
Anatomical pulmonary resections for primary lung cancer in octogenarians within a dedicated care protocol
Introduction
With the growing number of octogenarians in Western countries and the increase in cancers diagnosed at an advanced age, the question of proper treatment of this population is of gaining interest.
We focus on the treatment of non-small cell lung cancer (NSCLC) in octogenarians. Analyzing the literature, we noticed that older patients are less frequently operated on than younger one, for early-stage NSCLC (1-4).
Oncologists and surgeons are concerned with operative morbidity and mortality and the impact on the quality of life after surgery. Even without objective contra-indications, octogenarians are more frequently oriented towards alternative treatments such as SBRT (stereotactic body radiation therapy) or exclusive radio-chemotherapy (4). The risk is to offer these patients a suboptimal therapy based on the belief that they tolerate surgery poorly because of their age.
This study aims to establish if surgeons and oncologists are right to be afraid of performing major pulmonary resection for early stage NSCLC in octogenarians. To do so, we describe the results of post-operative morbidity, mortality, and survival of rigorously selected patients operated on.
Methods
Study population
We retrospectively collected data of 55 patients hospitalized consecutively in a single center, between January 2005 and December 2016. Inclusion criteria were: age greater than 80 years at the time of surgery, no contra-indication for surgery, and availability of geriatric assessment (GA). The lesion operated on had to be non-small cell primary lung cancer. We included only resectable lesions, without induction treatment, following to the current recommendation (5). All patients had an anatomical resection (pneumonectomy, lobectomy or segmentectomy) plus radical lymphadenectomy (stations 2, 4, 7, 8, 9 on right side, stations 5, 6, 7, 8, 9 on left side).
Patients undergoing wedge resection were excluded from the study.
Staging of the lesion
We performed standard clinical examinations, laboratory tests, computed-tomography (CT) scans of the brain, chest and upper abdomen, fiber optic endoscopies and systematic PET TDMs to define the cTNM status based on the seventh edition of the UICC (6).
Pre-operative evaluation
GA was systematically performed in a dedicated consultation (7), and patients were classified according to Balducci criteria (8), and Charlson comorbidity index (9). Dedicated team of oncogeriatricians was assessing general function of the patient, within a formalized consultation, of a minimum of one hour. They were taking into account: past medical history, former medication, complementary exam, but also: functional ability, cognition and mental health, socioenvironmental organization, balance, nutrition, earing and vision. The aim is to separate robust patients from frail and occult frail patients to adapt the treatment and follow-up plans to each patient and its particular limitations.
Patients’ ECOG Performance Status was systematically defined (10). Pulmonary function was preoperatively assessed by spirometry, and arterial blood gas measurements.
Patients underwent cardiac evaluation with systematic ECG and trans-thoracic echocardiography, and complementary evaluation at the discretion of the cardiologist.
We conformed strictly to the recommendation of operability published by ACCP (11).
Each decision of treatment was made within a multidisciplinary team, composed of oncologists, surgeons, radiologists, radiotherapists and oncogeriatricians.
Peri-operative management of the patients
Each patient was included in a specific protocol of care based on GA. There was no prescription of pre-operative benzodiazepine. Peridural analgesia was systematically performed, to limit the utilisation of morphine (IV or PO). Tramadol was never used in the post-operative period. Patients were mobilised at day one, with daily physiotherapy. Chest tubes were removed on day 2 and day 4. They were all discharged towards a specific recovery center for 3 weeks of a rehabilitation program.
Data collection
Demographic data and characteristics of the tumors were collected from medical charts. As it was a retrospective analysis, with no modification of our current practice, this study was not registered as a clinical trial, and there was no need of ethics approval. All patients had signed an informed consent form before surgery.
Data included were: patient age at diagnosis, surgical procedure performed, location of the cancer, clinical stage, pathological stage, histological subtype, type of resection, duration of hospital stay, type of complications, severity of complications according to Clavien (12), date of the first recurrence of the cancer based on date of the first CT-scan describing the lesion, site of recurrence based on this Ct-scan, date of the most recent follow-up or death.
The diagnosis of pneumopathy was based on the presence of a pulmonary infiltrate on chest X-ray and one or more of the following criteria: hyperthermia >38.4 °C, leucocythemia >10,000/mm3 or <4,000/mm3; or two or more of the following criteria: cough, purulent tracheobronchial secretions, or requirement of oxygenotherapy or ventilation assistance. Acute respiratory distress syndrome was defined by application of Berlin definition (13)
Patients were followed for 5 years after the surgery. A CT-scan was performed every six months for the first two years and annually for the 3 following years.
