Pretreatment platelet-to-lymphocyte ratio (PLR) as a predictor of response to first-line platinum-based chemotherapy and prognosis for patients with non-small cell lung cancer
Introduction
The mortality associated with lung cancer is one of the highest among malignant tumors (1). One reason for the poor outcome is the fact that the majority of non-small cell lung cancer (NSCLC) patients are diagnosed in advanced stages. Systemic combination chemotherapy has become the standard approach in the treatment of these patients (2-6). Unfortunately, an objective response is seen in only about 30% of NSCLC patients treated with chemotherapy. Furthermore, despite the progress in diagnosis and treatment of these patients, the overall 5-year survival rate remains unchanged at 15% over ten years (7).Thus, it is important to explore the potential role of tumor markers in predicting objective response and overall survival (OS) in patients with NSCLC.
Inflammation is increasingly recognized as being closely associated with cancer development (8). Inflammation can enhance tumor growth, invasion, angiogenesis, and, eventually, metastasis (9). Therefore, markers of inflammation may provide useful information for prognosis. Many markers of inflammation and immunology are repeatable, inexpensive and widely available in clinical practice, including platelet, lymphocyte, neutrophil, and C-reactive protein counts. Previous studies showed an association between these inflammatory markers and prognosis in many types of cancer. Our earlier study demonstrated that elevated platelet counts were associated with lymph node metastasis and poor survival in patients with NSCLC (10). Also, in recent years, many studies have focused on the role of an elevated neutrophil-to-lymphocyte ratio (NLR) (11,12) and platelet-to-lymphocyte ratio (PLR) (12-14) in the prognosis of different cancer populations. However, to our knowledge, no research has evaluated the role of PLR in predicting response to first-line chemotherapy and prognosis for patients with NSCLC.
The aim of the present study was to characterize the association between the pretreatment PLR and response to first-line chemotherapy and prognosis for patients with NSCLC.
Materials and methods
Patients and treatment evaluation
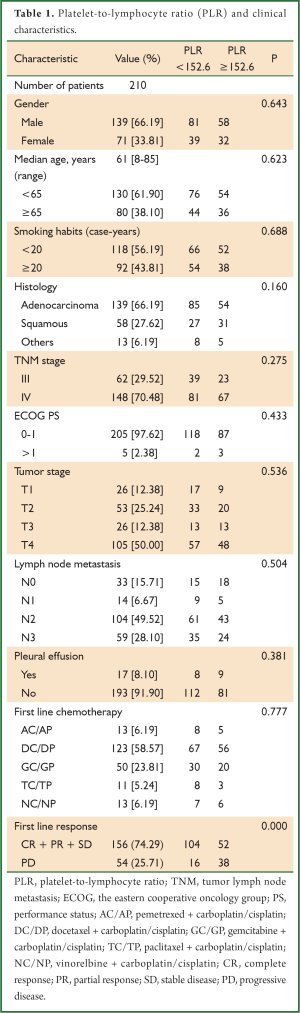
We consecutively enrolled 210 patients who were diagnosed with NSCLC in Jinling hospital (Nanjing, China) between January 2001 and August 2012 (Table 1). All diagnoses were confirmed by pathologic examination. Patients with systemic inflammation were excluded. All enrolled NSCLC patients were classified as stage III to stage IV, according to the guidelines of the tumor-node-metastasis (TNM) staging system of the Union for International Cancer Control (7th edition) (15). The stage assessment was based on computed tomography (CT) scans of the thorax and upper abdomen, magnetic resonance imaging (MRI) scans of the brain, and emission CT scans of bone. All patients received at least two cycles of standardized platinum-based chemotherapy. Tumor responses to treatment were assessed after two cycles of chemotherapy, based on the rules established by the Response Evaluation Criteria in Solid Tumors (RECIST) (16). Patients were considered to have achieved disease control (DC) if they had a complete response (CR), a partial response (PR), or stable disease (SD) according to RECIST. CR and PR were claimed only if the criteria for each are met at a subsequent time point as specified (generally four weeks later). Progressive disease (PD) was considered to represent disease progress. OS was determined for all patients who were censored at the time of last follow-up (31 August 2012).
