Bronchoplasty for treating the whole lung atelectasis caused by endobronchial tuberculosis in main bronchus
Introduction
Endobronchial tuberculosis (EBTB) is an inflammatory disease caused by the invasion of Mycobacterium tuberculosis in bronchial mucosa or submucosa (1,2), which is more common in young people and female than in male (3-7), and in the left than in the right main bronchus (5,6). The bad compliances with anti-TB chemotherapy as well as the increased drug resistance (8-10) may lead to the bronchial stenosis or even occlusion formed by a long-term repeated unhealed scar, which may cause partial or whole lung atelectasis (4,11), and even destroy the lung in the end (12). Therefore, most of these patients need to be treated by surgery. Currently, patients with the whole lung atelectasis tend to undergo pneumonectomy (12,13); however, it may result in a large number of complications and poor quality of life (14).
Recently, we treated 8 young cases of the whole lung atelectasis caused by EBTB in main bronchus, who had accepted long-term anti-tuberculosis treatment but failed. Eventually, they were successfully cured by bronchoplasty instead of pneumonectomy. The purpose of this study is to report our experience in the surgical management of the disease and to discuss the clinical features, postoperative complications, and prognosis.
Methods
Clinical information
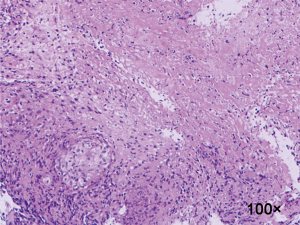
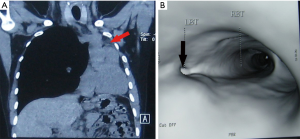
We retrospectively reviewed clinical materials of 8 patients with whole lung atelectasis caused by EBTB, who were diagnosed as the whole lung atelectasis and cured by surgery between Jun. 2006 to Dec. 2009. The associated pathology results showed a characteristic of granulomatous inflammation (Figure 1). The mean age of 2 male patients and 6 female patients was 24 years, and mean disease duration was 17 months. For the patients in this study, the early symptoms of the disease were cough and hemoptysis, and the late main symptoms were gasp and chest tightness. Pre-operative sputum smear and culture of M. tuberculosis showed negative results. The chest CT and bronchial three-dimensional reconstruction showed stenosis and occlusion in the main bronchus, the whole lung atelectasis and a shift of mediastinum to the affected side (Figure 2A,B). Of these 8 patients, 6 were in the left main bronchus, 2 in the right. Pre-operative fiberoptic bronchoscopy, by which 5 patients were pathologically diagnosed, revealed the narrow and occlusion of the proximal main bronchus and that the distal end was not seen. Additionally, 6 patients admitted with a long-term (>12 months) history of pulmonary tuberculosis.
The patients were intubated with a double-lumen endotracheal tube, turned to the contralateral decubitus position and underwent a standard posterolateral thoracotomy. Through intraoperative exploration, the left whole lungs of 3 cases were good, which can’t be resected, so bronchoplasty was performed in the left main bronchus. Because of tuberculosis lesion in the left upper lobe, 3 cases were admitted for sleeve resection of left upper lobe and reanastomosis of the left lower lobe to the left main bronchus, and due to fibrosis and consolidation in the right middle and upper lobes, 2 cases underwent sleeve lobectomy of right upper and middle lobes with anastomosis between the right lower lobe bronchus and the main bronchus. The bronchial anastomosis was then wrapped with a pedicle of pericardium. Flexibility and expansion of the remained lung were found to be good and there was no anastomotic leakage during the surgery. Then we closed the chest conventionally. At the end of surgery, the double-lumen tube was replaced by a single-lumen endotracheal tube, and the patient received short-term mechanical ventilation with positive end-expiratory pressure (PEEP). On the first day after surgery, weaning from ventilator and extubation were planned. They were advised to have follow-up examinations after discharge at regular intervals.
