Early reoperation performed for the management of complications in patients undergoing general thoracic surgical procedures
Introduction
General thoracic surgery is associated with the development of a wide range of postoperative complications. The commonest observed postoperative complications are atelectasis, supraventricular arrhythmias, prolonged air leak and bleeding. Postoperative retention pneumonia, empyema, bronchopleural fistula formation, chyle leak, wound problems, lobar torsion and nerve injuries are observed less frequently (1,2). In addition, other specific complications are related with specific procedures (i.e., pneumonectomy, resection of Pancoast tumors, esophagectomy). Most of the postoperative complications in general thoracic surgery are managed conservatively or with minor interventions, such as fiberoptic bronchoscopy, reinsertion of chest tube drain, chest needle drainage or the application of a vacuum assisted closure (VAC) device (1,2).
The need to proceed with reoperation in the immediate postoperative period in order to resolve a postoperative complication is not common and the observed rate after general thoracic surgery procedures or isolated lung parenchyma resection is reported to vary between 1.2% and 3.7% (3-7). Interestingly, most of such series in Medline (where an abstract is at least available to read) are published in German, Russian or Italian language (3,4,6-10), while only one series even though coming from Germany is published in English (5). These series are mostly historical and include patients who underwent thoracic surgery from 1963 until recently. The current study evaluates early reoperations which are performed after general thoracic surgical procedures in order to resolve a postoperative complication, focusing mainly in the observed rate and the predisposing factors for the development of such complications in almost the whole spectrum of the procedures performed in the era of modern general thoracic surgery.
Patients and methods
During the time period 2009-2012 (four years), 719 patients (mean age: 54±19 years, male: 71.62%) underwent a variety of general thoracic surgery procedures under general endotracheal anesthesia in an Academic/teaching Cardiothoracic department. Minor procedures, such as diagnostic/interventional fiberoptic or rigid bronchoscopy, thoracoscopy under local anesthesia, chest tube insertion, pericardiocentesis, insertion of a tracheal “T” tube, biopsies of scalene lymph nodes, open and dilational tracheostomy were excluded from the study.
All patients who underwent early reoperation for the management of surgical postoperative complications were retrospectively collected from the electronic database of the department. Early reoperation was defined as any reoperation performed to control a postoperative complication within the four weeks immediate postoperative period or within the initial postoperative hospitalization if this exceeding more to four weeks. Reoperations that were performed electively during the 4-week immediate postoperative period for oncologic reasons (completeness of resection) were excluded from the analysis. The notes of hospitalization and the notes of surgical procedures were assessed to retrieve all the required information for identifying the predisposing factors for reoperation.
A cumulative list of the procedures that were performed during the study period is presented in Table 1. Staging mediastinoscopies, anterior mediastinotomies and thoracoscopies that were performed at the same time with thoracotomy were not recorded as isolated procedures. The surgical access used to accomplish the above mentioned procedures were: thoracotomy in 405 cases (56.3%), video-assisted thoracoscopy in 99 cases (13.7%), cervical incision (including the 47 video-mediastinoscopies) in 63 (8.7%), median full or upper sternotomy in 51 (7.1%), anterior mediastinotomy in 24 (24%), axillary minithoracotomy in 21 (2.9%), suxiphoid incision in 12 (1.6%) and other incisions used for the resection of chest wall tumors and the correction of chest wall deformities (which do not fall within the typical thoracic approaches) in 44 cases (6.1%). Statistical analysis was performed with the IBM SPSS Statistics v 10.0.
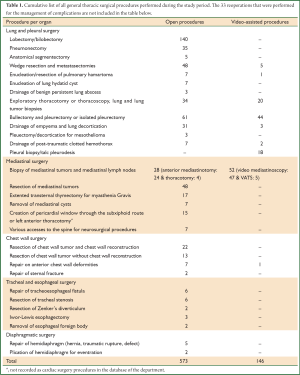
Full table
Results
Overall, 33 out of the 719 patients (4.6%) underwent early re-operation for managing surgical postoperative complications during the 4-year study period. A list of all the postoperative complications observed during the study period is presented in Table 2. The overall incidence of complications observed in the 719 patients was 22.2%.
