Lung volume reduction followed by lung transplantation—considerations on selection criteria and outcome
Introduction
As chronic obstructive pulmonary disease (COPD) remains one of the leading causes of morbidity and mortality in the developed world, tremendous effort has been undertaken to improve medical therapy and surgical treatment options for affected patients. Surgical treatment options in end-stage COPD patients are lung volume reduction surgery (LVRS) and lung transplantation (LuTX). Bronchoscopic LVR techniques have evolved in parallel, however no prospective randomized data comparing surgical and bronchoscopic LVR techniques are available yet. LVR is considered independent of LuTX however might also serve as a means to delay the listing for LuTX and bridge patients to the transplant procedure. The choice which procedure is suitable in individual situations has been highly debated in literature and different algorithms have been suggested for individual decision-making (1). It is unanimously accepted that LVRS does not preclude subsequent LuTX. The first report on LVRS as an option to bridge patients to LuTX dates back to 1995 (2). In addition, LVRS has been reported on the contralateral side after previous single lung transplantation (SLuTX) to reduce hyperinflation of the remaining native lung (3). In this review will discuss optimal patient selection, special consideration and published outcomes of patients undergoing both LVR and LuTX.
COPD patient selection for LuTX
Generally, patients with emphysema are referred to LuTX after exhaustion of all other treatment options. It is of utmost importance to select candidates whose quality of life and disease related survival will improve after LuTX (1,4), since particularly in COPD patients the overall survival benefit of LuTX is not as clear as in other indications.
According to the guidelines published by the International Society of Heart and Lung Transplantation (ISHLT) in 2014, patients suffering from emphysema should be referred early to a lung transplant for assessment of transplant suitability (5). Criteria for referral are: ongoing progression of disease despite maximum therapy, hypercapnia (paCO2 >50 mmHg), hypoxemia (paO2 <60 mmHg) or a significantly reduced lung function (FEV1 <25% of predicted). Alternatively, a BODE index of or above five has been suggested as a suitable threshold for referral (6). Further, those guidelines suggest a listing for LuTX when certain functional criteria are fulfilled. The proposed criteria are: heavily reduced lung function (FEV1 <20% of predicted), frequent exacerbations (≥3/Y), hypercapnic respiratory failure, a BODE Index ≥7 or an associated pulmonary hypertension (only one criterion needed). However, the optimal timing for listing is also depending on the local organ availability and the allocation algorithm in use.
There are only few absolute contraindications for LuTX, however there is a long list of relative contraindications which have to be considered on an individual basis. Those vary in detail depending on the center approach. Absolute contraindications are: multi organ failure (with the exception of temporary kidney failure in selected patients and planned multi-organ transplantation), recent malignancy, incompliance or an untreatable infectious disease (5,7). Relative contraindications include: age >65 years, obesity, cachexia, osteoporosis, hepatitis/HIV infection, acute respiratory failure (under mechanical ventilation or ECMO) and cardiac comorbidities.
Patient selection for LVRS
To this date, the strongest evidence regarding LVRS derives from the NETT-trial (national emphysema treatment trial) published in 2003 and updated in 2006 after a four-year follow-up (8,9). In this series of 1,218 patients (randomized 1:1 into a LVRS group and a best SOC group) four subgroups were identified based on their differential risk and benefit after LVRS. Their inclusion criteria in terms of patient candidacy for LVRS are still valid and with the knowledge about the patient outcome an algorithm was suggested to decide between LVRS and LuTX (1). Briefly, LVRS should be considered for patients with an upper-lobe predominant emphysema, an FEV1 between 45% and 20% of the predicted value and a DLCO not less than 20% as those patients will have a significant advantage in exercise capacity and dyspnea related quality of life (10). Findings of the NETT trial have been reproduced in a study of the STS database (11) and in the Canadian Lung Volume Reduction Surgery (CLVRS) trial (12). The first, compared post-operative results of 538 patients to the data published in the NETT trial (n=608). Although a significantly higher 30-day mortality was observed in comparison with the NETT non-high-risk subset (5.6% vs. 2.2%; P=0.005), the analysis with the total NETT cohort didn’t showed those differences. This demonstrates the importance of precise and strict patient selection. The latter study assed the long-term survival of patients randomized within the multicentric Canadian Lung Volume Reduction Surgery (CLVRS) trial. Although not significant, an improved median survival was observed for the LVRS group compared to the best-medical-care group (63 vs. 47 months; P=0.2) leading the authors to conclude that LVRS offers better outcomes for patients who survive the initial increased mortality within the early post-operative period.
Even repeat LVRS (Re-LVRS) has been described to be successful in highly selected patients (13). In this series of 22 patients, lung function was improved and breathlessness reduced after Re-LVRS with outcomes comparable by any means (Hospitalization, drainage time; surgical revisions, perioperative mortality) to those after the patients first LVRS.