Results
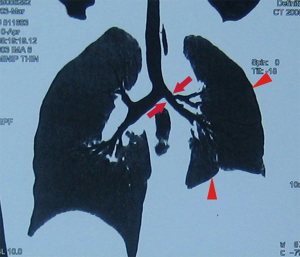
All patients had successful surgeries, and the mean inpatient stay was 14.3 days after the surgery. The pathological diagnoses of them were EBTB in main bronchus. They were re-examined on the 14th postoperative day by the chest CT and bronchial three-dimensional reconstruction image as well as bronchoscopy, which were all showing good patency of the bronchial anastomosis and good expansion of the remaining lung (Figure 3). Until the patients were discharged from hospital, they have not complained about chest tightness, fever, irritating cough, and sputum, and there was no bronchopleural fistula and lung atelectasis. Only 1 case had hoarseness, which occurred on the third day after surgery, and he was recovered after 2 months. And 2 cases had a small residual cavity in the right upper chest, which had not been larger within 11 months of follow-up. After surgery, these patients received anti-TB regimen (2 HRZSE/4 to 6 HRE) lasting for 6–12 months (sometimes, the consolidation period may be extended to 10 months), a standard medication plan for the patients treated with anti-tuberculosis therapy before, which is in favor of consolidating the surgical effects and preventing recurrence of EBTB. They were followed up for 11 to 48 months, respectively, and no recurrence was observed.
Discussion
Early imaging tests of EBTB, such as the chest radiography and the chest CT, maybe normal. However, in the later period, obstructive pneumonia, whole lung atelectasis and a shift of mediastinum to the affected side can be clearly seen in the images. For the identification of the location of lesion in main bronchus, pre-operative examination with the chest CT and bronchial three-dimensional reconstruction is necessary. And the bronchoscopy could not only show the endobronchial changes but also increase the positive rate of preoperative diagnosis by bronchoscopic biopsy, which can help to rule out malignancy and other inflammation and be unmatched by any other imaging examination (2,7,15,16).
Indications for operation
According to the characteristics of the patients in the study group, the indications for bronchoplasty are summarized as follows: (I) all the patients in this study suffered one or more symptoms, including cough, hemoptysis, gasp and chest tightness, which could not been well relieved through drug therapies; (II) bronchial stenosis, which is more than 2/3 of diameter, and even occlusion of the main bronchus as well as the failure of a long-term medical treatment, including dilatation or bronchial stenting; (III) tissue from the bilateral area of bronchial anastomosis must be healthy, which at least remains ≥0.5 cm distance from either side of the distal and proximal bronchus for anastomosis; (IV) at least the remaining one or more lobes of the total atelectasis lung should have good elasticity and no lesions; (V) TB is inactive or well controlled, and pre-operative anti-TB treatment has at least lasted for 3–6 months; (VI) patients are in good general condition, who can tolerate a lobectomy.
Experiences with the critical technique of operation
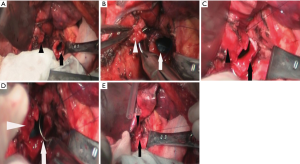
The difficulty and key points of the surgery vary depended on the locations of the EBTB in main bronchus, length of lesions, and situations of remaining lung of delivery. Our experiences are as follows: (I) the resectable range of lesions—we suggest that the stenosis lesion should be resected as completely as possible, and the surgical margin should be greater than 0.5 cm to avoid recurrence (Figure 4A). Besides, in order to get a negative margin result, intraoperative rapid pathological diagnosis was used. (II) The protection of bronchial blood supply—besides the requisite of completely resection, we should minimize the dissection of normal bronchial tissue surrounding bronchial anastomotic sites. And the appropriate dividing distance from the lesion is about 1 cm, which can remain surrounding blood supply as much as possible, help anastomosis to heal, and further prevent the occurrence of anastomotic leakage. (III) Avoiding injury to the recurrent laryngeal nerve—when we divided the proximal of left main bronchus, we should not do so far that directly causes injury to one side of the recurrent laryngeal nerve. Otherwise, patients may complain of irreversible hoarseness, coughing with weakness, which is harmful for expectoration and the expansion of the remaining lung and affect the prognosis. (IV) The treatment of the bronchial lumen—since there are often lots of secretion in distal bronchial lumen, after incision of the bronchus we should suck it out as soon as possible and then disinfect both ends of the lumen with 1% diluted povidone-iodine. In addition, double-lumen tube should be used to prevent intrusion of blood and secretion into the contralateral lung, which may threaten life (or which have death risk). (V) Anastomosis of the left main bronchus—since the right main bronchus is always exposed well, we could continuously suture with an absorbable line (3-0 Prolene suture). In comparison with that, the left main bronchus is often exposed poorly and is considered to be performed as the following steps: first, we anastomosed inner half of these two bronchi with one suture and tightened lines to involute the inner part (Figure 4B) and then anastomose the other half part of lateral bronchi (Figure 4C,D). In the end, the two ends of lines should be tightened and tied, respectively (Figure 4E). All the patients were anastomosed well at one time without leakage in the water during the surgery.