Full table
Measurement of PLR
The platelet and lymphocyte counts of peripheral blood were measured with a hematology analyzer (Sysmex XE2100, Sys-mex, Japan) before chemotherapy. PLR was defined as the absolute platelet count divided by the absolute lymphocyte count.
Statistical analyses
All statistical analyses were performed with SPSS 17.0 software. A receiver operator characteristic (ROC) curve was constructed to reveal an association between PLR and tumor response after two cycles of first-line chemotherapy. The chi-squared (χ2) test was used to evaluate the relatedness between PLR and baseline clinical characteristics. A logistic regression model was used to analyze independent risk factors for response after two cycles of first-line chemotherapy. The Kaplan-Meier method was used to calculate the survival probability. A log-rank test was performed to assess heterogeneity in survival within each prognostic factor. A Cox regression model was used to analyze independent prognostic risk factors. A P value <0.05 was considered statistically significant.
Results
PLR and response to first-line chemotherapy
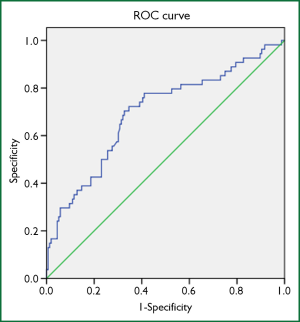
The NSCLC patients were divided into two groups according to the response to chemotherapy: 156 patients with DC (CR + PR + SD) and 54 patients with PD. As shown in Figure 1, the area under the ROC curve was 0.696 (95% CI: 0.610-0.781) and a PLR of 152.6 was identified as the cut-off value for predicting response, with a sensitivity of 70.4% and a specificity of 67.3%.
Patients were then divided into two groups according to PLR: 120 patients with PLR <152.6 and 90 patients with PLR ≥152.6. As shown in Table 1, PLR was significantly associated with response to first-line chemotherapy (P=0.000). No associations were observed between PLR and other clinical characteristics.
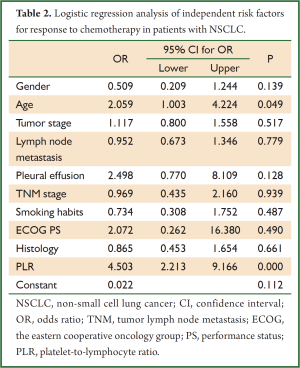
A logistic regression model was used to analyze independent risk factors for response after two cycles of chemotherapy. Table 2 shows that pretreatment PLR was the independent risk factor for response to chemotherapy in patients with NSCLC (OR, 4.503, 95% CI: 2.213-9.166).
Full table
PLR and OS
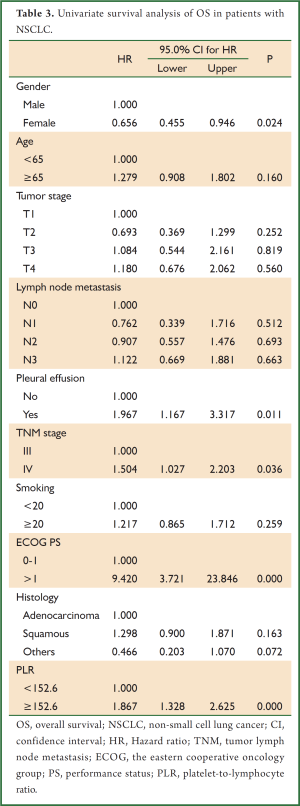
Univariate survival analysis demonstrated that high PLR (≥152.6) was associated with poor prognosis in NSCLC (HR, 1.867; 95% CI: 1.328-2.625). Pleural effusion, TNM stage (IV) and The Eastern Cooperative Oncology Group (ECOG) performance status (PS) (>1) were also associated with poor prognosis in NSCLC (HR, 1.967; 95% CI: 1.167-3.317; HR, 1.504; 95% CI: 1.027-2.203; HR, 9.420; 95% CI: 3.721-23.846, respectively). While, females showed a better prognosis in NSCLC than men (HR, 0.656; 95% CI: 0.455-0.946) (Table 3).