Recently, all evidence about the effectiveness of LVRS has been analyzed in a systematic review by the Cochrane airways group (14). The currently applied techniques for lung volume reduction surgery have evolved from the initially used sternotomy in the NETT trial towards standard minimally invasive approaches. Recent studies describe a prolonged overall benefit by a staged bilateral approach (15).
LVRS prior to LuTX
Literature research identifies twelve published reports about LuTX after previous LVRS (Table 1) presenting a total of 254 patients (not considering patients included in multiple reports). Those papers which made a comparison between LVRS/LuTX patients (n=201) and sole LuTX patients (n=1,030) differed broadly in terms of patient outcomes which will be depicted hereinafter. However, several confounders limit a structured comparison of those reports. First, the indications for LVRS as well as the choice of procedure (VATS or Sternotomy, uni- or bilaterally, laser vs. stapler, target regions) were not standardized in the pre-NETT era. Secondly, subsequent LuTX had been performed either double sidedly or single sidedly (either contralaterally or ipsilaterally) and according to center specific approaches (thoracotomy vs. clamshell; no mechanical support vs. CPB vs. ECMO). All but one report are retrospective non-randomized analyses of single center experiences which further depicts the scarcity of evidence in this matter.
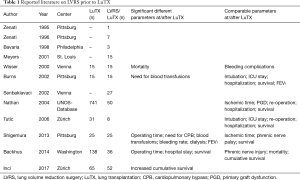
Full table
Impact of LVRS on pre-LuTX physical condition
Early on, LVRS has been postulated to postpone the need for LuTX, making it a valid “bridge to transplantation” (2,16-18). This assumption is substantiated by the fact that patients who had an improvement in FEV1 after LVRS had also a reduction of preoperative PCO2 (17) and a significant increase of their preoperatively reduced BMI (18). Those same “responders to LVRS” showed also a significantly lower 3-month mortality after LuTX. These findings from the group in Vienna were confirmed later on, as patients who were already eligible for LuTX at time of LVRS had a significant improvement in lung function within the first year after LVRS (19).
In a multicenter analysis of the UNOS database comparing 50 patients with LVRS before LuTX with 741 patients transplanted in the same period of time, it has been shown that both groups bared no difference in their disease severity (pulmonary function and pulmonary artery pressure) but that patients with previous LVRS had a slightly longer waiting period (343 vs. 211 days; P=0.014) (20). Further, this analysis showed a significantly higher occurrence of pneumothorax in the LVRS group during the time between listing and transplant (8% vs. 1.5%; P=0.01).
Surgical risks and technical considerations
Although all publications report a higher occurrence of adhesions during LuTX after previous LVRS, their severity has been characterized very differently. The group from Pittsburgh observed moderate to severe adhesions of the chest wall in 92% of LVRS patients (12% in sole LuTX group) and 20% of moderate to severe adhesions to the hilum (0% in sole LuTX) (21). In contrary, the authors of the most recent comparative study described more loose adhesions located in the apex and rare mediastinal adhesions which could be easily mobilized from adjacent structures (22).
As a logical consequence of these adhesions, one would inevitably think of a longer operation time, a longer ischemic time of the graft, a bigger loss of blood, a higher requirement for transfusions and ultimately a higher rate of re-operations due to hemothorax. Interestingly those risk factors and complications had only been observed in a higher rate in three (21,23,24) out of the seven comparative reports.
On the other hand, the four other series (17,19,20,22) showed no increased perioperative risk whatsoever. This considerable variation in terms of intraoperative difficulties and their subsequent implications can possibly be explained by looking at the choice of surgical approach for LVRS, which is unfortunately only reported in four of the present series. It is striking that the center with the lowest peri- and postoperative morbidity (22) performed 94% (n=49/52) of LVRS by means of thoracoscopy (n=3 by thoracotomy, n=0 by sternotomy) compared to the center with the biggest differences in outcome after LVRS/LuTX (21) which used thoracotomy (n=7/25; 28%) and sternotomy (n=4/25; 16%) way more deliberately, supposedly leading to a higher rate of complications.
Another explanation can be seen in the use of mechanical circulatory support during the transplant procedure. Because of the need of full heparinization, cardiopulmonary bypass (CPB) has been associated with a higher risk of bleeding during and after LuTX compared to ECMO or total lack of extracorporeal mechanical support (25-34). Interestingly, those two above mentioned papers are the only ones depicting their use of intra-operative extracorporeal support during LuTX. A higher use of CPB (44% in LVRS/LuTX vs. 16% in sole LuTX) correlated with a higher occurrence of bleeding (21) whereas this was not observed in the series where extracorporeal support was used less (19% in LVRS/LuTX vs. 23% in sole LuTX; not specified if ECMO or CPB) (22).
