Farrero E, Prats E, Povedano M, et al. Survival in amyotrophic lateral sclerosis with home mechanical ventilation: the impact of systematic respiratory assessment and bulbar involvement. Chest 2005;127:2132-8. [Crossref] [PubMed]
- Aboussouan LS, Khan SU, Meeker DP, et al. Effect of noninvasive positive-pressure ventilation on survival in amyotrophic lateral sclerosis. Ann Intern Med 1997;127:450-3. [Crossref] [PubMed]
- Lo Coco D, Marchese S, La Bella V, et al. The amyotrophic lateral sclerosis functional rating scale predicts survival time in amyotrophic lateral sclerosis patients on invasive mechanical ventilation. Chest 2007;132:64-9. [Crossref] [PubMed]
- Kacmarek RM, Chipman D. Basic principles of ventilator machinery. In: Tobin MJ. editor. Principles and Practice of Mechanical Ventilation. 2nd edition. New York: McGraw-Hill, 2006:53-95.
- Jiao GY, Newhart JW. Bench Study on Active Exhalation Valve Performance. Respir Care 2008;53:1697-702. [PubMed]
- Sanders MH, Kern N. Obstructive sleep apnea treated by independently adjusted inspiratory and expiratory positive airway pressure via nasal mask. Chest 1990;98:317-24. [Crossref] [PubMed]
- Chatburn RL. Classification of mechanical ventilators. In: Tobin MJ. editor. Principles and Practice of Mechanical Ventilation. 2nd edition. New York: McGraw-Hill, 2006:37-52.
- Ferguson GT, Gilmartin M. CO2 rebreathing during BiPAP ventilatory assistance. Am J Respir Crit Care Med 1995;151:1126-35. [PubMed]
- Lofaso F, Prigent H, Tiffreau V, et al. Long-term mechanical ventilation equipment for neuromuscular patients: meeting the expectations of patients and prescribers. Respir Care 2014;59:97-106. [Crossref] [PubMed]
- Bach JR, Bianchi C, Aufiero E. Oximetry and indications for tracheostomy for amyotrophic lateral sclerosis. Chest 2004;126:1502-7. [Crossref] [PubMed]
- Borg GA. Psychophysical bases of perceived exertion. Med Sci Sports Exerc 1982;14:377-81. [Crossref] [PubMed]
- Johns MW. A new method for measuring daytime sleepiness: the Epworth Sleepiness Scale. Sleep 1991;14:540-5. [Crossref] [PubMed]
- Torrance GW, Feeny D, Furlong W. Visual analog scales: do they have a role in the measurement of preferences for health states? Med Decis Making 2001;21:329-34. [Crossref] [PubMed]
- Greene JG, Smith R, Gardiner M, et al. Measuring behavioural disturbance of elderly demented patients in the community and its effects on relatives: a factor analytic study. Age Ageing 1982;11:121-6. [Crossref] [PubMed]
- Ulstein I, Bruun Wyller T, Engedal K. The relative stress scale, a useful instrument to identify various aspects of carer burden in dementia? Int J Geriatr Psychiatry 2007;22:61-7. [Crossref] [PubMed]
- Khirani S, Louis B, Leroux K, et al. Harms of unintentional leaks during volume targeted pressure support ventilation. Respir Med 2013;107:1021-9. [Crossref] [PubMed]
- Lofaso F, Brochard L, Touchard D, et al. Evaluation of carbon dioxide rebreathing during pressure support ventilation with airway management system (BiPAP) devices. Chest 1995;108:772-8. [Crossref] [PubMed]
- Hill NS, Carlisle C, Kramer NR. Effect of a Nonrebreathing Exhalation Valve on Long-term Nasal Ventilation Using a Bilevel Device. Chest 2002;122:84-91. [Crossref] [PubMed]
- Lofaso F, Aslanian P, Richard JC, et al. Expiratory valves used for home devices: experimental and clinical comparison. Eur Respir J 1998;11:1382-8. [Crossref] [PubMed]
- Douglas SL, Daly BJ. Caregivers of long-term ventilator patients. Physical and psychological outcomes. Chest 2003;123:1073-81. [Crossref] [PubMed]
- Vianello A, Arcaro G, Palmieri A, et al. Survival and quality of life after tracheostomy for acute respiratory failure in patients with amyotrophic lateral sclerosis. J Crit Care 2011;26:329.e7-14. [Crossref] [PubMed]
- Kuerner T. Essential rules and requirements for global clinical trials in rare lung diseases: a sponsor’s standpoint. Respir Investig 2015;53:2-6. [Crossref] [PubMed]