Full table
The average OS of the 210 NSCLC patients censored at the last follow-up was 558 days. The average OS of the 120 patients with a low PLR (PLR <152.6) was 648 days. The average OS for the remaining 90 patients whose PLR was ≥152.6 was 434 days. A Kaplan-Meier survival analysis showed that the OS of the NSCLC patients whose PLR was <152.6 was significantly longer than that of the NSCLC patients whose platelet count was ≥152.6 (log-rank test, P=0.000) (Figure 2).
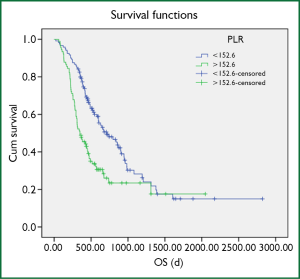
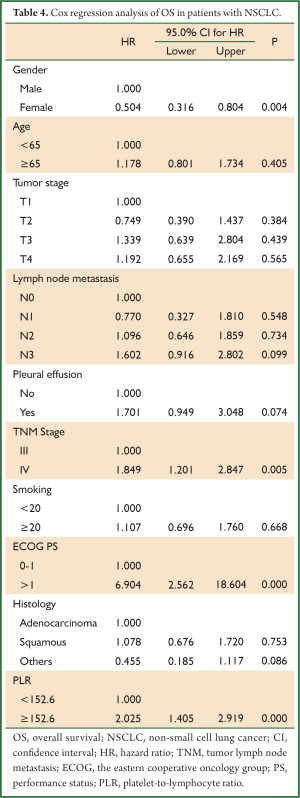
A Cox regression analysis showed that pretreatment PLR was an independent prognostic factor in NSCLC patients (HR, 2.025; 95% CI: 1.405-2.919). The risk of death for patients with a PLR ≥152.6 was more than twice higher than patients with a PLR <152.6. Clinical characteristics including gender, ECOG PS and TNM stage were also independent prognostic factors in patients with NSCLC (HR, 0.504; 95% CI: 0.316-0.804; HR, 6.904; 95% CI: 2.562-18.604; HR, 1.849; 95% CI: 1.201-2.847, respectively) (Table 4).
Full table
Discussion
Our study demonstrated that pretreatment PLR is an independent risk factor for response to first-line chemotherapy in patients with NSCLC. It is also associated with prognosis for NSCLC patients. An elevated PLR (≥152.6) predicted a poor response to first-line chemotherapy and a poor prognosis. We also showed that gender, stage, ECOG PS and response were independent prognostic factors for NSCLC patients.
Previous studies have identified a role of PLR in many malignant tumors, including colorectal cancer, breast cancer, ovarian cancer, and pancreatic ductal adenocarcinoma, but not NSCLC (12-14,17-21). To the best of our knowledge, this is the first study to explain the role of PLR in response to first-line chemotherapy and prognosis in NSCLC. Our results were similar to previous studies of other tumor types (12-14,19,22), but several studies have shown different results. For example, Bhatti et al. found that PLR was not a significant independent prognostic indicator in patients with resected pancreatic ductal adenocarcinoma. Azab et al. showed that higher PLR was associated with a trend of higher mortality in breast cancer patients with normal lymphocyte count (17). Perhaps, the role of PLR is different in different types of tumors. However, Smith et al. reported that preoperative PLR is a significant independent prognostic index in patients with resected pancreatic adenocarcinoma (14). These results are contradictory to the results of Bhatti, even though the same tumor type was evaluated. Clearly, the role of PLR in tumors still requires further clinical research.