The risk of injury (and subsequent palsy) of the phrenic nerve during extensive adhesiolysis has been addressed by all authors. Nevertheless, only two groups reported any occurrence of phrenic nerve injury, with one center having had a comparable incidence regardless of prior LVRS (2.2% vs. 5.6%; P=0.3) and the other one observing it in 3 patients with previous LVRS (12% vs. 4% in sole LuTX) (21,23). Those 3 patients had subsequent “phrenic nerve surgery” which was not further defined. To reduce the risk of phrenic nerve injury in cases where the mediastinal pleura is adherent it is possible to incise the diseased lung laterally of the adhesion and thereby leaving little visceral pleura over the nerve (22).
Another factor, which has been described to worsen pleural adhesions, is the use of buttressed staplers (17,18). Some authors postulated that the use of bovine pericardium to reinforce stapler lines would ultimately lead to dense adhesion especially at the diaphragm. Although plausible, this assumption has not been validated by other centers having used pericardium buttressed staplers (16,22,24). Even though buttressing the stapler lines with either PTFE or pericardium was well established until the early 2000 to reduce air leaks after LVRS (35,36) later reports suggested the use of autologous fibrin sealant (37) or bovine albumin (38) to overcome antigenic impact and reduce air-leak more efficiently. Currently a widely adopted method of choice to enforce staple lines in highly emphysematous parenchymal resection is to use bioabsorbable polymer buttress or preloaded buttress materials. Nevertheless, no evidence could be found on the likelihood of any of these materials to adhere to the pleura and thereby their impact in terms of LVRS/LuTX remains still unclear.
Another technique to cover resection lines and to reduce residual intrathoracic space after resection is the so-called pleura tent (39,40). This method was/is being routinely used by individual centers in the context of LVRS (41,42). As there is yet no report about LuTX after such an approach, no pertinent recommendations can be made.
Additional perioperative risks
Although reports on hemodynamic effects on patients before and after LVRS showed mixed results (43-46), patients suffering from pulmonary hypertension are considered to have a high surgical risk for LVRS and should thereby be favored for LuTX over LVRS (1). However, recently a single center analysis from the Zurich group reported on good outcomes of LVRS in a limited series of 10 patients with preoperative systolic pulmonary artery pressures >35 mmHg (47). Within the context of LVRS followed by LuTX, pulmonary hypertension was addressed only once in literature (21). The authors found out in a multivariate analysis that severe pulmonary Hypertension developed after LVRS (>60 mmHg) is as significant risk factor for mortality after subsequent LuTX [OR 1.91 (1.86–2.02) P=0.05]. Further the authors advocated to be “very selective in the use of LVRS as a bridge to later lung transplantation and to provide careful follow-up for patients with prior LVRS, making every effort to perform the lung transplantation before the patient develops severe pulmonary hypertension.”
Primary graft dysfunction (PGD) is an early form of (ischemia/reperfusion) lung injury. It is the major cause of short term morbidity and mortality after LuTX and is associated with a worse long term outcome and more specifically with an earlier occurrence of chronic rejection (48). In all three reports about occurrence of PGD after LVRS/LuTX no significant differences were found compared to a sole LuTX control group (8% vs. 4%, 4% vs. 9% and 7% vs. 9% of PGD Grade 3) (20-22).
Mid- and long-term post-operative outcomes
Two out of three reports providing data on lung function and physical condition of patients after LuTX (17,21,24), showed no significant differences between patients who had LVRS prior to LuTX compared to sole LuTX patients. The Vienna group reported on comparable FEV1 (87.2% vs. 84.3%), TLC (97% vs. 95%), PaO2 (83 vs. 77 mmHg) and BMI (24 vs. 23) in both groups after LuTX. In the report by Burns et al., longitudinal spirometry (up to 34-month follow-up) showed a steady but nonsignificant post-LuTX decline in lung function which was not different between both groups (2-year FEV1: 1.36 vs. 1.6 L; 3-year FEV1: 1.09 vs. 1.43 L). However, in a more recent analysis (LuTX performed between 2002 and 2009) at the same center (21), the authors observed a significantly inferior graft function in patient after LVRS/LUTX (peak FEV1: 57 vs. 79%; peak 6MWT 801 vs. 1,311 ft.; P<0.05). No hypothesis is given to explain such an important difference but supposedly it was associated to the higher rates of re-thoracotomy, phrenic nerve palsy and PGD observed in LVRS/LuTX patients, ultimately leading to worse outcomes.