Statistical analysis
Continuous variables were described using means or medians and ranges, and categorical variables using percentages. Overall survival (OS) corresponded to the length of time between surgery and death, loss of follow-up or time to evaluation. Recurrence-free survival (RFS) corresponded to the length of time between surgery and the first recurrence or loss of follow-up, at which time data were censored. They were calculated using the Kaplan-Meier method.
The statistical analysis was performed with R software (http://www.r-project.org).
Results
Patients and tumor characteristics
A total of 55 patients met the inclusion criteria. Patients and tumor characteristics are given in Table 1. Mean age was 82.1 years (80–86 years). Mean FEV1 was 94% and mean FVC was 98%. ECOG PS was 0 for all patients. Median Charlson’s comorbidity score was 0 (0–3). All patients were classified Balducci 1 or 2. No patient had adjuvant chemotherapy. Clinical staging of the lesion was cT1N0 in 16 cases (30%), cT1N1 in 4 (7%), cT2N0 in 20 (36%), cT2N1 in 4 (7%), cT3N0 in 7(13%), cT3N1 in 1 (2%) and cT4N0 in 3 (5%). Eleven patients had an up-gradation of T status (20%). Five patients had an up-gradation from cT1 to pT2 (9%), 1 from cT1 to pT3 (2%), 4 from cT2 to pT3 (7%) and one from cT3 to pT4 (2%). Twelve patients had an up-gradation of the tumor N stage after surgery (21%). Six patients had an up-gradation from cN0 to pN1 (11%). Three patients had an up-gradation from cN0 to pN2 (5%) and 3 from cN1 to cN2 (5%).
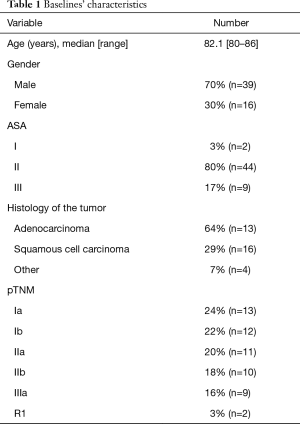
Full table
Feasibility of surgery in octogenarians
Details of type of surgical resection are shown in Table 2. Forty-eight interventions were made by conventional open surgery and seven by video-assisted thoracic surgery (VATS) (one right lower lobectomy, one apicodorsal segmentectomy, two right upper lobectomies, and three left lower lobectomies).
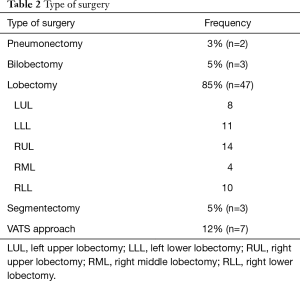
Full table
The median hospital stay was 11.5 days (7–31 days). The 30-day mortality rate was 3% (two deaths at days 15 and 29 after right upper lobectomy and right lower lobectomy respectively). The in-hospital morbidity rate was 45% with 11% of confusional syndrome (n=6). There were 29% of Clavien 2 complications (n=16), 11% Clavien 3 (n=6), 2% Clavien 4 (n=1) and 4% Clavien 5 (n=2). The total of relevant clinical complications (Clavien 3 to 5) was 16% (n=9). Details of complications are shown in Table 3.
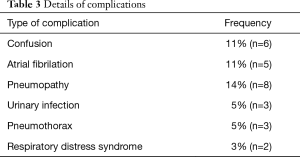
Full table
Survival analysis
The mean follow-up was 39 months (28–140 months). Seventeen patients died during the follow-up. Among them six died within the first year (2 post-operative death at day 15 and 29, one cardiogenic death, one digestive hemorrhage, two early recurrence of the disease). On the following year, 10 died because of NSCLC recurrence and one from other cancer.
The OS at one, three and five years were, respectively: 83% (95% CI: 75–95%); 70% (95% CI: 56–87%); 58% (95% CI: 43–79%) (Figure 1). The median RFS was 5.46 years (95% CI: 1.9–not reached). RFS at one, three and five years were respectively: 73% (95% CI: 61–89%); 62% (95% CI: 48–81%); 56% (95% CI: 40–79%) (Figure 2).
