
Evaluation of high-risk pulmonary nodules and pathologic correlation in patients enrolled in a low-dose computed tomography (LDCT) program
Introduction
Lung cancer is one of the leading causes of death in both men and women within the United States. The American Cancer Society estimates that lung cancer is responsible for 155,870 deaths: 84,590 in men and 71,280 in women. Additionally, research shows that each year, more people die of this particular type of cancer than of breast, prostate, and colon cancer combined (1). The number of lung cancer cases continues to rise annually while the rates of survival remain relatively low (2). In the United States, the 5-year survival rate for lung cancer is currently 18% (3).
In an effort to reduce lung cancer mortality statistics, many major medical societies have recommended annual low-dose CT screenings for high-risk individuals. The Centers for Disease Control and Prevention (CDC) has recommended yearly lung cancer screening with LDCT for people who are classified as high-risk individuals. High-risk individuals, according to the CDC are those who are between ages 55 to 80 years who have a 30 pack-year smoking history and currently smoke or have quit within the past 15 years (4). Additionally, the U.S Preventive Services Task Force has given a grade B recommendation for low-dose screenings (5)—demanding private insurance companies to cover all low-dose screening costs.
Although controversy continues to exist regarding the effectiveness of low-dose CT screenings, recent research conducted by the National Lung Cancer Screening Trial (NLST) showed a 20% decrease in lung-cancer mortality with the use of annual low-dose CT screenings (6). Other studies have focused on the effectiveness of low-dose programs in detecting asymptomatic malignancies. In addition to focusing on the effectiveness of our program, this study focused on studying the evolution of nodules, and the pathological nature of the malignancies found.
Methods
In February 2014, our institution implemented a low-dose program. Our team received ethics approval through Englewood Health’s IRB (IRB#-19-762) for a retrospective analysis of the program outcomes which did not require informed consent. We analyzed patients seen between the dates of February 21, 2014 and June 1, 2017. Our studied population consisted of 639 total patients. We established a prospective database examining multiple parameters such as evolution of change in lung-RADS classification, histology of tumors, follow up rates, and overall success of the program.
Three scanners were used to perform the low-dose CT screenings. The scanners included the GE Lightspeed VCT, Toshiba Aquilion, and the Toshiba Prime. All of the scanners capture up to 64 simultaneous anatomical slices in a single gantry rotation. These scanners were chosen for use in the program because they are able to enhance patient safety utilizing low-dose radiation while providing high quality CT images.
Patients within our program were screened prior to their first low-dose scan to make sure they met all eligibility criteria. The eligibility criteria used at our institution mirrored the patient eligibility criteria posted on the American College of Radiology’s Lung Cancer Screening website (7). In order for an individual to be evaluated within our low-dose CT screening program, the individual must be between the ages of fifty-five and seventy-seven, asymptomatic, have a thirty-pack years or greater history of smoking, and a current smoker or have quit within the last 15 years. The patients were then assessed in a standardized fashion for radiographic risk of nodules according to the ACR Guidelines (7). As depicted in the Lung-RADS Version 1.0 Assessment Categories released by the ACR (7), the guidelines categorize patients based on the presence of nodules, the size, and the nature of the nodule.
Results
Six hundred and thirty-nine total patients were evaluated over the course of the study and there were 759 total scans conducted. As a result of the continued success of the program, we saw the highest number of patients in 2016. From February 2014 to June 2017, the total number of patients screened increased by 32.5%. As a major goal was to identify frequency of pulmonary nodules, we noted that 43.9% (333/759) of scans showed the presence of pulmonary nodules.
These nodules were found in 207 out of 639 patients (32.4%). Using the guidelines given by the American College of Radiology, we classified 36 out of 639 patients as high-risk individuals [4A, B, X (5.63%)]. The classification of nodules noted is tabulated in Table 1, which shows that only 5.63% of the total population were classified as high-risk. Table 1 also shows that a majority of the studied scans showed results that categorized the patient as either lung-RADS 1 or lung-RADS 2 (low risk), totaling 87.1% of the total scans.

Full table
We next studied the evolution of nodules over the time period of the study with respect to lung-RADS. The most common change seen was migrating from lung-RADS 1 to lung-RADS 2. Eleven patients migrated from lung-RADS 1 to 3 and ten patients migrated from 2 to 3. Table 2 shows that only two (0.3%) patients out of the total population migrated from a low-risk. Lung-RADS classification (lung-RADS 1-3) to a high-risk lung-RADS classification (lung-RADS 4A, B, X). This table also shows that 8% of the total population changed in lung-RADS over the course of the study period.