The cut-off value of PLR was important in our evaluation. Cut-off values were 300 and 150 in previous studies. And even patients were categorized into quartiles according to their baseline 25th, 50th, and 75th PLR percentiles (17). In our study, the cut-off value for PLR was calculated with an ROC curve according to response after two cycles of first-line chemotherapy. One meta-analysis showed that median survival time was significantly associated with both the objective response rate and DC rate after first-line chemotherapy in patients with advanced NSCLC treated with systemic chemotherapy (23). Therefore, we believed that OS in NSCLC patients could be predicted more accurately with a preoperative PLR cut-off value of 152.6. However, more studies are needed to confirm the clinical relevance of this value.
NLR, which is defined as the absolute neutrophil count divided by the absolute lymphocyte count, is another predictor of prognosis in many tumors. Several studies reported that NLR was superior to PLR as a predictor of prognosis (13,22). However, other studies concluded the opposite (12). As with PLR, the significance of NLR may be associated with tumor type. Our previous study showed that NLR was associated with response to therapy and prognosis of advanced NSCLC patients treated with first-line platinum-based chemotherapy (24). Our present study also found that PLR was a predictor of response to first-line chemotherapy and prognosis for patients with NSCLC. However, further research is still needed to confirm whether NLR is more useful than PLR in NSCLC.
Platelet count and lymphocyte count are repeatable, inexpensive and widely available in clinical practice, making PLR a potentially useful biomarker for predicting response to first-line chemotherapy and prognosis in NSCLC. However, there are several limitations to our study. First, it was an observational, single-center study, and the sample of patients was not very large. Second, platelet count and lymphocyte count may be influenced by other factors. Third, patients treated with epidermal growth factor receptor tyrosine kinase inhibitor (EGFR-TKI) in the first line was not included in this study. Despite the above limitations, our study was the first to describe the relationship between PLR and response to first-line chemotherapy and prognosis in NSCLC.
In Conclusion, our study shows a significant association between the pretreatment PLR and response to first-line chemotherapy and prognosis in NSCLC. Pretreatment PLR was an independent risk factor for response to first-line chemotherapy in patients with NSCLC. An elevated PLR (>152.6) predicted a poor response to first-line chemotherapy and a poor prognosis.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (NO. 81170064) and the Natural Science Foundation of Jiangsu Province (NO. BK2011658).
Disclosure: The authors declare no conflict of interest.
References
- Siegel R, Naishadham D, Jemal A. Cancer statistics, 2013. CA Cancer J Clin 2013;63:11-30. [PubMed]
- Molina JR, Yang P, Cassivi SD, et al. Non-small cell lung cancer: epidemiology, risk factors, treatment, and survivorship. Mayo Clin Proc 2008;83:584-94. [PubMed]
- Wakelee HA, Bernardo P, Johnson DH, et al. Changes in the natural history of nonsmall cell lung cancer (NSCLC)--comparison of outcomes and characteristics in patients with advanced NSCLC entered in Eastern Cooperative Oncology Group trials before and after 1990. Cancer 2006;106:2208-17. [PubMed]
- Ramalingam S, Belani C. Systemic chemotherapy for advanced non-small cell lung cancer: recent advances and future directions. Oncologist 2008;13:5-13. [PubMed]
