“Valve in valve” through femoral approach to allow the later implantation of long-term left ventricle assist device: a case report
Long-term left ventricular assist devices (LVADs) may be indicated as a bridge to heart transplantation, as a bridge to candidacy or as a destination therapy, and their use in our environment is continuously growing. The selection of the patient and his perioperative management are determinant factors to the success of this treatment (1).
We present a 42-year-old male patient with advanced heart failure. His cardiac medical history began 7 years earlier with a severe aortic stenosis due to a bicuspid valve that was treated with aortic valve replacement with a biological aortic prosthesis Mitroflow 21 mm. A biological prosthesis was implanted by choice of the patient due to the desire not to follow anticoagulant treatment. The patient suffered a perioperative myocardial infarction. After a prolonged hospital stay, he was discharged with moderate left ventricular dysfunction and moderate pulmonary hypertension.
In the following years, the patient was clinically compensated with the New York Heart Association (NYHA) functional class II/IV. An internal cardiac defibrillator was implanted in the context of syncopal ventricular tachycardia.
Three months before the current admission he developed progressive dyspnea despite of intensive medical treatment. Finally, the patient was admitted to the hospital with congestive heart failure and deterioration of renal function (functional class NYHA IV/IV).
The echocardiogram showed very severe left ventricular dysfunction (12% by Simpson method) with degeneration of the aortic prosthesis with moderate central regurgitation. Cardiac catheterization showed no coronary stenosis, moderate aortic regurgitation (Figure 1) and severe pulmonary hypertension with negative vasodilator challenge test.

In this scenario we decided to implant HeartMate 3TM long-term LVAD as a bridge to candidacy. Since it was mandatory to correct the prosthetic insufficiency in order to have a well-functioning LVAD, it was decided to perform a transcatheter aortic valve implantation (TAVI). The rationale for such decision was based on the concept that concomitant aortic valve surgery and LVAD implantation would increase cardiopulmonary bypass time and so the risk of complications.
TAVI was performed through femoral approach with implantation of an Evolut R 23 mm (Medtronic Minn) under sedation without general anesthesia and without complications. Seven hours later HeartMate 3TM LVAD was implanted (Figures 2,3). Finally, the patient was discharged 16 days later with improvement of heart failure.

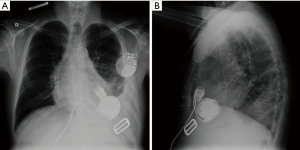
Follow-up of the patient revealed reduction of pulmonary pressures and he was included in the cardiac transplant waiting list.
Discussion
The presence of significant aortic regurgitation (higher than mild) is a formal contraindication for the implantation of LVAD. The back flow in the aortic root worsens aortic regurgitation leading to failure of system effectiveness.
Several surgical strategies on the aortic valve have been described; valve repair, replacement with a biological prosthesis or closing the aortic valve. All of the three procedures require cardiopulmonary bypass and aortic clamping, which increases the risk of complications, especially if the patient has previously undergone cardiac surgery (4).
The favorable outcomes obtained with TAVI in patients with aortic stenosis and high or prohibitive risk for cardiac surgery have led to extend the possibilities of TAVI to other clinical situations such as “valve in valve” in case of dysfunction of biological prosthesis (5).
There is no much experience in TAVI and LVAD (6). In this regard, few cases had been reported as transapical or transaortic TAVI and LVAD implantation (7,8).
To the best of our knowledge, this is an uncommon case of a TAVI through femoral approach to correct significant aortic regurgitation of a degenerated bioprosthesis allowing the later implantation of a long term LVAD in a case of advanced heart failure.
We believe that our strategy has shortened the time of surgical intervention and reduced the risk of complications as compared to aortic valve replacement during the implantation of LVAD. This strategy should be considered as an alternative option in such complex situations.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Informed Consent: Written informed consent was obtained from the patient for publication of this manuscript and any accompanying images.
References
- Ponikowski P, Voors AA, Anker SD, et al. 2016 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure: The Task Force for the diagnosis and treatment of acute and chronic heart failure of the European Society of Cardiology (ESC)Developed with the special contribution of the Heart Failure Association (HFA) of the ESC. Eur Heart J 2016;37:2129-200. Erratum in: Eur Heart J 2018;39:860. [Crossref] [PubMed]
- Pascual I, Díaz-Molina B, Lambert JL, et al. Angiography showing moderate aortic regurgitation before transcatheter aortic valve implantation (TAVI). Asvide 2018;5:586. Available online: http://www.asvide.com/article/view/25561
- Pascual I, Díaz-Molina B, Lambert JL, et al. Angiography showing the correction of aortic regurgitation after “valve in valve”. Asvide 2018;5:587. Available online: http://www.asvide.com/article/view/25564
- Robertson JO, Naftel DC, Myers SL, et al. Concomitant aortic valve procedures in patients undergoing implantation of continuous-flow left ventricular assist devices: An INTERMACS database analysis. J Heart Lung Transplant 2015;34:797-805. [Crossref] [PubMed]
- Baumgartner H, Falk V, Bax JJ, et al. 2017 ESC/EACTS Guidelines for the management of valvular heart disease. Eur Heart J 2017;38:2739-91. [Crossref] [PubMed]
- Phan K, Haswell JM, Xu J, et al. Percutaneous Transcatheter Interventions for Aortic Insufficiency in Continuous-Flow Left Ventricular Assist Device Patients: A Systematic Review and Meta-Analysis. ASAIO J 2017;63:117-22. [Crossref] [PubMed]
- Murashita T, Joyce DL, Pochettino A, et al. Concomitant Valve-in-Valve Transcatheter Aortic Valve Replacement and Left Ventricular Assist Device Implantation. Innovations (Phila) 2017;12:147-9. [Crossref] [PubMed]
- Baum C, Seiffert M, Treede H, et al. Concomitant transcatheter aortic valve and left ventricular assist device implantation. ASAIO J 2013;59:90-2. [Crossref] [PubMed]


