Application of triple-chimney technique using C-TAG and Viabahn or Excluder iliac extension in TEVAR treatment of aortic arch dilation diseases
Introduction
Thoracic endovascular aortic repair (TEVAR), which is associated with lower mortality and morbidity, has been established as the preferred treatment strategy for aneurysms and dissection of the descending thoracic aorta due to its less invasive characteristics (1). The essential requirement of TEVAR is to have an enough proximal landing zone (PLZ) longer than 15 mm (2,3). Therefore, aortic dilation diseases, including aortic aneurysm, dissection and pseudo-aneurysm involving aortic arch are still not suitable for treatment with standard TEVAR because of an insufficient PLZ (4). Consequently, to obtain an adequate PLZ length and ensure satisfactory sealing, the supra-aortic branches have to be sacrificed (5,6). Numerous approaches have been applied to preserve the blood perfusion of the supra-aortic branches while providing a sufficient landing zone (7), including the hybrid technique, branched devices, fenestrated devices and others (8). However, these methods are not without limitations. Hybrid techniques, including debranching or extra-anatomical bypass, still need thoracic or cervical incision and carry the risk of prosthetic vessel infection. While these above mentioned novel endografts are still under development and are not readily available, especially in emergent circumstances (9). An alternative modality is the chimney technique that features the deployment of parallel grafts from each vital branch vessel into the thoracic aorta (10). This technique was first reported by Greenberg et al. (11) and was later performed by various groups for visceral and supra-aortic branch preservation (12). Many centres have attempted to combine TEVAR with the chimney technique to preserve perfusion in the left subclavian artery (LSA) or in the LSA and left common carotid artery (LCCA) simultaneously and have achieved satisfactory outcomes (4,13). To the best of our knowledge, the triple-chimney technique has not been frequently utilized in TEVAR for the management of aortic arch dilation diseases. The objective of this study was to report the experience of a single centre on the application of the triple-chimney technique using C-TAG together with Viabahn or Excluder iliac extension devices for the endovascular management of aortic arch dilation diseases (Figure 1).
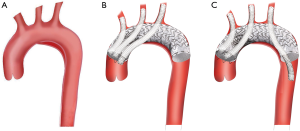
Methods
Inclusion criteria
The Institutional Research Ethic Committee Review Board of our hospital approved this study. Written informed consent was obtained from each patient. From July 2016 to August 2017, patients fulfilling the inclusion criteria were enrolled in this study. The inclusion criteria were as follows: (I) lesions involving zone 0–2; (II) elderly patients or those with multiple comorbidities considered to pose high surgical risk; (III) patients with previous thoracic open surgery; and (IV) patients who refused open thoracotomy treatment and were willing to undergo the triple-chimney technique.
Treatment procedure
All patients were diagnosed via contrast-enhanced CT scan (Somatom Definition Flash, Siemens, Germany). The 3 Mensio work station (Medical Imaging B.V., Bilthoven, Netherlands) was utilized for reconstruction and measurement of the landing zone length, the diameters of the landing zone and supra-aortic branches based on centre-line images. Procedures were performed under general anaesthesia in a hybrid suite. Intraoperatively, the left elbow and bilateral neck area were cut down to expose the left brachial artery, as well as the left and right carotid arteries. After puncture, sheathes were placed to establish access, and 0.035 Amplatz guidewires (Boston Scientific, Heredia, Costa Rica) were advanced into the ascending aorta through each sheath. Femoral artery access was established using the Seldinger puncture technique, and two Perclose ProGglide devices (Abbott Vascular, Santa Clara, CA, USA) were pre-placed. A 0.035-in Terumo guide wire (Terumo Corporation, Tokyo, Japan) and a 5 F pigtail catheter (Cordis Corporation, Miami Lakes, FL, USA) were advanced into the ascending aorta through the right femoral artery sheath. The Terumo guide wire and MPA catheter (Cordis Corporation, Miami Lakes, FL, USA) were advanced into the LSA through the left femoral artery sheath, and the Terumo guide wire was exchanged into a Supracore guidewire (Abbott Vascular, Santa Clara, CA, USA). Then, angiography was performed to check the lesion, the ostium of the supra-aortic branches and the coronary arteries. A Lunderquist guidewire (Cook Medical, Bjaeverskov, Denmark) and a Gore sheath (W.L. Gore and Assoc. Inc., Flagstaff, AZ, USA) were exchanged through the right femoral artery. Afterwards, a C-TAG (W.L. Gore and Assoc. Inc., Flagstaff, AZ, USA) stent-graft was advanced into the aortic arch. Three chimney stents were advanced through the pre-placed stiff wire into the ascending aorta with their proximal ends extending 5–10 mm beyond the stent-graft (three forward chimneys), and the distal ends extended approximately 20 mm into the supra-aortic branches. The C-TAG stent-graft was deployed first, followed by immediate deployment of the chimney stents. An aortic balloon and peripheral balloons were placed into the aortic and chimney stents for simultaneous dilation. A final angiogram was performed to confirm the sealing of the lesion, accuracy of chimney stent’s placement and blood flow in the supra-aortic branches. All patients were scheduled for follow-up assessment, including routine office visits and contrast CT scan at 1, 3, 6, and 12 months after procedure and annually thereafter.