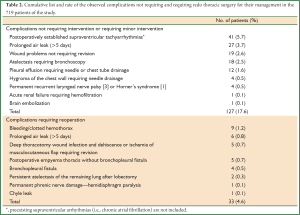
Full table
Reoperation for bleeding and/or clotted hemothorax
Nine patients underwent reoperation to stop bleeding and/or to remove the accumulated blood clots from the chest. The initial access was a thoracotomy incision in all the nine cases. None from a total of the 99 performed video-assisted thoracic surgery (VATS) procedures (not including VATS lobectomies and thymectomies) was complicated with postoperative bleeding. Reopening of the thoracotomy incision took place between the day of surgery and the 5th postoperative day (median: 1.0 day). Details on initial procedures that were complicated with postoperative bleeding, day of reoperation, active bleeding detection and predisposing factors for bleeding are presented in Table 3. The effect of reoperation on overall postoperative hospital stay was moderate, since the median hospital stay from reoperation to discharge was eight days.
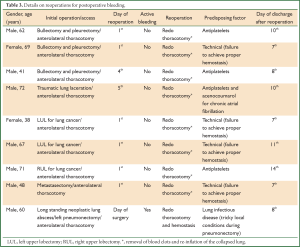
Full table
The majority of reoperations were performed on the 1st postoperative day. It is notable that four patients were receiving antiplatelet drugs before surgery (clopidogrel or the combination of aspirin and clopidogrel) for cardiovascular diseases which were discontinued 5 to 7 days before surgery in the three elective procedures. One patient, under treatment with aspirin and warfarin for coronary artery disease and chronic atrial fibrillation, who was initially operated for traumatic lung laceration, bled the 5th postoperative day since the re-administration of aspirin and warfarin.
Reoperation for the control of prolonged air leak
Six patients underwent reoperation to control a persistent prolonged air leak. Table 4 presents the initial procedures, the predisposing factors for the prolonged air leak which did not respond to conservative treatment, the site of air leak detected during reoperation and the accesses used during reoperation. Regarding the operative technique used in the initial procedures, stapling lines were not reinforced with synthetic materials during the study period. One-way intrabronchial valves were not used in the attempt to control the prolonged air leak. Reoperations were performed between the 9th and 29th postoperative day (median: 10.5 days) resulting in air leak cessation and full lung expansion in all cases. The time from reoperation to discharge from the hospital varied between 3 and 14 days (median: 5.5 days) (Table 4).
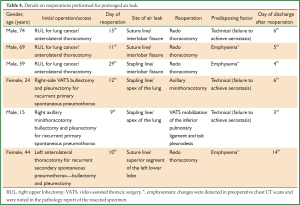
Full table
The decision to reoperate was always made after failure of the conservative treatment manoeuvres, such as repeated blood pleurodesis or position of a chest tube drain in the 2nd intercostal space in the midclavicular line after the three upper lobectomies. The criteria to reoperate were: (I) failure of the lung or remaining lung to fully expand after discontinuation of the applied to the chest tube(s)/suction (–20 cm H2O) and (II) a trend obtained by the Thopaz digital drainage system (Medela AG, Switzerland) with no tendency of the leak to diminish during several days, even in the lung was fully expanded in plain chest radiography.
Reoperation to drain a post-thoracotomy empyema (without fistula)
Reoperation for the drainage of empyema was made after thoracotomy in four cases and after a VATS procedure in the last case. Details of the initial procedures and subsequent reoperations are presented in Table 5. Reoperations were performed between the 8th and 29th postoperative day (median: 15.2 days), the earlier being performed for drainage of a post-pneumonectomy empyema. Reoperations for postoperative empyema resulted in serious prolongation of the hospital stay, since the median hospital stay from reoperation to discharge was 23 days.
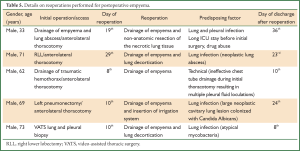
Full table
Reoperation for bronchopleural fistula
Details regarding initial surgery and reoperations for bronchopleural fistula formation are presented in Table 6. Technical issues were implicated in bronchopleural fistula formation in 3 out of the 4 patients. Reoperations were performed between the 10th and 17th postoperative day (median: 12.5 days). Reoperations to control a bronchopleural fistula were associated with increase of the hospital stay (median time from reoperation to discharge: 18.5 days).
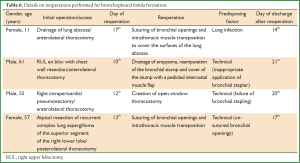
Full table
Reoperation to revise the thoracotomy incision or an ischemic musculocutaneous flap
Four patients underwent reoperation to repair the thoracotomy incision due to deep thoracotomy wound suppuration and dehiscence in three cases, lung herniation in one and venous congestion of a musculocutaneous muscle flap resulting in partial necrosis in the last one. Details regarding these operations are presented in Table 7. The median elapsed time from initial surgery to reoperation was 15.0 days. Discharge from the hospital was made possible within 14-21 days since the day of reoperation (median time from reoperation to discharge: 18.5 days).