Discussion
Life expectancy for octogenarians is constantly rising. In France, life expectancy at 80 years old is 8.4 years for men and 10.5 years for women (14). In the United States, octogenarians account for 14% of all lung cancers (15,16).
What to do for octogenarians with resectable NSCLC is a frequent question in oncological daily practice. Because of their supposed or real frailty, those patients are currently undertreated (1-4), and advanced age per se is a poor prognostic factor, even in patients with stage I NSCLC in large cohort studies (17). Some papers have proven that patients older than 70 have comparable surgical issues to younger ones (1,18-20), but for older one, literature is less provided. Surgeons and oncologists remain preoccupied by the consequences of major pulmonary resection in octogenarians.
This study aimed to determine results of major pulmonary resection for primary lung cancer in rigorously selected octogenarians in a dedicated care protocol. With a morbidity of 45% and a mortality of 3% and a 5-year OS rate of 58%, our series is in the best range of the results of the literature. In a review of the literature on lung surgery in the elderly, Pallis et al. (21) reported a morbidity of 10% to 60%, a mortality of 0 to 9% and a 5-year OS rate of 32% to 57%.
Our rate of 11% for post-operative delirium (POD) is inferior to the one in literature, even if all patients spent 24 to 48 h in ICU after surgery. Pisani et al. (22) described an 80% rate of POD in the case of hospitalization in ICU for elderly patients. Our low rate of POD may be related to the realization of a systematic geriatric assessment. The consultation aimed to distinguish robust patients from frail and occult frail patients. Frail and occult-frail patients were oriented toward alternative treatment such as SBRT, and only robust patients were operated on. We also applied a rigorous policy of restriction of the use of benzodiazepine and morphinics in octogenarians. Early mobilization and physiotherapy were performed as early as post-operative day 1.
The majority of the surgical procedures were performed through a conventional approach. We did not use video-assisted thoracic surgery techniques at the beginning of this series, because it was the beginning of our learning curve of the technique. We were waiting to have similar duration of intervention before implementing the technic to elderly. As published by Port et al. (23), the implementation of the use of video-assisted thoracic surgery techniques will help us to improve morbidity, by lowering the rate of pneumonia and atelectasis.
All octogenarian patients spent 3 weeks in a recovery center. Our length of stay of eleven days, with only 16% of relevant complications, is explained by the delay between theoretic discharge possibilities and availability of places in the recovery center.
Our study has some limitations. First, its retrospective nature induces some selection bias. Patients were included only after extensive pre-operative evaluation. We do not have the data for octogenarians treated by other options during the same period as we are taking care of patients coming from various oncology units. We performed extremely rigorous pre-operative work-up, with systematic geriatric assessment. Surgery was performed only in case where octogenarians were having normal lung and cardiac function. We also report a single center experience, with therapeutic decisions made in the same pluri-disciplinary meeting, with the same team and with the same “sensibility and subjectivity” of physicians. However, that is also the strength of this study: in a dedicated care protocol, with rigorous selection of patients, based on the stage of the disease, but also on an extensive and objective evaluation of the patient’s clinical state, by a systematic geriatric assessment, surgery is a valuable option to be considered seriously.
Major pulmonary resection for primary lung cancer in octogenarians seems to be safe, with an acceptable morbidity, mortality and long-term survival rate when performed in a dedicated care protocol. Age per se is not a contra-indication to major lung surgery with curative intent.
Acknowledgments
We want to thank the Geriatric Department of the University hospital of Nice for its collaboration.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. As it was a retrospective analysis, with no modification of our current practice, this study was not registered as a clinical trial, and there was no need of ethics approval. All patients had signed an informed consent form before surgery.