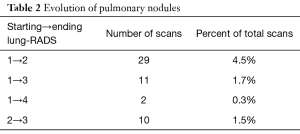
Full table
Table 3 demonstrates the histopathological characteristics of the tumors found. This includes the type of cancer as well as staging. Within the study period, twenty-five high-risk patients were recommended a biopsy based on their low-dose screening results. Seventeen of the twenty-five (68%) were benign while eight were found to be malignant. These malignancies included one squamous cell carcinoma, five adenocarcinomas, one non-small cell carcinomas, and one lymphoma.
The youngest patient with a malignancy was 64 years of age while the oldest patient was 77 years of age. Additionally, six of the eight malignancies were found in males. The malignancies were found in different locations: two were found in the right lower lobe, two in the right upper lobe, one in the left lower lobe, and three were multiple sites. Case IV showed metastatic cancer originating in the lung and migrating to the lymph nodes (Table 3). Case VII showed metastatic malignancy originating in the lung and migrating to the liver. Case VIII showed metastatic lymphoma migrating to the bone marrow and lungs.
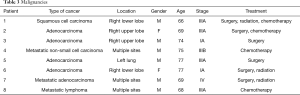
Full table
With respect to pathologic staging, 3 out of 8 were noted to be stage III (37.5%). There were also two primary adenocarcinomas that were found to be stage I (25%). 3 of the 8 malignancies (37.5%) were found to be non-lung primary and resulted in treatments such as chemotherapy and radiation.
We found the prevalence of malignancy to be 1.09% in our studied population. The sensitivity of the low-dose program was found to be 100% while the specificity was found to be 96.4%. Our sensitivity demonstrates that we have correctly identified all patients with the disease. On the other hand, our specificity demonstrates that we have correctly reported 96.4% of patients without the disease as a true negative.
Discussion
By examining the number of patients screened per year, we were able to determine that the total number of patients getting screened from 2014 to 2016 increased by 32.5%. A potential cause of this increase in screenings could be the increase in physician awareness throughout the institution. By the end of 2016, there was an increase in the number of physicians referring their patients to receive low-dose screenings. This directly influenced the number of new patients entering the program, with an estimate of a 36.6% increase expected in 2017.
One of the ways to determine whether a nodule was suspicious was conducting multiple scans and observing the evolution of the nodule throughout the scans (8,9). Although the overall percentage of pulmonary nodules were 32.4% of scans, we found only eight to be malignant.
This suggests that the rate of benign nodule detection is relatively common even in a high-risk population. Although our rate of benign nodule determination was high, based on the lung-RADS classification and observation over time, we only needed to perform twenty-five biopsies, resulting in seventeen being benign. Eleven of the twenty-five biopsies were surgical, six were bronchoscopies, and eight were CT-guided, needle, or aspiration.
A study conducted by the Division of Pulmonary and Critical Care Medicine and Internal Medicine at the Mayo Clinic found 2,244 noncalcified lung nodules in 66% of their 1,520 screened participants (10). Compared to our institution which found 33.2% of scans to include at least one nodule, these findings further suggest that benign nodule detection is relatively common.
One of the goals of this paper was to evaluate the characteristics of pulmonary nodules and their pathologic findings in a low-dose CT screening program. The specific goals of our research were to study the potential evolution of nodules for increasing risk, histopathology of malignancies, number and type of patients, stage of identified cancers.
With respect to the histopathology of tumors found, we were interested in looking at the type, stage, and location of the malignancies. With respect to location, we could not define any particular pattern. In contrast, when we examined the histology, we found that five of the eight malignancies were adenocarcinoma (62.5%). This is similar to another study conducted by the National Lung Screening Trial which revealed that adenocarcinomas were the most frequently occurring when studying malignancies of a high-risk population (11).
According to a study conducted by the National Cancer Institute (NCI), one in 100 high-risk persons enrolled in a study were found to have lung cancer on their first screening exam (11). These findings suggest that we should have found approximately six cancers. Among the individuals involved in our low-dose program, eight malignancies were demonstrated. This is consistent with the NCI prediction.