Complications and treatment
The early postoperative complications include atelectasis, pneumonia, respiratory failure and bronchopleural fistula. Sometimes these complications may have some relevance to each other and may appear in sequence. For instance, atelectasis may cause pneumonia, which finally leads to respiratory failure, and as a result, pulmonary infection is exacerbated. Furthermore, the infection of thoracic cavity is one of the high-risk factors of bronchopleural fistula. To avoid the early atelectasis, patients in the study group were ventilated mechanically with PEEP for an average of 22 hours, which can prevent the re-expanded alveoli from collapsing and reduce work of breathing. So, it benefits the renewal of postoperative pulmonary ventilation function. Additionally, we also use aerosol inhalation for dilution of sputum and relief of bronchial spasm, and susceptive antibiotics, postoperatively. If necessary, aspiration of sputum by fiberoptic bronchoscopy could be carried out. It is important to enhance the nursing work for helping to spit, such as forcefully slapping on the patients’ back and encouraging them to cough. Bronchopleural fistula always occurs within 7 days after bronchoplasty, especially in the patients with EBTB (17). In conclusion, besides good surgical techniques, early good expansion of the remaining lung and free drainage of pleural effusion and air in the chest cavity after surgery altogether contribute for the prevention of anastomotic leakage.
The late common complications are bronchial anastomotic stenosis and EBTB recurrence. The former is closely due to poor surgical techniques of anastomosis such as an obviously uniform diameter of both bronchial ends and high density of sutures. Additionally, anastomotic inflammation, which may be caused by the stitches and others stimuli, may lead to benign hyperplasia of granulation or scar, which is easy to be ignored. According to some reports, bronchoscopy can be considered to remove the granuloma and suture (18,19), and treat anastomotic stenosis with balloon dilatation or stent (20,21). However, all of these methods are not so effective. The latter is related to non-completely resection of lesions and postoperative irregular use of anti-TB drugs. The two late complications did not occur during the follow-up of our patients, and we believe that the critical beneficial factors are the improvement of surgical techniques and suture materials and the postoperative standard anti-TB treatment.
However, one patient in our group had hoarseness on the second postoperative day which lasted for 2 months. We presume that the recurrent laryngeal nerve might be oppressed by inflammatory edema or scar after surgery, and in that case, it might eventually recover. Otherwise, if there was a direct intraoperative injury to recurrent laryngeal nerve, such hoarseness would be not reversible (22).
Prognosis
Some patients suffering from the whole lung atelectasis caused by EBTB in main bronchus can be treated with bronchoplasty, by which the lesions are not only completely removed but also patients can avoid pneumonectomy. Compared with pneumonectomy, it may improve patients’ clinical symptoms and quality of life. Until now, no recurrence of these patients has been detected and the prognosis is good.
Conclusions
In summary, bronchoplasty is an effective treatment for atelectasis of the whole lung caused by EBTB in the main bronchus. It may improve patients’ clinical symptoms and quality of life. The favorable Prognosis is to be expected.
Acknowledgements
We are grateful to Leiming Wu for their secretarial assistance.
Funding: This work was supported by the project in science and education of Suzhou City (No. SYS201542) and National Natural Science Foundation of China (No. 81502275, 81702254).
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: Ethical approval was given by the Medical Ethics Committee of The First Affiliated Hospital of Soochow University. Consent to participate is not applicable.