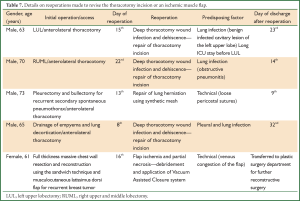
Full table
Reoperation for persistent atelectasis of the remaining lung
Two patients underwent completion pneumonectomy for persistent atelectasis of the remaining lobe after left lower lobectomy which was performed for lung cancer in the 1st case and after left upper lobectomy performed for vanishing lung syndrome in the 2nd case respectively. Repeated fiberoptic bronchoscopies failed to expand the remaining lobe. Reoperations were performed the 5th and 9th postoperative day respectively. Both patients had fatal outcome since persistent atelectasis led to infection of the remaining lobe, with subsequent sepsis and multiple organ failure. Technical issues were detected as the cause of persistent atelectasis in both cases. The application of bronchial stapler too close to the origin of the left upper lobe in the 1st case of left lower lobectomy and the extensive mobilization of the left lower lobe during left upper lobectomy which had as result the twisting of the main bronchus at some body positions. Bronchomalacia was also present in the second case as a second contributing factor.
Reoperation to plicate the hemidiaphragm
Damage to the phrenic nerve during resection of a thymoma of the right cardiophrenic angle resulted in elevation of the right hemidiaphragm to the level of the 4th intercostal space. Reoperation to plicate the right hemidiaphragm through reopening of the anterolateral thoracotomy incision was obviated dyspnea development on minimal effort since the mobilization of the patient. Reoperation was made on the 11th postoperative day, resulting in almost immediate serious improvement of the clinical condition of the patient.
Reoperation for chyle leak
One patient underwent reoperation for chyle leak after the resection of a large bronchogenic cyst. Conservative treatment with total parenteral nutrition and octreotide administration for ten days failed to reduce chyle fistula output to less than 800 mL per day. Reopening of the thoracotomy incision was made on the 13th postoperative day. The site of chyle leak was well detected and sutured with multiple pledgeted “Π” sutures. Recovery of the patient was uncomplicated and he was discharged from the hospital after seven days.
Factors responsible for the development of complications which required reoperation for their management
Searching within the hospitalization notes and the notes of the initial procedure and reoperation of the 33 patients allowed us to detect some responsible factors responsible for the development of complications requiring reoperation. These factors were considered to be technical in 17 cases (51.5%), when initial surgery was employed for lung or pleural infections in 9 cases (27.3%), the recent antiplatelet drug administration in 4 cases (12.1%) and the occurrence of diffuse lung emphysematous changes in 3 cases (9.1%). Lung and/or pleural infections were well documented by cultures obtained from the bronchial tree, pleural fluid or the resected specimen.
Advanced lung emphysematous changes were found in 3 out of the 6 patients (50%) with prolonged air leak. The emphysema was obvious in preoperative chest CT scan and was also noted by the surgeon in the operation notes. The recent use of antiplatelets was detected in 4 out of the 9 patients who underwent reoperation for postoperative bleeding. Initial surgery in the presence of lung and/or pleural infection was recorded in 5 out of the 9 patients who were complicated with postoperative empyema and bronchopleural fistulas. Surgery for infectious lung disease was also found in one patient who was complicated with postoperative bleeding. The infection of the lung and pleural cavity during initial surgery were considered as the responsible factor for postoperative bleeding due to the particular local conditions during mobilization of the lung and dissection of the hilum. Deep wound infection and wound dehiscence occurred in three patients who were initially operated for lung and/or pleural infections. Long ICU stay before initial surgery was also recorded in two patients as a further contributing factor to infection.
In more than half of the cases various technical issues were considered to be the predisposing factors for the occurrence of a complication requiring reoperation, comprising of failure to achieve hemostasis or aerostasis, failure or inappropriate use of bronchial and parenchymal staplers, loose pericostal sutures, ineffective chest tube drainage, venous congestion of a latissimus dorsi musculocutaneous flap and extensive mobilization of the residual lobe.