In terms of post-LuTX survival, presented results were equally heterogeneous. All but one of the reports showed no significant differences between compared group. In those, LVRS/LuTX patients had a 1-year survival ranging from 75% to 100% and a 5-year survival between 63% and 66.2%. In comparison, patients with LuTX alone had a 1-year survival rate between 81% and 87% and a 5-year rate ranging between 61% and 66% respectively. In accordance to that, overall 1-year survival in the UNOS/ISHLT Transplant Registry (1990–2015; n=53,396) was 80.7% and 5-year survival 54.8% (49). Most recent UNOS/ISHLT data (as of January 5, 2018 for Transplants performed between 2013 and 2016) bared an improved 1-year survival of 85.8% (n=10,847).
In one report, LVRS prior to LuTX had a negative impact on survival after transplantation (at 1 and 3 years: 72% and 49% vs. 87% and 66% for LuTX alone; P=0.008) (23). According to the authors, this reduced survival can partially be explained by the longer surgical time and a longer hospital length of stay, also pointing to the higher acuity of these patients. Nevertheless, as the median cumulative survival after LVRS (+LuTX) was statistically comparable with survival of patients undergoing either procedure alone (LVRS/LuTX 104 months; LVRS 103 months; LuTX 96 months) the authors concluded that LVRS was able to rescue more severely affected patients from otherwise reduced survival in the absence of LuTX.
In contrast, the most recent publication (and the one with the most patients in the LVRS/LuTX group; n=52) observed a significantly improved median survival after LVRS followed by LuTX (LVRS/LuTX 143 months; LuTX 86 months; P<0.001) (22). In this cohort LVRS led to the postponement of LuTX with a mean time of 45 months and the authors stated that patient selection is the crucial element for such promising results. Unfortunately, the interpretation of those findings is limited as no data were recorded neither about the initial strategy for LVRS (“definitive therapy” or “bridging therapy”), nor about the decision for LuTX candidacy after LVRS. Thereby, patients who were “bridged” by means of LVRS but ultimately didn’t receive a transplant (either ineligible for LuTX or deceased on waiting list) were not taken into account.
Endoscopic lung volume reduction (ELVR) prior to LuTX
ELVR techniques have been developed and are increasingly being used in the last decade as a less invasive alternative to LVRS. The most popular approach is the use of one-way valves to induce atelectasis of emphysematous lobes, which is however only feasible if no collateral ventilation of the lobes is present. Other methods—with however no adequate evidence to substantiate routine use outside of clinical trials—are the instillation of chemical sealant, the placement of metal coils and thermal ablation of small airways by means of vapor. Aside from three case reports/case series (n=1/4/5) addressing the use of endobronchial valves or sealant in patients subsequently undergoing LuTX (50-52) only one single-center analysis presenting post LuTX data was published so far (53). In this report 20 patients who had ELVR (valves: n=17; vapor therapy n=1; coils n=1; sealant n=1) followed by LuTX after a median time of two years were compared to matched cohort of 40 sole LuTX patients. All of the intraoperative, functional and short-term outcome parameters were comparable between both groups (surgery duration: 252 vs. 260 min; hospitalization 21 vs. 24 days; 6MWT: 397 vs. 380 meters; 1-year survival: 95% vs. 97.5%). Interestingly, patients who had prior ELVR, showed a significantly higher rate of bacterial airway colonization after LuTX (50% vs. 15%; P=0.004) even though this difference was not so apparent before LuTX (25% vs. 10%; P=0.13). Strikingly, Stenotrophomonas maltophilia predominated in the stain cultures (n=4/10). The authors concluded that the higher prevalence of bronchiectasis seen in the CT scans prior to LuTX might potentially explain the higher rate of pathological contamination and that bronchiectasis could possibly have been favored by ELVR as other reports suggested (54,55). Nevertheless, as early outcomes were unaffected by previous ELVR and the possible risk of colonization, further research is necessary to asses any possible impact on long-term survival.
LVR after or during LuTX
Native lung hyperinflation (NLH) is a complication unique to SLuTX for lung emphysema. It is characterized by a radiographic evidence of graft compression and the decrease in lung function and exercise tolerance. Even if the first reports about LVRS as a method of treatment of this condition date back to 1997, only 11 case reports/case series with a total of 44 patients have been published so far (3,56-65). Also, LVRS of the native lung at the same time of LuTX was reported in four patients were hyperinflation was to be expected (65-67). More recently ELVR was used to counteract NLH (51,65,68-71). All reports share a high rate of success in terms of improving lung function and reducing subjective breathlessness of patients affected by NLH. Also, none of those cases had a mortality attributed to the LVR measure itself. All published reports about LVR after SLuTX are summarized in Table 2.