Results
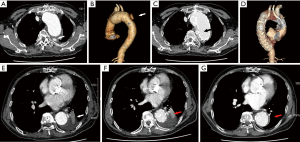
Of all, 7 patients (5 males) with an average age of 56.1±10.8 years, including 4 with aortic arch aneurysms, 2 with acute aortic dissections and 1 with a type I endoleak after previous TEVAR, were treated using the triple-chimney technique in our centre (Table 1). Among these, 6 innominate arteries (IAs) were deployed with Excluder iliac extensions, and one with a Viabahn cover-stent. All LCCAs and LSAs were placed with Viabahn cover-stents. Reverse chimney technique was applied in 4 patients to preserve the LSA. Three (42.0%) type I endoleaks were found in the completion angiogram. Two of these endoleaks were apparently reduced, and one completely disappeared after balloon dilation. In one patient of these three (completely disappeared), type I endoleak was detected again by the CTA check upon discharge at 3 days after operation (Figure 2) , but this patient refuse to take any further treatment. One patient suffered from iliac artery rupture during Gore sheath withdrawal and was managed by laparotomy and prosthetic vessel reconstruction. All patients were scheduled for regular follow-up evaluation. The median follow-up time was 15.7 months (9–20 months). One patient died of myocardial infarction at 6 months after discharge. The type I endoleak detected by pre-discharging CTA check in one patient was found completely disappeared at the 3-month’s follow up (Figure 2). The type I endoleak in other two patients (reduced by balloon dilatation) was found disappeared by CT scan at 1-month’s follow up. No other complications, including stent-graft migration, occlusion, type II endoleak or neurological complications occurred (Table 2).

Full table

Full table
Discussion
The triple-chimney technique features endovascular chimney-stents deployment parallel with the main aortic endograft to preserve the flow to supra-aortic branches and to permit proximal extension of landing zones (14). It has shown positive results with high technical success and satisfactory patency rate of preserving supra-aortic branches and visceral arteries (13,15). This technique provides an alternative strategy that appears to be less invasive with lower mortality and morbidity comparing to open surgery (9,16,17) and hybrid techniques (6,18). The hybrid approach consists of various types of supra-aortic vessel debranching and reconstruction manoeuvres, and the complexity and invasiveness are notably increased because the PLZ is near the aortic valve. The chimney technique is helpful to avoid reconstruction by extra-anatomic bypasses. Due to the customized design characteristic of the fenestrated and branched grafts, at least 4 to 6 weeks are required to prepare for the procedure, making it extremely costly. Therefore, these new devices couldn’t be applied in emergency cases or symptomatic patients. Currently, the triple-chimney technique is the only total endovascular approach available in emergency situations (19,20).
Indications for the triple-chimney technique with TEVAR remain unclear. Theoretically, it is suitable for patients whose lesions involve zone 0 and no other better option is available or for those in whom a pending rupture is present, requiring emergent repair (6).