Mortality of reoperations
Mortality of re-operations was 6.1% (2 out of the 33 patients) and it was associated with the need to proceed with completion pneumonectomy in both cases which developed postoperatively persistent atelectasis of the remaining lung resulting in infection of the remaining lung parenchyma. Both patients died of sepsis and multiple organ failure within a few days since the day of completion pneumonectomy procedure. In the rest 29 out of the 33 cases, early reoperation solved the problem allowing recovery of the patient.
Type of procedures which were complicated with complications requiring reoperation
The higher rate of complications requiring reoperation was observed after surgery performed for trauma and lung/pleural infections, followed by lung parenchyma resection and pleurectomy (Table 8). Complications requiring reoperation for their management were necessary in 2 out of the 99 video-thoracoscopic procedures (2%). Procedures performed via median full or upper sternotomy, mediastinoscopies and mediastinotomies were not associated with complications requiring reoperation during the study period.
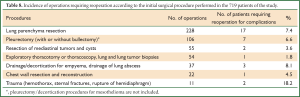
Full table
Discussion
Historical series on reoperation for postoperative complications after thoracic surgery report the rate of bleeding or clotted hemothorax to be the leading cause for reoperation. This accounts for 52-75% of all reoperations (3-5), with the exception of the series by Ermolov and Stonogin published in 1996, where rethoracotomy rate for intrathoracic bleeding was only 2.7% (11). Bronchopleural fistulas accounted for 17.8-25.5% of all reoperations in the above mentioned series (3-5). However, most of these series include patients who underwent thoracic surgical procedures since the early 60s. During the last five decades the evolution of general thoracic surgery with the ongoing use of minimally invasive techniques, the progress in surgical technology and biomaterials and the progress in lung isolation techniques made serious changes in general thoracic surgery practice. A range of procedures performed in the past are completely abandoned, other procedures are performed only through minimally invasive techniques, while aggressive surgery for locally advanced lung cancer became easier with the use of neo-adjuvant therapy and consequently the reoperation rate for complications in modern thoracic surgery needs reappraisal.
In the current study, the reoperation rate was higher (4.6%) than that reported in other series (1.2-3.7%) (3-7). In contrary, mortality rate of reoperations was lower (6.1%) when compared with the mortality rate reported in other published series (13.3-37.7%) (3,5,11). Fatal outcome in the present series was associated with the need to proceed with completion pneumonectomy for persistent atelectasis of the remaining lung after lobectomy. The use of completion pneumonectomy for failure of the residual lung to re-expand after primary surgery is reported by Montesano et al. [2002] to carry a high risk for fatal outcome (8). Guggino et al. reported also high mortality rate for completions pneumonectomies performed for complications of the initial treatment or as emergent/salvage procedure (12).
Reoperation is generally agreed to be necessary for the management of particular complications (i.e., bleeding, bronchopleural fistula, empyema), while it is debatable for others, especially in the management of prolonged air leak (1,2). In our practice we do not hesitate to reoperate if the prolonged air leak does not resolve with conservative treatment, if the lung fails to fully expand after discontinuation of the applied suction to the chest tubes and if the modern digital suction systems—such as the Thopaz system used in our unit—show a stable trend of the air leak during several (>5) days. One-way intrabronchial valves are not used in our practice to control a prolonged air leak, as their application with this indication still remains questionable and the success not well documented (13). The majority of patients with prolonged air leak are usually well recovered from the initial operation and otherwise fully ambulatory, making that way the complication very embarrassing for the patient. In addition, we have to outline that discharge of a patient with a Heimlich valve and other similar maneuvers are not an acceptable option in our policy (2,14,15). Reoperations for prolonged air leak with proper indications in the current study allowed fast recovery and discharge of the patient from the hospital (median hospital stay after reoperation: 5.5 days). This fact is very important for patients who will undergo adjuvant treatment for malignancy or for patients undergoing surgery for benign conditions and need to return soon to their daily activities. According to Varela et al. [2005], prolonged air leaks are associated with significant morbidity and increased hospital cost and therefore aggressive surgical management should be performed with proper indications to shorten the hospital stay (16). According to the results of our study, bullectomy and right upper lobectomy are the most vulnerable procedures for prolonged air leak. Right upper lobectomy has increased risk for prolonged air leak because probably of the two interlobar fissures to divide and especially if the lungs exhibit emphysematic changes.
The surgical management of complications requiring reoperation is debatable among thoracic surgeons and hence the aim of the current study is not to support or to propose any strategy or technique of management. The study focuses mainly on the rate of complications requiring reoperation and on the detection of possible predisposing factors for reoperation. The possible different opinions, strategies and surgical techniques used to resolve the complication or even the appropriate time to proceed with reoperation are out of the scope of the current study and they are not discussed.