- Fathi AT, Brahmer JR. Chemotherapy for advanced stage non-small cell lung cancer. Semin Thorac Cardiovasc Surg 2008;20:210-6. [PubMed]
- Schiller JH, Harrington D, Belani CP, et al. Comparison of four chemotherapy regimens for advanced non-small-cell lung cancer. N Engl J Med 2002;346:92-8. [PubMed]
- Jemal A, Siegel R, Ward E, et al. Cancer statistics, 2009. CA Cancer J Clin 2009;59:225-49. [PubMed]
- Grivennikov SI, Greten FR, Karin M. Immunity, inflammation, and cancer. Cell 2010;140:883-99. [PubMed]
- Balkwill F, Mantovani A. Inflammation and cancer: back to Virchow? Lancet 2001;357:539-45. [PubMed]
- Liu HB, Gu XL, Ma XQ, et al. Preoperative platelet count in predicting lymph node metastasis and prognosis in patients with non-small cell lung cancer. Neoplasma 2013;60:203-8. [PubMed]
- Cedrés S, Torrejon D, Martínez A, et al. Neutrophil to lymphocyte ratio (NLR) as an indicator of poor prognosis in stage IV non-small cell lung cancer. Clin Transl Oncol 2012;14:864-9. [PubMed]
- Kwon HC, Kim SH, Oh SY, et al. Clinical significance of preoperative neutrophil-lymphocyte versus platelet-lymphocyte ratio in patients with operable colorectal cancer. Biomarkers 2012;17:216-22. [PubMed]
- Asher V, Lee J, Innamaa A, et al. Preoperative platelet lymphocyte ratio as an independent prognostic marker in ovarian cancer. Clin Transl Oncol 2011;13:499-503. [PubMed]
- Smith RA, Bosonnet L, Raraty M, et al. Preoperative platelet-lymphocyte ratio is an independent significant prognostic marker in resected pancreatic ductal adenocarcinoma. Am J Surg 2009;197:466-72. [PubMed]
- Sobin LH, Gospodarowicz MK, Wittekind C. TNM Classification of Malignant Tumours, 7th Edition. Oxford: Wiley-Blackwell. 2009.
- Eisenhauer EA, Therasse P, Bogaerts J, et al. New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur J Cancer 2009;45:228-47. [PubMed]
- Azab B, Shah N, Radbel J, et al. Pretreatment neutrophil/lymphocyte ratio is superior to platelet/lymphocyte ratio as a predictor of long-term mortality in breast cancer patients. Med Oncol 2013;30:432. [PubMed]
- Bhatti I, Peacock O, Lloyd G, et al. Preoperative hematologic markers as independent predictors of prognosis in resected pancreatic ductal adenocarcinoma: neutrophil-lymphocyte versus platelet-lymphocyte ratio. Am J Surg 2010;200:197-203. [PubMed]
- Raungkaewmanee S, Tangjitgamol S, Manusirivithaya S, et al. Platelet to lymphocyte ratio as a prognostic factor for epithelial ovarian cancer. J Gynecol Oncol 2012;23:265-73. [PubMed]
- Seretis C, Seretis F, Lagoudianakis E, et al. Enhancing the accuracy of platelet to lymphocyte ratio after adjustment for large platelet count: a pilot study in breast cancer patients. Int J Surg Oncol 2012;2012:653608.
- Smith RA, Ghaneh P, Sutton R, et al. Prognosis of resected ampullary adenocarcinoma by preoperative serum CA19-9 levels and platelet-lymphocyte ratio. J Gastrointest Surg 2008;12:1422-8. [PubMed]
- He W, Yin C, Guo G, et al. Initial neutrophil lymphocyte ratio is superior to platelet lymphocyte ratio as an adverse prognostic and predictive factor in metastatic colorectal cancer. Med Oncol 2013;30:439. [PubMed]
- Hotta K, Fujiwara Y, Kiura K, et al. Relationship between response and survival in more than 50,000 patients with advanced non-small cell lung cancer treated with systemic chemotherapy in 143 phase III trials. J Thorac Oncol 2007;2:402-7. [PubMed]
- Yao Y, Yuan D, Liu H, et al. Pretreatment neutrophil to lymphocyte ratio is associated with response to therapy and prognosis of advanced non-small cell lung cancer patients treated with first-line platinum-based chemotherapy. Cancer Immunol Immunother 2013;62:471-9. [PubMed]