The triple-chimney technique has the advantage of fewer surgical injuries than open surgery and hybrid techniques and is associated with lower medical expenses and less operating difficulty comparing with fenestrated and branched devices. Nevertheless, several concerns are associated with this technique. The most serious one is the risk of type I endoleak through the gutters among the aortic wall, aortic main body and chimney stents revascularizing the supra-aortic branches (21). In a clinical and systematic review, Shahverdyan et al. (9) reported that the incidence of type I endoleak was 19.7%. Several methods may be useful to decrease the incidence of type I endoleak. The first one is to deploy slightly more oversized aortic stent-grafts and chimney stents. Some authors recommended that oversize should be approximately 20–30% for aortic stent-grafts and 15–20% for chimney stents (9,22). In our series, diameter of the main body was chosen basically according to the formula proposed by Chou et al. (23). Of these 7 patients, the oversize of the main body and branch chimney stents were all larger than 20%. Aiming to reduce gutter further, another useful way is to select a combination of main body and chimney stents with the same design and compliant metal struts. The C-TAG is a flexible endograft specifically designed to conform to the geometry of the aortic arch (24). The chimney stents used in our report were Excluder iliac extension and Viabahn cover-stent, also from Gore. These stents have the same design (nitinol outer skeleton and expanded polytetrafluoroethylene fabric) (25) that provide high flexibility and adaptability (9), which is theoretically advantageous because it allows them to coexist without being compressed during deployment and kissing balloon dilation. A study described displacement of the main endograft by the chimney stent during its deployment, resulting in the development of the “gutter” and leading to endoleak (12). To address this issue, the aortic balloon and peripheral balloons were placed into aortic endografts and chimney stents for kissing dilation. In our series, the reverse chimney technique was applied in four patients. Chimney stents were deployed in parallel in the IA and LCCA, and the reverse chimney technique (periscope) was applied in the LSA (Figure 3). This distribution avoids the potential overlap of three forward chimneys and displacement of the aortic endograft, adding benefit by reducing the gutter and decreasing the occurrence of endoleak. The blood flow in the gutter is slow due to its narrowness, so that the chance of sealing is proportional to the length of this blood flow in the overlapping segment (6). Therefore, to induce thrombosis, a longer gutter length is suggested. Some experts recommended an overlap of at least 2 to 3 cm between the endograft and chimney stents (26). Regarding the reverse chimney, the length of overlap should be much longer than the 2 cm requirement, which would slow the retrograde manner of blood flow further in the gutter from the distal aorta and promote thrombosis formation.
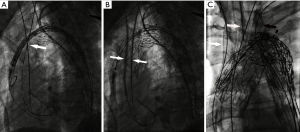
Another serious complication is the cerebrovascular stroke, which is common for any surgery involving the aortic arch. The incidence of stroke for any such surgery is 5% to 12%, according to Thomas et al. (27). Hogendoorn et al. (20) reported that the incidence of stroke was 5.3%, which is mainly due to the occurrence of cerebral vascular embolization caused by aortic embolism.
Mangialardi et al. (10) analysed their 11-year experience of performing the single or double chimney technique. They stated that the occurrence of stroke was related to the excessive operation of guidewires and catheters in the aortic arch, resulting in the dislodgement of mural thrombi. In theory, the triple-chimney technique can increase the incidence of stroke due to multiple manipulations of the guidewires and catheters. In addition, other risk factors including a “shaggy” aorta, stenosis in the carotid artery (20,21,28), cardiac insufficiency (14), and air embolism originating from the circulatory system and coverage of branch vessels (29) would also increase the incidence of neurological complications.
Regarding the aortic arch branches, the IA and LCCA obviously must be preserved in any situation; however, revascularization of the LSA remains controversial. According to the literatures, if the cerebral arterial circle (circle of Willis) is complete and the right vertebral artery is dominant, 90% of patients with LSA coverage will have no incidence of cerebral ischaemia or LSA steal syndrome (30,31). Nevertheless, the Society for Vascular Surgery (SVS) (32) practice guidelines states that coverage of the LSA might increase the risk of postoperative stroke and spinal cord ischaemia (33,34). Due to the concern of ischemia of cerebellar circulation, spinal cord and left upper limb, in our 7 patients, all the LSA are preserved and reverse chimney technique (periscope) was applied in 4 patients.
No neurological ischaemic-related complications occurred in our serials. Several reasons might contribute to this result. First is the average age of our group was relatively young (56.1±10.8 years). In addition, all patients in our group had undergone a comprehensive evaluation before surgery, and none had severe atherosclerotic degeneration. Furthermore, careful irrigation of the deployment system, meticulous manipulation of the guidewire and catheter during the procedure may help to avoid air emboli and mural thrombosis displacement. Although no neurological complications occurred in our series, they cannot be ignored in the long-term clinical application of the triple-chimney technique.
Our initial data demonstrate that the triple-chimney technique has acceptable short- and mid-term outcomes. However, the long-term durability remains unclear, mainly related to the continuous interactions of the endograft, chimney stent and aortic wall. Moreover, this interactions would be more complex in tortuous arches with high velocity pulsatile status (6) and respiratory movement (14), which may increase the incidence of retrograde type A dissection (35,36) and the risk of intimal tears (6). Long-term follow-up is particularly important in terms of observing the consequences of these interactions.
