European institutional accreditation of general thoracic surgery
Introduction
General thoracic surgery (GTS) must be performed by qualified surgeons, specialized in GTS according to European or national regulations, and practicing in dedicated GTS units with appropriate characteristics.
Unfortunately, in Europe the educational and qualification pathways of GTS vary from country to country.
To obviate this situation and improve standardization of GTS practice across Europe, the European Society of Thoracic Surgeons (ESTS) has implemented a program of Institutional Accreditation.
The aim of this program is to set standards of good clinical practice across Europe with the intent to improve the quality of care according to published guidelines.
European institutional accreditation: rules of engagement
To be eligible for accreditation, the units must be active contributors to the ESTS Database. A contribution of high quality data of at least 2 years and a sizable number of patients is required. This pre-requisite is necessary to reliably calculate the composite performance score (CPS), which is the metric used to evaluate the Institutional performance.
In addition to Database participation and a positive CPS (score >0), units must meet certain structural, procedural and qualification criteria based and modified from those published more than 10 years ago in a joint EACTS/ESTS document (1) and recently updated and revised by Brunelli and coll on behalf of the ESTS and EACTS thoracic domain (2).
The CPS
It is now recognized that clinical outcomes are only one measure of overall health care quality (3).
In fact, like intelligence or musical ability, quality is an abstract construct that is characterized by many latent variables. To quantify abstract constructs a combination of measurable surrogates, which are associated with that underlying variables is needed.
A recent document from the Society of Thoracic Surgeons (STS) quality measurement task force elegantly explained the conceptual and the statistical considerations in the development of CPSs in Cardiac Surgery (4).
Based on that methodology, ESTS has developed a CPS for lung surgery (5).
The method consists in developing standardized outcome and process indicators covering all temporal domains of the lung resection care. The indicators were selected based on their evidence-based level and possibly according to published guidelines.
For the preoperative domain, we selected the proportion of patients with DLCO measured before major lung resection (6), and the proportion of patients with clinically suspicious N2 nodes at CT scan or PET scan submitted to some types of preoperative mediastinal invasive staging (7).
For the intraoperative domain, we selected the proportion of patients with primary neoplastic disease submitted to major anatomic resections and lobe-specific or systematic nodal dissection (8).
For the postoperative domain, we selected the risk-adjusted in-hospital cardiopulmonary and mortality rates (9).
Each of these indicators has been rescaled according to their standard deviation in the entire population to obtain individual standardized indicators. These were then summed to obtain the composite score for each unit.
To derive the regression models for morbidity and mortality, univariate screening of the following variables: age, gender, BMI, type of resection, ppoFEV1, induction therapy, extended resection, presence of cardiac co-morbidity. Variables with P-level<0.1 were used as independent predictors in backward logistic regression analysis validated by bootstrap resampling technique. Only significant (P<0.05) and reliable (bootstrap significancy frequency >50%) were retained in the final model.
The logistic regression equation for mortality (c-index 0.74; Hosmer Lemeshow statistics, P=0.9)
Logit: –3.22 + 1.049 × pneumonectomy (coded as 1 vs. 0 lobectomy) + 0 .928 × cardiac comorbidity (coded as 1 and including CAD, any previous cardiac surgery, history and treatment for arrhythmia, congestive heart failure, hypertension) – 0.0175 × ppoFEV1%.
The logistic regression equation for cardiopulmonary morbidity (c-index 0.66; Hosmer-Lemeshow statistics, P=0.4)
Logit: –3.52 + 0.659 × pneumonectomy + 0.403 × extended resection (coded as 1 and including chest wall resection, pleuropneumonectomy, completion operation, intrapericardial operation) + 0.322 × cardiac comorbidity – 0.0065 × ppoFEV1% + 0.0315 × age.
Standardized scores are then calculated by subtracting the observed risk-adjusted outcome or process incidence minus the average observed outcome or process incidence. The difference is then divided by the standard deviation of the observed outcome or process in the entire population. A CPS >0 is the threshold selected by the Database Committee as the eligibility criteria for accreditation.
Structure and qualification of GTS unit
In addition to their CPS, units must have certain structural, procedural and professional characteristics to be certified. These characteristics needs to be assessed and audited along a sample of data submitted to the database. To this purpose, ESTS has subcontracted an external auditing company, which together with a thoracic surgeon is appointed to visit the applicant units and produce a report, which is then evaluated by the Database Committee. If the report is judged satisfactory, the Accreditation is finally granted by the ESTS Council.
The followings are the required structural/procedural/professional characteristics (2):
Institutional status
GTS units should have dedicated staff and institutional resources and ideally a separate budget whenever feasible.
They should have a dedicated and separated personnel and institutional resources and should be headed by an UEMS EBTS certified surgeon or by a surgeon with an equivalent certification issued by a UEMS recognized body (i.e., national diploma of specialization) with a minimum experience of 5 years of practice as qualified GTS surgeon.
Qualification of surgeons
GTS units should have a dedicated staff including at least one UEMS EBTS certified (or with an equivalent certification recognized by UEMS—i.e., National Diploma of Specialization) surgeon supervising surgical activity and acting as Head of the unit plus a number of qualified (preferably UEMS EBTS certified or with a UEMS recognized certification of specialization—i.e., National Certificate of Specialization) general thoracic surgeons performing at least 100 certifiable major thoracic procedures per year per surgeon. Ideally, there should be one staff qualified GTS surgeon each 100 major thoracic procedures. A minimum staff of two qualified GTS surgeons should be in place to allow for adequate coverage of patient care and to ensure adequate on-call arrangements.