An additional study, “Cost-Effectiveness of CT Screening in the National Lung Screening Trial”, was conducted that analyzed the histological nature of malignancies within a population (12). These findings showed, once again, that adenocarcinoma was the most common type of malignancy found within the population, which is consistent with our findings. This study also analyzed the role of subsequent scans in terms of cost/benefit. The study found that the cost-effectiveness ratio of a single baseline low-dose CT scan was 2,500 US dollars per year of life saved (12). The article also showed that the screening program would be expected to increase survival by 0.1 year at an incremental cost of 230 US dollars (12).
With respect to stage, we found that five of the eight (62.5%) were not early stage, but instead were found to be stage III or IV. In contrast, an additional study demonstrated somewhat different results. This study found twenty-eight of thirty-six (77.7%) malignancies to be early stage IA and the rest to be either stage II or III (13). However, our program detected more malignancies in the later stage. These included two patients within stage I, three patients within stage III, and two within stage IV. This is potentially due to the fact that our institution had a lower volume of patients compared to larger institutions.
Only two patients significantly increased from low lung-RADS classification to a higher classification which demonstrates that it is possible to change from a low lung-RADS classification to a higher classification within a 3-year period (Table 2). Our study as well as others have demonstrated the importance of following patients over time. Although there was migration between lung-RADS categories among the high-risk population (4A, B, X), only 8% migrated from a low-risk category to a higher-risk category. This demonstrates not only the strength of the low-dose program, but also the need to continue the annual follow-ups of high-risk individuals.
In addition to demonstrating the merits of the low-dose CT screening program, our study has demonstrated, amongst the large number of patients, very low rates of migration from low lung-RADS classification to a higher risk category, the finding of more advanced stages of malignancies, and a preponderance of adenocarcinoma within the eight malignancies found.
Acknowledgements
We gratefully thank Dr. Steven Brower, Medical Director of the Lefcourt Family Cancer Treatment and Wellness Center, for his assistance and guidance throughout the entire study.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: Our team received ethics approval through Englewood Health’s IRB (IRB#-19-762) for a retrospective analysis of the program outcomes which did not require informed consent.
References
- American Cancer Society. Key Statistics for Lung Cancer. Retrieved January 11, 2018, Available online: https://www.cancer.org/cancer/non-small-cell-lung-cancer/about/key-statistics.html
- Torre LA, Bray F, Siegel RL, et al. Global cancer statistics, 2012. CA Cancer J Clin 2015;65:87-108. [Crossref] [PubMed]
- World Health Organization-WHO. Cancer. 2011. Available online: (accessed 12 December 2017).http://www.who.int/mediacentre/factsheets/fs297/en/
- Center for Disease Control. Lung Cancer. (2017, July 26). Retrieved January 11, 2018, Available online: https://www.cdc.gov/cancer/lung/basic_info/screening.htm
- US Preventative Services Task Force. Lung Cancer. (2017, July 26). Retrieved December 9, 2017, Available online: https://www.uspreventiveservicestaskforce.org/Page/Document/UpdateSummaryFinal/lung-cancer-screeningrom
- The Patient Protection and Affordable Care Act, HR 3590, 111th Congress, 2nd Session (2010), §6301. Available online: https://www.congress.gov/bill/111th-congress/house-bill/3590?q=%7B%22search%22%3A%5B%22affordable+care+act%22%5D%7D&s=3&r=1
- American College of Radiology. Lung Rads. Retrieved December 8, 2017 Available online: http://www.acr.org/Quality-Safety/Resources/LungRADS
- Jemal A, Siegel R, Xu J, et al. Cancer statistics, 2010. CA Cancer J Clin 2010;60:277-300. [Crossref] [PubMed]
- Marchianò A, Calabrò E, Civelli E, et al. Pulmonary nodules: volume repeatability atmultidetector CT lung cancer screening. Radiology 2009;251:919-25. [Crossref] [PubMed]
- Bach PB, Mirkin JN, Oliver TK, et al. Benefits and harms of CT screening for lung cancer: a systematic review. JAMA 2012;307:2418-29. [Crossref] [PubMed]
- National Lung Screening Trial Research Team. Reduced lung-cancer mortality with low-dose computed tomographic screening. N Engl J Med 2011;365:395-409. [Crossref] [PubMed]
- Black WC, Keeler EB, Soneji SS. Cost-effectiveness of CT screening in the National Lung Screening Trial. N Engl J Med 2015;372:388. [PubMed]
- Nawa T, Nakagawa T, Kusano S, et al. Lung cancer screening using low-dose spiral CT: results of baseline and 1-year follow-up studies. Chest 2002;122:15-20. [Crossref] [PubMed]