Conclusions
The triple-chimney technique using the C-TAG and Viabahn or Excluder iliac extension devices in the TEVAR treatment as an endovascular alternative to preserve supra-aortic branches appears to be safe, feasible, and effective and is associated with low mortality and morbidity. Type I endoleak is the main peri-procedural concern, and it should be detected and treated expeditiously. Despite these encouraging preliminary results, long-term durability and efficacy must be investigated in larger cohorts of patients before this advanced technique can be widely applied.
Acknowledgements
Funding: This work was supported by the National Natural Science Foundation of China (Grant number: 81570438) and Shanghai Science and Technology Commission Innovation Fund (Grant number: 18441902400).
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethics Statement: The study was approved by the Institutional Review Board of Zhongshan Hospital, and written informed consent was obtained from all patients.
References
- Bavaria JE, Appoo JJ, Makaroun MS, et al. Endovascular stent grafting versus open surgical repair of descending thoracic aortic aneurysms in low-risk patients: A multicenter comparative trial. J Thorac Cardiovasc Surg 2007;133:369-77.e4. [Crossref] [PubMed]
- Caronno R, Piffaretti G, Tozzi M, et al. Intentional coverage of the left subclavian artery during endovascular stent graft repair for thoracic aortic disease. Surg Endosc 2006;20:915-8. [Crossref] [PubMed]
- Peterson BG, Eskandari MK, Gleason TG, et al. Utility of left subclavian artery revascularization in association with endoluminal repair of acute and chronic thoracic aortic pathology. J Vasc Surg 2006;43:433-9. [Crossref] [PubMed]
- Yang J, Xiong J, Liu X, et al. Endovascular Chimney Technique of Aortic Arch Pathologies: A Systematic Review. Ann Vasc Surg 2012;26:1014-21. [Crossref] [PubMed]
- Malina M, Resch T, Sonesson B. EVAR and complex anatomy: an update on fenestrated and branched stent grafts. Scand J Surg 2008;97:195-204. [Crossref] [PubMed]
- Mangialardi N, Ronchey S, Malaj A, et al. Value and limitations of chimney grafts to treat arch lesions. J Cardiovasc Surg (Torino) 2015;56:503-11. [PubMed]
- Schoder M, Lammer J, Czerny M. Endovascular Aortic Arch Repair: Hopes and Certainties. Eur J Vasc Endovasc Surg 2009;38:255-61. [Crossref] [PubMed]
- Meng Q, Shi Z, Huang H, et al. Clinical experience of preprocedural fenestrated technique during TEVAR for aortic arch disease to preserve supra-aortic branch. J Surg Concepts Pract 2017;22:322-6.
- Shahverdyan R, Gawenda M, Brunkwall J. Triple-barrel Graft as a Novel Strategy to Preserve Supra-aortic Branches in Arch-TEVAR Procedures: Clinical Study and Systematic Review. Eur J Vasc Endovasc Surg 2013;45:28-35. [Crossref] [PubMed]
- Mangialardi N, Serrao E, Kasemi H, et al. Chimney technique for aortic arch pathologies: an 11-year single-center experience. J Endovasc Ther 2014;21:312-23. [Crossref] [PubMed]
- Greenberg RK, Clair D, Srivastava S, et al. Should patients with challenging anatomy be offered endovascular aneurysm repair? J Vasc Surg 2003;38:990-6. [Crossref] [PubMed]
- Larzon T, Gruber G, Friberg O, et al. Experiences of intentional carotid stenting in endovascular repair of aortic arch aneurysms--two case reports. Eur J Vasc Endovasc Surg 2005;30:147-51. [Crossref] [PubMed]
- Zhao Y, Shi Y, Wang M, et al. Chimney Technique in Supra-Aortic Branch Reconstruction in China: A Systematic and Critical Review of Chinese Published Experience. Vasc Endovascular Surg 2017;51:429-35. [Crossref] [PubMed]
- Gehringhoff B, Torsello G, Pitoulias G, et al. Use of Chimney Grafts in Aortic Arch Pathologies Involving the Supra-Aortic Branches. J Endovasc Ther 2011;18:650-5. [Crossref] [PubMed]
- Donas KP, Pecoraro F, Torsello G, et al. Use of covered chimney stents for pararenal aortic pathologies is safe and feasible with excellent patency and low incidence of endoleaks. J Vasc Surg 2012;55:659-65. [Crossref] [PubMed]
