Limited prognostic value of myocardial viability assessment in patients with coronary artery diseases and severe left ventricular dysfunction
Introduction
Coronary artery disease (CAD) is the leading cause of heart failure which continues to pose an unequivocal challenge to the health care system worldwide. The optimal management of ischemic heart failure remains unclarified and relies typically on the totality of symptom burden, patient risks and disease severity assessed anatomically and functionally (1). It’s now generally accepted that coronary revascularization improves substantially left ventricular (LV) function in many patients with ischemic LV dysfunction (2,3). The concept of myocardial hibernation consequent to ischemic insults evokes great interests of performing myocardial viability assessment to predict functional recovery following revascularization. A large body of observational evidence has suggested that the identification of viable myocardium predicts also improved survival after surgical revascularization (4-7).
Among multiple imaging modalities used to test myocardial viability, positron emission tomography using 18F-fluorodeoxyglucose (18FDG-PET) is considered as the most sensitive one and holds the advantages of combining high spatial resolution with quantitative measure of myocardial perfusion (2).The 1- and 5-year follow-up results of PARR-2 study has demonstrated that PET-assisted management benefits patients adhering to imaging-based recommendations (8,9). In contrast, the Surgical Treatment of Ischemic Heart Failure (STICH) viability substudy showed that identification of viable myocardium by Single-photon Emission Computed Tomography (SPECT) or Dobutamine stress echocardiography did not add prognostic value in patients selected for coronary artery bypass grafting (CABG) (10). In brief, non-invasive myocardial viability testing in contemporary practice of managing ischemic LV dysfunction remains somewhat controversial and its prognostic value is a debating issue (6,11-13).
The objective of the present study was to investigate the impact of myocardial viability evaluation using 18FDG-PET as a decision-making tool on clinical outcomes of patients with CAD and severe LV dysfunction.
Methods
Study design
It’s a retrospective, single center, observational study of patients with documented CAD and LV dysfunction who were referred to our department for surgical revascularization or surgical revascularization work-up between January 2012 and December 2016. The study conformed to the ethical guidelines of the Declaration of Helsinki revised in 2015 and was approved by the institutional ethics committee (No: IORG0003571).
Patients
Inclusion criteria were: (I) baseline left ventricular ejection fraction ≤35% as evaluated by echocardiography, (II) a luminal narrowing ≥50% in at least one major coronary artery, and (III) myocardial viability assessed using 18FDG-PET. Those with myocardial infarction within 2 months and those receiving concomitant valvular surgery were excluded.
PET imaging and data analysis
The PET perfusion imaging was performed at rest with a standard protocol with N-13-ammonia and the static 18FDG-PET imaging was acquired using a standard protocol as previously described (11). The perfusion/FDG imaging data were submitted to the core laboratory for standard processing and quantified measures of the extent of ischemic myocardium and scar tissue.
Operative details
In CABG groups, anastomosis of left internal mammary artery to left anterior descending artery (LIMA-LAD) was routinely performed and completed by venous grafts to other coronary branches; off-pump CABG was conducted in 33 patients (62.3%).
Follow-up
The data including clinical findings, laboratory tests, serial echocardiographic studies and treatment histories were collected and recorded in an electronic database. Follow-up was completed with routine out-patient visits and telephone interview. All patients underwent at least once echocardiographic assessment at 3-month interval post-operatively. The primary endpoint was all cause mortality during follow-up; the secondary endpoint included a composite of all-cause mortality and major adverse cardiac and cerebrovascular events (MACCE) defined as acute coronary syndrome, repeat revascularization, hospitalization for heart failure and stroke.
Statistical analysis
Continuous variables are expressed as mean ± standard deviation (SD) and categorical variables as percentages. Comparisons were made using one-way analysis of variance (ANOVA) for continuous variables and Chi Square or Fisher’s exact test for categorical data. Actuarial estimate of survival was made using the Kaplan-Meier method and Kaplan-Meier survival curves were compared with the log-rank test. Cox’s proportional hazards model was used as appropriate. Statistical analysis was performed using the statistical Package for Social Science, version 23.0 (SPSS, Chicago, IL, USA).
Results
Eighty-one patients with ischemic left ventricular dysfunction fulfilled the inclusion criteria and were selected for this study. According to the viability assessment using 18FDG-PET and the final treatment options, the patients were divided into CABG group with the extent of myocardial scar ≥10% (n=30), CABG group with the extent of myocardial scar <10% (n=23) and the OMT group (n=28). The patients in OMT group were contraindicated for CABG for the coronary anatomy unsuitable for surgical revascularization.