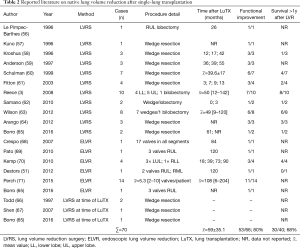
Full table
Discussion
Although LVRS and LuTX are both established adjunctive therapies for patients suffering from severe COPD, criteria for one or the other option differ tremendously. LVRS generally aims at patients with less functional impairment (FEV1 >20, DLCO >20). Nevertheless, a thorough understanding of the indications, contraindications, risks, and benefits of each procedure, as well as the patient’s goals and preferences, should guide the decision-making process (1). Also, previous LVRS followed by (initially not considered) LuTX has been shown a feasible strategy for patients where LVRS didn’t bring the looked-for benefit or the patients’ status deteriorated after an initial improvement. According to the NETT-sub group analysis, patients with an upper-lobe predominant emphysema are those who would benefit from LVRS. This applies also for patients who are already transplant candidates. Thereby LVRS should be considered if the patients would benefit regarding their lung function and nutritional status until a suitable organ is allocated to them.
In literature, LVRS has been shown to delay the need for LuTX in selected patients and to improve their physical condition prior to LuTX giving them “a better start “after LuTX. Although it has been shown in Literature that prior LVRS can increase perioperative risk and thereby mortality after LuTX, todays LVRS practice via VATS (compared to sternotomy and thoracotomy) reduces the occurrence of severe adhesions making a subsequent transplantation less challenging and less risky. Another development which noticeably decreased the risk of bleeding perioperatively is the ongoing paradigm shift regarding the preference of ECMO compared to CPB as intra-operative mechanical support.
As eligibility criteria for LuTX or LVR such as the surgical approach changed substantially over the years it remains difficult to translate past findings to current state-of-the-art. Nevertheless, it is important to know which risk factors can lead to a higher morbidity for LuTX when patients had prior LVRS. Those identified by literature in a single center cohort are (21): advanced age (>65 years), severe pulmonary hypertension (PA >60), prolonged CPB time (>4 hours), high transfusion requirements (>20 units) and “emergency” LuTX after LVRS failure (17). As those observations have not been validated so far, further investigations are needed to clearly identify not only the patients which wouldn’t get a benefit from LVRS but those who would get a higher risk of mortality because of the combination of both treatments.
Endoscopic approaches of LVR are rather new compared to surgical approaches. Despite their less important improvement of functional parameters of the patients, the scarce evidence did not show an increase in perioperative risks or mortality making endoscopic valves a valid alternative to LVRS in highly selected patients.
To conclude, most publications agree on the fact that LVRS does not impair survival after LuTX in patients with severe COPD. As LVRS does not preclude technically the possibility for LuTX it should always be considered for highly selected patients which at best would not only have a QOL and functional benefit from LVRS but possibly a reduced peri-operative risk. Clearly, further multi-centric analysis with comparable approaches is needed to improve decision making and best possible timing.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
- Patel N, DeCamp M, Criner GJ. Lung transplantation and lung volume reduction surgery versus transplantation in chronic obstructive pulmonary disease. Proc Am Thorac Soc 2008;5:447-53. [Crossref] [PubMed]
- Zenati M, Keenan RJ, Landreneau RJ, et al. Lung reduction as bridge to lung transplantation in pulmonary emphysema. Ann Thorac Surg 1995;59:1581-3. [Crossref] [PubMed]
- Reece TB, Mitchell JD, Zamora MR, et al. Native lung volume reduction surgery relieves functional graft compression after single-lung transplantation for chronic obstructive pulmonary disease. J Thorac Cardiovasc Surg 2008;135:931-7. [Crossref] [PubMed]
- De Meester J, Smits JM, Persijn GG, et al. Listing for lung transplantation: life expectancy and transplant effect, stratified by type of end-stage lung disease, the Eurotransplant experience. J Heart Lung Transplant 2001;20:518-24. [Crossref] [PubMed]
- Weill D, Benden C, Corris PA, et al. A consensus document for the selection of lung transplant candidates: 2014--an update from the Pulmonary Transplantation Council of the International Society for Heart and Lung Transplantation. J Heart Lung Transplant 2015;34:1-15. [Crossref] [PubMed]
- Celli BR, Cote CG, Marin JM, et al. The body-mass index, airflow obstruction, dyspnea, and exercise capacity index in chronic obstructive pulmonary disease. N Engl J Med 2004;350:1005-12. [Crossref] [PubMed]