- Strauch JT, Spielvogel D, Lauten A, et al. Technical advances in total aortic arch replacement. Ann Thorac Surg 2004;77:581-90. [Crossref] [PubMed]
- Walsh SR, Tang TY, Sadat U, et al. Endovascular stenting versus open surgery for thoracic aortic disease: Systematic review and meta-analysis of perioperative results. J Vasc Surg 2008;47:1094-8.e3. [Crossref] [PubMed]
- Chiesa R, Tshomba Y, Melissano G, et al. Hybrid approach to thoracoabdominal aortic aneurysms in patients with prior aortic surgery. J Vasc Surg 2007;45:1128-35. [Crossref] [PubMed]
- Sugiura K SB, Akesson M, et al. The applicability of chimney grafts in the aortic arch. J Cardiovasc Surg (Torino) 2009;50:475-81. [PubMed]
- Hogendoorn W, Schlosser FJ, Moll FL, et al. Thoracic endovascular aortic repair with the chimney graft technique. J Vasc Surg 2013;58:502-11. [Crossref] [PubMed]
- Wang T, Shu C, Li QM, et al. First experience with the double chimney technique in the treatment of aortic arch diseases. J Vasc Surg 2017;66:1018-27. [Crossref] [PubMed]
- Yoshida RA, Kolvenbach R, Yoshida WB, et al. Total endovascular debranching of the aortic arch. Eur J Vasc Endovasc Surg 2011;42:627-30. [Crossref] [PubMed]
- Chou HW, Chan CY, Wang SS, et al. How to size the main aortic endograft in a chimney procedure. J Thorac Cardiovasc Surg 2014;147:1099-101. [Crossref] [PubMed]
- Bockler D, Brunkwall J, Taylor PR, et al. Thoracic Endovascular Aortic Repair of Aortic Arch Pathologies with the Conformable Thoracic Aortic Graft: Early and 2 year Results from a European Multicentre Registry. Eur J Vasc Endovasc Surg 2016;51:791-800. [Crossref] [PubMed]
- Möllenhoff C, Katsargyris A, Steinbauer M, et al. Current status of Hemobahn/Viabahn endografts for treatment of popliteal aneurysms. J Cardiovasc Surg (Torino) 2013;54:785-91. [PubMed]
- Lachat M, Frauenfelder T, Mayer D, et al. Complete Endovascular Renal and Visceral Artery Revascularization and Exclusion of a Ruptured Type IV Thoracoabdominal Aortic Aneurysm. J Endovasc Ther 2010;17:216-20. [Crossref] [PubMed]
- Thomas M, Li Z, Cook DJ, et al. Contemporary results of open aortic arch surgery. J Thorac Cardiovasc Surg 2012;144:838-44. [Crossref] [PubMed]
- Cires G, Noll RE Jr, Albuquerque FC Jr, et al. Endovascular debranching of the aortic arch during thoracic endograft repair. J Vasc Surg 2011;53:1485-91. [Crossref] [PubMed]
- Spear R, Haulon S, Ohki T, et al. Editor's Choice - Subsequent Results for Arch Aneurysm Repair with Inner Branched Endografts. Eur J Vasc Endovasc Surg 2016;51:380-5. [Crossref] [PubMed]
- Liu H, Shu C, Li X, et al. Endovascular aortic repair combined with chimney technique in the treatment of stanford type B aortic dissection involving aortic arch. Ann Vasc Surg 2015;29:758-63. [Crossref] [PubMed]
- Li Y, Hu Z, Wang J, et al. Endovascular Chimney Technique for Aortic Arch Pathologies Treatment: A Systematic Review and Meta-Analysis. Ann Vasc Surg 2018;47:305-15. [Crossref] [PubMed]
- Matsumura JS, Lee WA, Mitchell RS, et al. The Society for Vascular Surgery Practice Guidelines: management of the left subclavian artery with thoracic endovascular aortic repair. J Vasc Surg 2009;50:1155-8. [Crossref] [PubMed]
- Chung J, Kasirajan K, Veeraswamy RK, et al. Left subclavian artery coverage during thoracic endovascular aortic repair and risk of perioperative stroke or death. J Vasc Surg 2011;54:979-84. [Crossref] [PubMed]
- Patterson BO, Holt PJ, Nienaber C, et al. Management of the left subclavian artery and neurologic complications after thoracic endovascular aortic repair. J Vasc Surg 2014;60:1491-8.e1. [Crossref] [PubMed]
- Dong ZH, Fu WG, Wang YQ, et al. Retrograde type A aortic dissection after endovascular stent graft placement for treatment of type B dissection. Circulation 2009;119:735-41. [Crossref] [PubMed]
- O'Callaghan A, Mastracci TM, Greenberg RK, et al. Outcomes for supra-aortic branch vessel stenting in the treatment of thoracic aortic disease. J Vasc Surg 2014;60:914-20. [Crossref] [PubMed]