The baseline characteristics of the study population are listed in Table 1. There was no significant difference among groups with respect to variables such as age, gender, body mass index and the prevalence of hypertension, dyslipidemia, chronic obstructive pulmonary diseases (COPD) and previous myocardial infarction (MI). The mean age was 54.8±12.5 and male gender was prevalent (67/81, 82.7%). The pre-operative LVEF ranged from 24% to 35% (mean 32.1%±3.0%) and the LV diastolic dimension from 4.8 to 8.9 cm (mean 6.2±0.9 cm). The mean number of diseased coronary vessels was 2.9±0.5. The extent of scar tissue and perfusion/FDG activity mismatch are respectively 18.1%±14.2% (range: 0–54%) and 20.7%±18.9% (range: 0–72%). The preoperative mean logistic European System for Cardiac Operative Risk Evaluation II (EuroSCORE II) calculation was 5.4%±4.2%. The preoperative investigation revealed significant difference among groups regarding myocardial ischemic burden; otherwise, the demographic parameters, the prevalence of comorbidities were comparable among 3 groups.
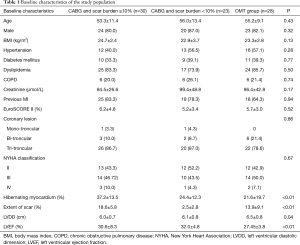
Full table
The direct comparison between two groups with respect to the peri-operative variables is presented in Table 2. The cardiopulmonary bypass (CPB) time and aortic cross clamping time were not significantly different; the percentage of off-pump surgery were comparable. There was no significant difference between two groups with respect to the use of intra-aortic balloon counterpulsation (IABP), the duration of mechanical ventilation and the use of continuous renal replacement therapy (CRRT) in post-operative evolution; nevertheless, the post-operative in-hospital stay were significantly longer in the group with myocardial scar ≥10%.

Full table
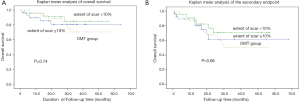
After a median follow-up of 32 months, as shown in Table 3, all-cause mortality was 18.5% (15/81) and the cumulative rate of secondary endpoint was 33.3% (27/81) in all patients. The Kaplan-Meier analysis showed no statistically significant difference in either overall survival or secondary endpoint among three groups (Figure 1). There was no significant interaction between either myocardial hibernation or myocardial scarring with respect to all-cause mortality or incidence of MACCE (all P>0.05).
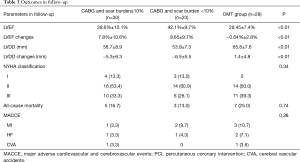
Full table
Discussion
The main finding of the present study is that myocardial viability assessment might be helpful for decision-making in the management of CAD with severely depressed LV systolic function, especially in those potentially eligible for surgical revascularization. However, the viability-guided therapeutic strategy is not related to improved clinical outcomes, since the overall mortality and incidence of MACCE were observed to be similar with three treatment options.
The American College of Cardiology/American Heart Association guideline recommends to perform CABG in patients with poor LV function with significant viable revascularizable myocardium (Class II B, level of evidence: B). However, challenges in the application of viability testing to clinical practice are posed by the complexity of myocardial substrate abnormalities, with combinations of stunned, hibernating and scarred myocardium being identified in the same patient. It remains unclarified whether the amount of hibernating myocardium or the extent of scar tissue to be a better predictor of myocardial functional recovery following revascularization, and their cutoffs are not well defined (9-11). In our study, we shared the standpoint of Dr. Beanlands et al. and dichotomized the patients by the cut-off level of myocardial scar extent being 10%. According to the definition, myocardial scarring means an irreversible ischemic damage regardless of therapies while myocardial hibernation represents a status in which the ischemic myocardium is potentially functionally recoverable following a successful revascularization. In this sense, myocardial scar might be a better parameter to identify the severity of ischemic insult and to predict outcomes following revascularization.
The present study confirmed that surgical coronary revascularization instead of optimal medical treatments resulted in structural and functional improvements of ischemic failing hearts; however, the improvements were independent of the baseline viability status. The extent of scar failed to predict clinical outcomes following surgical revascularization. The disparity in the extent of myocardial hibernation between two groups could be an issue and a confounding factor. As many previous studies have also shown that the amount of hibernating myocardium predicts function recovery after revascularization (6,14). The interpretation of the viability evaluation could be difficult in the scenario of co-existence of a substantial amount of scarring and hibernating myocardium, since the two entities play an opposing role in the process of reverse remodeling following revascularization.
No significant benefits of overall survival and MACCE-free survival were observed in two CABG groups as compared to OMT groups. We ascribed it in part to the relative high per-operative mortality associated with CABG procedure which counterbalanced its benefits on survival rates; another explanation is that surgical revascularization might be insufficient to improve the micro-circulation in the ischemic area and to salvage enough hibernating myocardium in the setting of severe ischemic LV dysfunction.
Taken together, our study suggests that it might be of limited value to perform routinely myocardial viability assessment using 18FDG-PET in preoperative investigation of CAD with severe LV dysfunction. 18FDG-PET imaging is useful to select patients with substantial myocardial viability for surgical revascularization, but the functional recovery following CABG in terms of LVEF increasing is not necessarily translated to survival benefits.