References
- Dillman RO, Zusman DR, McClure SE. Surgical resection and long-term survival for octogenarians who undergo surgery for non-small-cell lung cancer. Clin Lung Cancer 2009;10:130-4. [Crossref] [PubMed]
- Brock MV, Kim MP, Hooker CM, et al. Pulmonary resection in octogenarians with stage I nonsmall cell lung cancer: a 22-year experience. Ann Thorac Surg 2004;77:271-7. [Crossref] [PubMed]
- Sigel K, Bonomi M, Packer S, et al. Effect of age on survival of clinical stage I non-small-cell lung cancer. Ann Surg Oncol 2009;16:1912-7. [Crossref] [PubMed]
- Rivera C, Dahan M, Bernard A, et al. Surgical treatment of lung cancer in the octogenarians: results of a nationwide audit. Eur J Cardiothorac Surg 2011;39:981-6. [Crossref] [PubMed]
- Thomas P, Dahan M, Riquet M, et al. Practical issues in the surgical treatment of non-small cell lung cancer. Recommendations from the French Society of Thoracic and Cardiovascular Surgery. Rev Mal Respir 2008;25:1031-6. [Crossref] [PubMed]
- Goldstraw P, Crowley J, Chansky K, et al. The IASLC Lung Cancer Staging Project: proposals for the revision of theTNM stage groupings in the forthcoming (seventh) edition of the TNM Classification of malignant tumours. J Thorac Oncol 2007;2:706-14. [Crossref] [PubMed]
- Fukuse T, Satoda N, Hijiya K, et al. Importance of a comprehensive geriatric assessment in prediction of complications following thoracic surgery in elderly patients. Chest 2005;127:886-91. [Crossref] [PubMed]
- Balducci L, Extermann M. Management of cancer in the older person: a practical approach. Oncologist 2000;5:224-37. [Crossref] [PubMed]
- Charlson ME, Pompei P, Ales KL, et al. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis 1987;40:373-83. [Crossref] [PubMed]
- Oken MM, Creech RH, Tormey DC, et al. Toxicity and response criteria of the Eastern Cooperative Oncology Group. Am J Clin Oncol 1982;5:649-55. [Crossref] [PubMed]
- Brunelli A, Kim A, Berger K, et al. Physiologic evaluation of the patient with lung cancer being considered for resectional surgery. Diagnosis and management of lung cancer 3rd ed: American College of Chest Physicians evidence-based clinical practice guidelines. Chest 2013;143:e166S-90S.
- Dindo D, Demartines N, Clavien PA. Classification of surgical complications. New proposal with evaluation in a cohort of 6336 patients and result of a survey. Ann Surg 2004;240:205-13. [Crossref] [PubMed]
- Fanelli V, Vlachou A, Ghannadian S, et al. Acute respiratory distress syndrome: new definition, current and future therapeutic option. J Thorac Dis 2013;5:326-34. [PubMed]
- Statistiques. Available online: https://www.insee.fr/fr/statistiques
- Owonikoko TK, Ragin CC, Belani CP, et al. Lung cancer in elderly patients: an analysis of the surveillance, epidemiology, and end results database. J Clin Oncol 2007;25:5570-7. [Crossref] [PubMed]
- Parkin DM, Bray F, Ferlay J, et al. Global cancer statistics, 2002. CA Cancer J Clin 2005;55:74-108. [Crossref] [PubMed]
- Ou SH, Zell JA, Ziogas A, et al. Prognostic factors for survival of stage I non- small cell lung cancer patients: a population-based analysis of 19,702 stage I patients in the California Cancer Registry from 1989 to 2003. Cancer 2007;110:1532-41. [Crossref] [PubMed]
- Chambers A, Routledge T, Pilling J, et al. In elderly patients with lung cancer is resection justified in terms of morbidity, mortality and residual quality of life? Interact CardioVasc Thorac Surg 2010;10:1015-21. [Crossref] [PubMed]
- Fanucchi O, Ambrogi M, Dini P, et al. Surgical treatment of non-small cell lung cancer in octogenarians. Interact CardioVasc Thorac Surg 2011;12:749-53. [Crossref] [PubMed]
- Burfeind WR Jr, Tong BC, O’Branski E, et al. Quality of life outcomes are equivalent after lobectomy in the elderly. J Thorac Cardiovasc Surg 2008;136:597-604. [Crossref] [PubMed]
- Pallis AG, Gridelli C, van Meerbeeck J, et al. EORTC elderly task force and lung cancer group and international society for geriatric oncology (SIOG) expert’s opinion for the treatment of non-small-cell lung cancer in an elderly population. Ann Oncol 2010;21:692-706. [Crossref] [PubMed]
- Pisani MA, Murphy TE, Araujo KL, et al. Benzodiazepine and opioid use and the duration of ICU delirium in an older population. Crit Care Med 2009;37:177-83. [Crossref] [PubMed]
- Port JL, Mirza FM, Lee PC, et al. Lobectomy in octogenarians with non-small cell lung cancer: ramifications of increasing life expectancy and the benefits of minimally invasive surgery. Ann Thorac Surg 2011;92:1951-7. [Crossref] [PubMed]