References
- Lehtinen P, Kankanen L, Kulonen E. Properties of plasma membranes from granulation tissue with reference to extracellular matrix. Acta Chem Scand B 1975;29:963-8. [Crossref] [PubMed]
- Qian YF. Diagnostic value of bronchoscopy in endobronchial tuberculosis: a comparative study between bronchoscopic observations and pathological findings in 208 cases (author's transl). Zhonghua Jie He He Hu Xi Xi Ji Bing Za Zhi 1979;2:19-21. [PubMed]
- Qingliang X, Jianxin W. Investigation of endobronchial tuberculosis diagnoses in 22 cases. Eur J Med Res 2010;15:309-13. [Crossref] [PubMed]
- Sucena M, Amorim A, Machado A, et al. Rev Port Pneumol 2004;10:383-91. [Endobronchial tuberculosis -- clinical and bronchoscopic features]. [Crossref] [PubMed]
- Kikuchi K, Kobayashi K. Surgical treatment for tracheobronchial tuberculosis. Kekkaku 1997;72:43-8. [PubMed]
- Lee JY, Yi CA, Kim TS, et al. CT scan features as predictors of patient outcome after bronchial intervention in endobronchial TB. Chest 2010;138:380-5. [Crossref] [PubMed]
- Lee JH, Park SS, Lee DH, et al. Endobronchial tuberculosis. Clinical and bronchoscopic features in 121 cases. Chest 1992;102:990-4. [Crossref] [PubMed]
- Espinal MA, Laszlo A, Simonsen L, et al. Global trends in resistance to antituberculosis drugs. World Health Organization-International Union against Tuberculosis and Lung Disease Working Group on Anti-Tuberculosis Drug Resistance Surveillance. N Engl J Med 2001;344:1294-303. [Crossref] [PubMed]
- Aziz MA, Wright A. The World Health Organization/International Union Against Tuberculosis and Lung Disease Global Project on Surveillance for Anti-Tuberculosis Drug Resistance: a model for other infectious diseases. Clin Infect Dis 2005;41 Suppl 4:S258-62. [Crossref] [PubMed]
- Carvalho AC, Migliori GB, Cirillo DM. Tuberculosis in Europe: a problem of drug resistance or much more? Expert Rev Respir Med 2010;4:189-200. [Crossref] [PubMed]
- Park IW, Choi BW, Hue SH. Prospective study of corticosteroid as an adjunct in the treatment of endobronchial tuberculosis in adults. Respirology 1997;2:275-81. [Crossref] [PubMed]
- Hoheisel G, Chan BK, Chan CH, et al. Endobronchial tuberculosis: diagnostic features and therapeutic outcome. Respir Med 1994;88:593-7. [Crossref] [PubMed]
- Watanabe Y, Murakami S, Iwa T. Bronchial stricture due to endobronchial tuberculosis. Thorac Cardiovasc Surg 1988;36:27-32. [Crossref] [PubMed]
- Leo F, Scanagatta P, Vannucci F, et al. Impaired quality of life after pneumonectomy: who is at risk? J Thorac Cardiovasc Surg 2010;139:49-52. [Crossref] [PubMed]
- Chung HS, Lee JH. Bronchoscopic assessment of the evolution of endobronchial tuberculosis. Chest 2000;117:385-92. [Crossref] [PubMed]
- Cakir E, Uyan ZS, Oktem S, et al. Flexible bronchoscopy for diagnosis and follow up of childhood endobronchial tuberculosis. Pediatr Infect Dis J 2008;27:783-7. [Crossref] [PubMed]
- van der Werf TS, Mahama AR. Pulmonary tuberculosis complicated by bronchopleural fistula. Trop Doct 1989;19:31-3. [Crossref] [PubMed]
- Baba K, Nagao K, Matsuda M, et al. Kyobu Geka 1996;49:1048-51. [An operative case of suture-granuloma which resulted from an intra-pulmonary treatment 10 years ago and manifested hemoptysis]. [PubMed]
- Baumgartner WA, Mark JB. Bronchoscopic diagnosis and treatment of bronchial stump suture granulomas. J Thorac Cardiovasc Surg 1981;81:553-5. [PubMed]
- Chao YK, Liu YH, Hsieh MJ, et al. Controlling difficult airway by rigid bronchoscope--an old but effective method. Interact Cardiovasc Thorac Surg 2005;4:175-9. [Crossref] [PubMed]
- Phillips MJ. Stenting therapy for stenosing airway diseases. Respirology 1998;3:215-9. [Crossref] [PubMed]
- Chiang FY, Lu IC, Kuo WR, et al. The mechanism of recurrent laryngeal nerve injury during thyroid surgery--the application of intraoperative neuromonitoring. Surgery 2008;143:743-9. [Crossref] [PubMed]