- Orens JB, Estenne M, Arcasoy S, et al. International guidelines for the selection of lung transplant candidates: 2006 update--a consensus report from the Pulmonary Scientific Council of the International Society for Heart and Lung Transplantation. J Heart Lung Transplant 2006;25:745-55. [Crossref] [PubMed]
- Fishman A, Martinez F, Naunheim K, et al. A randomized trial comparing lung-volume-reduction surgery with medical therapy for severe emphysema. N Engl J Med 2003;348:2059-73. [Crossref] [PubMed]
- Naunheim KS, Wood DE, Mohsenifar Z, et al. Long-term follow-up of patients receiving lung-volume-reduction surgery versus medical therapy for severe emphysema by the National Emphysema Treatment Trial Research Group. Ann Thorac Surg 2006;82:431-43. [Crossref] [PubMed]
- Mora JI, Hadjiliadis D. Lung volume reduction surgery and lung transplantation in chronic obstructive pulmonary disease. Int J Chron Obstruct Pulmon Dis 2008;3:629-35. [Crossref] [PubMed]
- Decker MR, Leverson GE, Jaoude WA, et al. Lung volume reduction surgery since the National Emphysema Treatment Trial: study of Society of Thoracic Surgeons Database. J Thorac Cardiovasc Surg 2014;148:2651-8.e1. [Crossref] [PubMed]
- Agzarian J, Miller JD, Kosa SD, et al. Long-term survival analysis of the Canadian Lung Volume Reduction Surgery trial. Ann Thorac Surg 2013;96:1217-22. [Crossref] [PubMed]
- Kostron A, Horn-Tutic M, Franzen D, et al. Repeated lung volume reduction surgery is successful in selected patients. Eur J Cardiothorac Surg 2015;48:710-5. [Crossref] [PubMed]
- van Agteren JE, Carson KV, Tiong LU, et al. Lung volume reduction surgery for diffuse emphysema. Cochrane Database Syst Rev 2016;10. [PubMed]
- Oey IF, Morgan MD, Spyt TJ, et al. Staged bilateral lung volume reduction surgery - the benefits of a patient-led strategy. Eur J Cardiothorac Surg 2010;37:846-52. [Crossref] [PubMed]
- Zenati M, Keenan RJ, Sciurba FC, et al. Role of lung reduction in lung transplant candidates with pulmonary emphysema. Ann Thorac Surg 1996;62:994-9. [Crossref] [PubMed]
- Wisser W, Deviatko E, Simon-Kupilik N, et al. Lung transplantation following lung volume reduction surgery. J Heart Lung Transplant 2000;19:480-7. [Crossref] [PubMed]
- Senbaklavaci O, Wisser W, Ozpeker C, et al. Successful lung volume reduction surgery brings patients into better condition for later lung transplantation. Eur J Cardiothorac Surg 2002;22:363-7. [Crossref] [PubMed]
- Tutic M, Lardinois D, Imfeld S, et al. Lung-volume reduction surgery as an alternative or bridging procedure to lung transplantation. Ann Thorac Surg 2006;82:208-13; discussion 213. [Crossref] [PubMed]
- Nathan SD, Edwards LB, Barnett SD, et al. Outcomes of COPD lung transplant recipients after lung volume reduction surgery. Chest 2004;126:1569-74. [Crossref] [PubMed]
- Shigemura N, Gilbert S, Bhama JK, et al. Lung transplantation after lung volume reduction surgery. Transplantation 2013;96:421-5. [Crossref] [PubMed]
- Inci I, Iskender I, Ehrsam J, et al. Previous lung volume reduction surgery does not negatively affect survival after lung transplantation. Eur J Cardiothorac Surg 2018;53:596-602. [Crossref] [PubMed]
- Backhus L, Sargent J, Cheng A, et al. Outcomes in lung transplantation after previous lung volume reduction surgery in a contemporary cohort. J Thorac Cardiovasc Surg 2014;147:1678-83.e1. [Crossref] [PubMed]
- Burns KE, Keenan RJ, Grgurich WF, et al. Outcomes of lung volume reduction surgery followed by lung transplantation: a matched cohort study. Ann Thorac Surg 2002;73:1587-93. [Crossref] [PubMed]
- Ko WJ, Chen YS, Lee YC. Replacing cardiopulmonary bypass with extracorporeal membrane oxygenation in lung transplantation operations. Artif Organs 2001;25:607-12. [Crossref] [PubMed]
- Aigner C, Wisser W, Taghavi S, et al. Institutional experience with extracorporeal membrane oxygenation in lung transplantation. Eur J Cardiothorac Surg 2007;31:468-73; discussion 473-4. [Crossref] [PubMed]
- Bittner HB, Binner C, Lehmann S, et al. Replacing cardiopulmonary bypass with extracorporeal membrane oxygenation in lung transplantation operations. Eur J Cardiothorac Surg 2007;31:462-7; discussion 467. [Crossref] [PubMed]
- Nagendran M, Maruthappu M, Sugand K. Should double lung transplant be performed with or without cardiopulmonary bypass? Interact Cardiovasc Thorac Surg 2011;12:799-804. [Crossref] [PubMed]
- Ius F, Kuehn C, Tudorache I, et al. Lung transplantation on cardiopulmonary support: venoarterial extracorporeal membrane oxygenation outperformed cardiopulmonary bypass. J Thorac Cardiovasc Surg 2012;144:1510-6. [Crossref] [PubMed]
- Bermudez CA, Shiose A, Esper SA, et al. Outcomes of intraoperative venoarterial extracorporeal membrane oxygenation versus cardiopulmonary bypass during lung transplantation. Ann Thorac Surg 2014;98:1936-42; discussion 1942-3.