Operating rooms
There should be one dedicated operating room per 300-400 major thoracic procedures per year.
Advanced care
GTS units should preferably have access to a dedicated thoracic Intensive Care Unit (ICU) or as an alternative to a multispecialty ICU with expertise to treat thoracic patients.
In addition, one Intermediate Care or High Dependency Unit bed per 100 major thoracic procedures should be available.
Thoracic ward
GTS units should have a dedicated thoracic surgical ward with dedicated paramedical staff and physiotherapists. Ideally, a GTS ward should have 4-6 beds available per 100 major thoracic procedures per year.
Outpatient care
GTS units should have sufficient facilities for outpatient visits allowing same day access to radiology, pulmonary function tests, endoscopy and cardiologic testing if needed.
Inpatient diagnostic facilities
GTS units must have access to the following support facilities:
- Haematological, biochemical and microbiological laboratories;
- Respiratory pathophysiology laboratory;
- Endoscopic examinations by bronchoscopy and oesophagoscopy (including EBUS and EUS);
- Radiological investigation by plain X-ray, contrast studies, ultrasound, vascular imaging, CT-scan, PET scan;
- CT or ultrasound needle biopsy;
- Cytology, histopathology and frozen section analysis.
Surgical activities
GTS units should perform a total number of major thoracic procedures greater than 150 (+/–50). The term major thoracic procedures, which is used in this paragraph refers to the generality of surgical procedures.
The process of accreditation
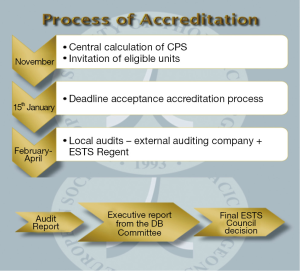
The process of accreditation is shown in Figure 1. CPS is automatically calculated centrally at the end of each year. Eligible units are invited to apply for the program. If they accept to participate they have to submit an application form and an application fee covering the expenses of the audit team visiting the unit and the audit process, which is performed by an external company subcontracted by ESTS.
The auditing team is composed by representatives from the auditing company and a thoracic surgeon selected by the Database Committee, who is generally the ESTS national regent of the unit Country.
The auditing team would inspect the unit during February-April. The audit focuses both on a random sample of data submitted to the ESTS Database to verify their quality and correspondence with source data file and on the structural/procedural and qualification characteristics of the units mentioned above.
Once the visit is complete, the team will produce an audit report to be sent to the members of the database committee for deliberation on the institutional accreditation of that unit.
The Database committee will send an executive report to the ESTS Executive Committee for their final decision on the accreditation.
The accreditation is valid for a 36 months period. After this period the unit must apply for revalidation. The first revalidation will consist in assessing the CPS only. No additional local audit is required. From the second revalidation the unit must reapply for repeating the entire process including local inspection if CPS would result positive.
Benefits of the institutional accreditation
The ESTS accreditation provides a number of benefits to certified institutions:
- Accredited units will be announced during the ESTS Annual meeting and their names listed in the ESTS website and in the ESTS Annual Report;
- Participation to ESTS quality improvement initiatives;
- Participation to high-profile scientific projects supported by the ESTS scientific committee;
- Accredited units may propose their own clinical research projects based on the data present in the ESTS database.
Acknowledgements
Disclosure: The authors declare no conflict of interest.
References
- Klepetko W, Aberg TH, Lerut AE, et al. Structure of general thoracic surgery in Europe. Eur J Cardiothorac Surg 2001;20:663-8. [PubMed]
- Brunelli A, Falcoz PE, D’Amico T, et al. European guidelines on structure and qualification of general thoracic surgery. Eur J Cardiothorac Surg 2014;45:779-86. [PubMed]
- Brunelli A, Rocco G. Clinical and nonclinical indicators of performance in thoracic surgery. Thorac Surg Clin 2007;17:369-77. [PubMed]
- Shahian DM, Edwards FH, Ferraris VA, et al. Quality measurement in adult cardiac surgery: part 1--Conceptual framework and measure selection. Ann Thorac Surg 2007;83:S3-12. [PubMed]
- Brunelli A, Berrisford RG, Rocco G, et al. The European Thoracic Database project: composite performance score to measure quality of care after major lung resection. Eur J Cardiothorac Surg 2009;35:769-74. [PubMed]
- Brunelli A, Charloux A, Bolliger CT, et al. The European Respiratory Society and European Society of Thoracic Surgeons clinical guidelines for evaluating fitness for radical treatment (surgery and chemoradiotherapy) in patients with lung cancer. Eur J Cardiothorac Surg 2009;36:181-4. [PubMed]
- De Leyn P, Lardinois D, Van Schil PE, et al. ESTS guidelines for preoperative lymph node staging for non-small cell lung cancer. Eur J Cardiothorac Surg 2007;32:1-8. [PubMed]
- Lardinois D, De Leyn P, Van Schil P, et al. ESTS guidelines for intraoperative lymph node staging in non-small cell lung cancer. Eur J Cardiothorac Surg 2006;30:787-92. [PubMed]
- Brunelli A, Rocco G, Van Raemdonck D, et al. Lessons learned from the European thoracic surgery database: the Composite Performance Score. Eur J Surg Oncol 2010;36 Suppl 1:S93-9. [PubMed]