Technical failures during the initial procedures were detected to be the major predisposing factor for reoperation, accounting for half of the cases in the present series. Even though technical issues can be involved as the cause of reoperation in any case, the presence of some clinical conditions or the use of antiplatelet agents was found to be related in the development of complications which required reoperation. Lung emphysematous changes are a well know predisposing factor for prolonged air leak, while initial surgery for lung and pleural infections is involved in wound suppuration and dehiscence, postoperative empyema and the formation of bronchopleural fistula (2,17,18). Moreover, lung infections are responsible for difficult to manage local conditions (heavy adhesions, fragile tissues, enlarge bronchial vessels) within the operated hemithorax which predispose to technical failures during surgery. Failure of bronchial or lung parenchyma stapling, if not recognized during surgery, can lead to serious postoperative problems mainly that of prolonged air leak and bronchopleural fistula (1,2). Especially, inappropriate stapling is very important during VATS procedures, where the stapling line is quite difficult to be checked for air leak after re-inflation of the lung. Thoracotomy wound dehiscence following wound suppuration is an uncommon complication after general thoracic surgery procedures, which in their majority are clean operations. Thus, surgery for lung and pleural infections (clean-contaminated or contaminated operations) is the commonest cause for the development of thoracotomy wound suppuration and dehiscence. Ischemia of a muscle flap used to cover a chest wall defect is mostly dependent to technical factors during mobilization or fixation of the flap (19).
The ongoing use of antiplatelets in medicine in order to prevent thrombosis in various cardiovascular pathologies seriously affects hemostasis in surgery, even if they are discontinued five days before surgery. Discontinuation of the antiplatelets carries high risk of thrombosis, especially in the clinical setting of drug eluting stents into coronary arteries and hence the most common policy is the discontinuation of antiplatelets and their replacement for a short-time before operation by a bridging agent, such as low molecular weight heparins (20). However, despite this policy platelet function is not completely normal during surgery and non-surgical bleeding from any raw surface in the operative field can occur. Areas of adhesiolysis, pleurectomy, mediastinal lymph node dissection, thoracotomy wound and site of chest tube insertion are the vulnerable sites for bleeding and accumulation of blood clots within the chest in general thoracic surgery. In a recently published [2012] retrospective series by Stock et al., the authors found that reoperation for bleeding rate was similar in patients who discontinued clopidogrel, either with the use of a bridging agent or without a bridging agent, as also in patients who underwent thoracic surgery on clopidogrel. However, in this series, general thoracic surgery performed under clopidogrel administration was related with increasing rate of postoperative blood transfusions (21). In our series, clopidogrel administration before surgery or re-administration after surgery was related with 4 out of the 8 reoperations for accumulation of blood clots within the hemithorax, where a site of active bleeding was not found during reopening of the thorax.
The main limitations of the current study are the retrospective design and the relatively small number of the included patients. In addition, the number of the included esophageal procedures is very low, not allowing conclusions for the specific complications of esophagectomy requiring reintervention (i.e., anastomotic dehiscence). Video-assisted lobectomies and thymectomies are not also included in the performed VATS procedures during the study period, which could possibly affect the low complications rate (2%) of thoracoscopic procedures observed in the study. On the other hand, with the exception of esophagectomies as well VATS lobectomies and thymectomies, the case-mix includes procedures from the whole spectrum of modern general thoracic surgery, allowing the extraction of safe conclusions concerning the type or complications requiring reoperation for their management in the contemporary practice.
In conclusion, the reoperation rate for the management of postoperative complications in the era of modern thoracic surgery is low and they are mostly observed after thoracotomy. Bleeding and clotted hemothoraces are the leading cause of reoperation and the use of antiplatelets, even if discontinued before surgery, is highly involved in the development of such complications. Infectious lung and pleura during the initial operation predispose to the development of postoperative empyema, bronchopleural fistula, wound suppuration and dehiscence. Emphysematic changes in the lungs predispose to the development of prolonged air leak. Despite the recognition of some predisposing conditions for major complications requiring redo-thoracic surgery to control, technical failures during the initial operation are the major source for the development of such complications. Completion pneumonectomy that is performed in the postoperative period as emergent procedure for the management of persistent atelectasis of the residual lobe is associated with high risk for fatal outcome.