- Biscotti M, Yang J, Sonett J, et al. Comparison of extracorporeal membrane oxygenation versus cardiopulmonary bypass for lung transplantation. J Thorac Cardiovasc Surg 2014;148:2410-5. [Crossref] [PubMed]
- Yu WS, Paik HC, Haam SJ, et al. Transition to routine use of venoarterial extracorporeal oxygenation during lung transplantation could improve early outcomes. J Thorac Dis 2016;8:1712-20. [Crossref] [PubMed]
- Magouliotis DE, Tasiopoulou VS, Svokos AA, et al. Extracorporeal membrane oxygenation versus cardiopulmonary bypass during lung transplantation: a meta-analysis. Gen Thorac Cardiovasc Surg 2018;66:38-47. [Crossref] [PubMed]
- Slama A, Besa V, Abu Issa O, et al. Which Extracorporeal Support Should We Use During Lung Transplantation? - Impact of CPB and ECMO on Peri-Operative Outcome. J Heart Lung Transplant 2017;36:S403. [Crossref]
- Stammberger U, Klepetko W, Stamatis G, et al. Buttressing the staple line in lung volume reduction surgery: a randomized three-center study. Ann Thorac Surg 2000;70:1820-5. [Crossref] [PubMed]
- Hazelrigg SR, Boley TM, Naunheim KS, et al. Effect of bovine pericardial strips on air leak after stapled pulmonary resection. Ann Thorac Surg 1997;63:1573-5. [Crossref] [PubMed]
- Moser C, Opitz I, Zhai W, et al. Autologous fibrin sealant reduces the incidence of prolonged air leak and duration of chest tube drainage after lung volume reduction surgery: a prospective randomized blinded study. J Thorac Cardiovasc Surg 2008;136:843-9. [Crossref] [PubMed]
- Rathinam S, Naidu BV, Nanjaiah P, et al. BioGlue and Peri-strips in lung volume reduction surgery: pilot randomised controlled trial. J Cardiothorac Surg 2009;4:37. [Crossref] [PubMed]
- Miscall L, Duffy RW, Nolan RB, et al. The pleural tent as a simultaneous tailoring procedure in combination with pulmonary resection. Am Rev Tuberc 1956;73:831-52. [PubMed]
- Hansen JL. Parietal pleurolysis (the pleural tent) as a simultaneous space-reducing procedure in combination with pulmonary resection. Acta Chir Scand 1957;112:485-8. [PubMed]
- Cooper JD, Patterson GA, Sundaresan RS, et al. Results of 150 consecutive bilateral lung volume reduction procedures in patients with severe emphysema. J Thorac Cardiovasc Surg 1996;112:1319-29; discussion 1329-30. [Crossref] [PubMed]
- Venuta F, De Giacomo T, Rendina EA, et al. Thoracoscopic pleural tent. Ann Thorac Surg 1998;66:1833-4. [Crossref] [PubMed]
- Sciurba FC. Preoperative predictors of outcome following lung volume reduction surgery. Thorax 2002;57 Suppl 2:II47-II52. [PubMed]
- Criner GJ, Scharf SM, Falk JA, et al. Effect of lung volume reduction surgery on resting pulmonary hemodynamics in severe emphysema. Am J Respir Crit Care Med 2007;176:253-60. [Crossref] [PubMed]
- Haniuda M, Kubo K, Fujimoto K, et al. Different effects of lung volume reduction surgery and lobectomy on pulmonary circulation. Ann Surg 2000;231:119-25. [Crossref] [PubMed]
- Weg IL, Rossoff L, McKeon K, et al. Development of pulmonary hypertension after lung volume reduction surgery. Am J Respir Crit Care Med 1999;159:552-6. [Crossref] [PubMed]
- Caviezel C, Aruldas C, Franzen D, et al. Lung volume reduction surgery in selected patients with emphysema and pulmonary hypertension. Eur J Cardiothorac Surg 2018. [Epub ahead of print]. [Crossref] [PubMed]
- Lee JC, Christie JD, Keshavjee S. Primary graft dysfunction: definition, risk factors, short- and long-term outcomes. Semin Respir Crit Care Med 2010;31:161-71. [Crossref] [PubMed]
- Chambers DC, Yusen RD, Cherikh WS, et al. The Registry of the International Society for Heart and Lung Transplantation: Thirty-fourth Adult Lung And Heart-Lung Transplantation Report-2017; Focus Theme: Allograft ischemic time. J Heart Lung Transplant 2017;36:1047-59. [Crossref] [PubMed]
- Venuta F, Diso D, Anile M, et al. Bronchoscopic lung volume reduction as a bridge to lung transplantation in patients with chronic obstructive pulmonary disease. Eur J Cardiothorac Surg 2011;39:364-7. [Crossref] [PubMed]