Our study holds several limitations. First of all, the decision making in the context of CAD with severe LV dysfunction is multifactorial and dependent upon a large spectrum of interactive parameters including the LVEF, extent of LV remodeling, extent of scarring and hibernation, clinical symptoms, duration of dysfunction, coronary anatomy and the comorbidities. Due to the retrospective nature of the present study, the impact of myocardial viability assessment on decision making might be overstated. Secondly, the OMT was a compromise rather than an active treatment option for those unsuitable for CABG. Another limitation was due to a small sample size and a relatively short follow-up time that increases the probability of type II error. These limitations warrant a larger prospective randomized trial over longer follow-up durations.
Conclusions
Myocardial viability assessment using18FDG-PET is of limited value in the preoperative investigation in patients with CAD and severe LV dysfunction. In patients with substantial myocardial scar burden, cardiac functional recovery following surgical revascularization is not necessarily translated to survival benefits.
Acknowledgements
Funding: This work was supported by the National Key Research and Development Program of China (31330029), the National Natural Science Foundation of China (81600229) and Special Funds for Fundamental Scientific Research in Central Universities (011853031).
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: The study conformed to the ethical guidelines of the Declaration of Helsinki revised in 2015 and was approved by the institutional ethics committee (No: IORG0003571).
References
- Mielniczuk LM, Toth GG, Xie JX, et al. Can functional testing for ischemia and viability guide revascularization? JACC Cardiovasc Imaging 2017;10:354-64. [Crossref] [PubMed]
- Anavekar NS, Chareonthaitawee P, Narula J, et al. Revascularization in patients with severe left ventricular dysfunction. J Am Coll Cardiol 2016;67:2874-87. [Crossref] [PubMed]
- Pitt M, Lewis ME, Bonser RS. Coronary artery surgery for ischemic heart failure: Risks, benefits, and the importance of assessment of myocardial viability. Prog Cardiovasc Dis 2001;43:373-86. [Crossref] [PubMed]
- Sawada SG, Dasgupta S, Nguyen J, et al. Effect of revascularization on long-term survival in patients with ischemic left ventricular dysfunction and a wide range of viability. Am J Cardiol 2010;106:187-92. [Crossref] [PubMed]
- Sciagrà R, Pellegri M, Pupi A, et al. Prognostic implications of Tc-99m sestamibi viability imaging and subsequent therapeutic strategy in patients with chronic coronary artery disease and left ventricular dysfunction. J Am Coll Cardiol 2000;36:739-45. [Crossref] [PubMed]
- Pasquet A, Robert A, D’Hondt AM, et al. Prognostic value of myocardial ischemia and viability in patients with chronic left ventricular ischemic dysfunction. Circulation 1999;100:141-8. [Crossref] [PubMed]
- Siontis KC, Chareonthaitawee P. Prognostic value of positron emission tomography myocardial perfusion imaging beyond traditional cardiovascular risk factors: Systematic review and meta-analysis. Int J Cardiol Heart Vasc 2015;6:54-9. [Crossref] [PubMed]
- Beanlands RSB, Nichol G, Huszti E, et al. F-18-Fluorodeoxyglucose positron emission tomography imaging-assisted management of patients with severe left ventricular dysfunction and suspected coronary disease. J Am Coll Cardiol 2007;50:2002-12. [Crossref] [PubMed]
- Mc Ardle B, Shukla T, Nichol G, et al. Long-term follow-up of outcomes with F-18-Fluorodeoxyglucose positron emission tomography imaging-assisted management of patients with severe left ventricular dysfunction secondary to coronary disease. Circ Cardiovasc Imaging 2016.9. [PubMed]
- Bonow RO, Maurer G, Lee KL, et al. Myocardial Viability and Survival in Ischemic Left Ventricular Dysfunction. N Engl J Med 2011;364:1617-25. [Crossref] [PubMed]
- Beanlands RSB, Ruddy TD, deKemp RA, et al. Positron emission tomography and recovery following revascularization (PARR-1): the importance of scar and the development of a prediction rule for the degree of recovery of left ventricular function. J Am Coll Cardiol 2002;40:1735-43. [Crossref] [PubMed]
- Cleland JGF, Calvert M, Freemantle N, et al. The Heart Failure Revascularisation Trial (HEART). Eur J Heart Fail 2011;13:227-33. [Crossref] [PubMed]
- Orlandini A, Castellana N, Pascual A, et al. Myocardial viability for decision-making concerning revascularization in patients with left ventricular dysfunction and coronary artery disease: A meta-analysis of non-randomized and randomized studies. Int J Cardiol 2015;182:494-9. [Crossref] [PubMed]
- Pagano D, Townend JN, Littler WA, et al. Coronary artery bypass surgery as treatment for ischemic heart failure: the predictive value of viability assessment with quantitative positron emission tomography for symptomatic and functional outcome. J Thorac Cardiovasc Surg 1998;115:791-9. [Crossref] [PubMed]