Acknowledgements
Disclosure: The authors declare no conflict of interest.
References
- Pairolero PC, Payne SW. Postoperative care and complications in the thoracic surgery patient. In: Baue AE, Geha AS, Hammond GL, et al. eds. Glenn’s Thoracic and Cardiovascular Surgery. Appleton and Lange, 1991: 31-43.
- Faber P, Piccione W Jr. Complications of surgery in the lung cancer patient. In: Pass HI, Mitchell JB, Johnson DH, et al. eds. Lung cancer: principles and practice. Philadelphia: Lippincott Raven Publishers, 1996: 615-32.
- Plaksin SA, Petrov ME. Early rethoracotomies for diseases and chest traumas. Vestn Khir Im I I Grek 2012;171:20-3. [PubMed]
- Parshin VD, Biriukov IuV, Gudovskiĭ AM, et al. Rethoracotomy in thoracic surgery. Khirurgiia (Mosk) 2012;4-9. [PubMed]
- Sirbu H, Busch T, Aleksic I, et al. Chest re-exploration for complications after lung surgery. Thorac Cardiovasc Surg 1999;47:73-6. [PubMed]
- Pálffy G, Forrai I, Csekeö A, et al. Analysis of reoperations after 10,000 lung resections. Zentralbl Chir 1984;109:72-80. [PubMed]
- Van’t Westeinde SC, Horeweg N, De Leyn P, et al. Complications following lung surgery in the Dutch-Belgian randomized lung cancer screening trial. Eur J Cardiothorac Surg 2012;42:420-9. [PubMed]
- Montesano G, Nasti AG, Angelici AM, et al. Reoperations in lung surgery. Ann Ital Chir 2002;73:113-6; discussion 116-7. [PubMed]
- Sadovnikov AA, Kuznetsova AD. Rethoracotomies in the postoperative period. Vestn Khir Im I I Grek 1991;146:86-8. [PubMed]
- Thetter O, Habekost M, Izbicki JR. Rethoracotomy after lung resection. Langenbecks Arch Chir Suppl Kongressbd 1991;161-5. [PubMed]
- Ermolov AS, Stonogin VD. Rethoracotomy because of hemorrhage in the early postoperative period after operations on the lungs and mediastinal organs. Vestn Khir Im I I Grek 1996;155:67-70. [PubMed]
- Guggino G, Doddoli C, Barlesi F, et al. Completion pneumonectomy in cancer patients: experience with 55 cases. Eur J Cardiothorac Surg 2004;25:449-55. [PubMed]
- Mahajan AK, Doeing DC, Hogarth DK. Isolation of persistent air leaks and placement of intrabronchial valves. J Thorac Cardiovasc Surg 2013;145:626-30. [PubMed]
- Waller DA, Edwards JG, Rajesh PB. A physiological comparison of flutter valve drainage bags and underwater seal systems for postoperative air leaks. Thorax 1999;54:442-3. [PubMed]
- Cerfolio RJ, Bass C, Katholi CR. Prospective randomized trial compares suction versus water seal for air leaks. Ann Thorac Surg 2001;71:1613-7. [PubMed]
- Varela G, Jiménez MF, Novoa N, et al. Estimating hospital costs attributable to prolonged air leak in pulmonary lobectomy. Eur J Cardiothorac Surg 2005;27:329-33. [PubMed]
- Petrella F, Rizzo S, Radice D, et al. Predicting prolonged air leak after standard pulmonary lobectomy: computed tomography assessment and risk factors stratification. Surgeon 2011;9:72-7. [PubMed]
- Nadir A, Kaptanoglu M, Sahin E, et al. Post-thoracotomy wound separation (DEHISCENCE): a disturbing complication. Clinics (Sao Paulo) 2013;68:1-4. [PubMed]
- Chang RR, Mehrara BJ, Hu QY, et al. Reconstruction of complex oncologic chest wall defects: a 10-year experience. Ann Plast Surg 2004;52:471-9; discussion 479. [PubMed]
- Singla S, Sachdeva R, Uretsky BF. The risk of adverse cardiac and bleeding events following noncardiac surgery relative to antiplatelet therapy in patients with prior percutaneous coronary intervention. J Am Coll Cardiol 2012;60:2005-16. [PubMed]
- Paul S, Stock C, Chiu YL, et al. Management and outcomes of patients on preoperative plavix (clopidogrel) undergoing general thoracic surgery. Thorac Cardiovasc Surg 2013;61:489-95. [PubMed]