- Destors M, Aniwidyaningsih W, Jankowski A, et al. Endoscopic volume reduction before or after lung transplantation. Eur J Cardiothorac Surg 2012;42:897-8. [Crossref] [PubMed]
- Fruchter O, Fridel L, Kramer MR. The pathological features of bronchoscopic lung volume reduction using sealant treatment assessed in lung explants of patients who underwent lung transplantation. Respiration 2013;86:143-4. [Crossref] [PubMed]
- Fuehner T, Clajus C, Fuge J, et al. Lung transplantation after endoscopic lung volume reduction. Respiration 2015;90:243-50. [Crossref] [PubMed]
- Herth FJ, Noppen M, Valipour A, et al. Efficacy predictors of lung volume reduction with Zephyr valves in a European cohort. Eur Respir J 2012;39:1334-42. [Crossref] [PubMed]
- Fruchter O, Rosengarten D, Goldberg E, et al. Airway bacterial colonization and serum C-reactive protein are associated with chronic obstructive pulmonary disease exacerbation following bronchoscopic lung volume reduction. Clin Respir J 2016;10:239-45. [Crossref] [PubMed]
- Le Pimpec-Barthes F, Debrosse D, Cuenod CA, et al. Late contralateral lobectomy after single-lung transplantation for emphysema. Ann Thorac Surg 1996;61:231-4. [Crossref] [PubMed]
- Kuno R, Kanter KR, Torres WE, et al. Single lung transplantation followed by contralateral bullectomy for bullous emphysema. J Heart Lung Transplant 1996;15:389-94. [PubMed]
- Kroshus TJ, Bolman RM 3rd, Kshettry VR. Unilateral volume reduction after single-lung transplantation for emphysema. Ann Thorac Surg 1996;62:363-8. [Crossref] [PubMed]
- Anderson MB, Kriett JM, Kapelanski DP, et al. Volume reduction surgery in the native lung after single lung transplantation for emphysema. J Heart Lung Transplant 1997;16:752-7. [PubMed]
- Schulman LL, O'Hair DP, Cantu E, et al. Salvage by volume reduction of chronic allograft rejection in emphysema. J Heart Lung Transplant 1999;18:107-12. [Crossref] [PubMed]
- Fitton TP, Bethea BT, Borja MC, et al. Pulmonary resection following lung transplantation. Ann Thorac Surg 2003;76:1680-5; discussion 1685-6.
- Samano MN, Junqueira JJ, Teixeira RH, et al. Lung hyperinflation after single lung transplantation to treat emphysema. J Bras Pneumol 2010;36:265-9. [Crossref] [PubMed]
- Wilson H, Carby M, Beddow E. Lung volume reduction surgery for native lung hyperinflation following single-lung transplantation for emphysema: which patients? Eur J Cardiothorac Surg 2012;42:410-3. [Crossref] [PubMed]
- Arango E, Espinosa D, Illana J, et al. Lung volume reduction surgery after lung transplantation for emphysema-chronic obstructive pulmonary disease. Transplant Proc 2012;44:2115-7. [Crossref] [PubMed]
- Borro JM, Delgado M, Coll E, et al. Single-lung transplantation in emphysema: Retrospective study analyzing survival and waiting list mortality. World J Transplant 2016;6:347-55. [Crossref] [PubMed]
- Todd TR, Perron J, Winton TL, et al. Simultaneous single-lung transplantation and lung volume reduction. Ann Thorac Surg 1997;63:1468-70. [Crossref] [PubMed]
- Shen Y, Chen JY, Wei YC, et al. Nan Fang Yi Ke Da Xue Xue Bao 2007;27:895-6. [Single lung transplantation with concomitant contralateral lung volume reduction for end- stage emphysema]. [PubMed]
- Crespo MM, Johnson BA, McCurry KR, et al. Use of endobronchial valves for native lung hyperinflation associated with respiratory failure in a single-lung transplant recipient for emphysema. Chest 2007;131:214-6. [Crossref] [PubMed]
- Pato O, Rama P, Allegue M, et al. Bronchoscopic lung volume reduction in a single-lung transplant recipient with natal lung hyperinflation: a case report. Transplant Proc 2010;42:1979-81. [Crossref] [PubMed]
- Kemp SV, Carby M, Cetti EJ, et al. A potential role for endobronchial valves in patients with lung transplant. J Heart Lung Transplant 2010;29:1310-2. [Crossref] [PubMed]
- Perch M, Riise GC, Hogarth K, et al. Endoscopic treatment of native lung hyperinflation using endobronchial valves in single-lung transplant patients: a multinational experience. Clin Respir J 2015;9:104-10. [Crossref] [PubMed]


