
Surgery for limited stage small cell lung cancer
Introduction
Small cell lung cancer (SCLC) comprises 15% of all lung cancer diagnoses. It is known for aggressive tumor biology and overall poor prognosis. Despite advancements in oncologic management of non-SCLC (NSCLC), there has been minimal improvement in the treatment of SCLC among the last several decades. Overall survival rates for SCLC remain as low as 5–7% at 5 years (1).
The dismal prognosis of SCLC is directly related to both its rapid dissemination and high recurrence rate following initial treatment. Given these aggressive features, prior treatment strategies have focused on systemic therapy, with surgery often avoided due to the extent of disease at time of initial presentation. However, recent evidence suggests a role for surgery as part of a multimodal approach in a subset of patients presenting with limited stage disease. This review addresses past and present views on surgical management of SCLC, including optimal approach to surgical resection in the highly select cohort presenting with early stage disease.
Methods
We performed searches on PubMed and Embase without date limits using the following search terms: small cell lung cancer, limited stage, early stage, surgery, and lobectomy. Reference lists of selected articles were manually searched for additional relevant articles.
Staging
In the 1950s, the Veterans Administration Lung Study Group developed a staging method for SCLC that categorized the disease into two subgroups: limited stage and extensive stage (2). Limited stage disease is defined as disease restricted to the ipsilateral hemithorax that can be encompassed within a single radiation field. Overall, less than one-third of patients with SCLC are diagnosed with limited stage disease.
The tumor, node, metastases (TNM) staging system, classically used for NSCLC, has more recently been applied to SCLC. Studies examining SCLC and TNM staging have shown a correlation between TNM stage and survival, providing an additional tool for both clinical and pathologic staging (3,4). The prognostic utility offered by the TNM staging system marks a shift from the reliance on the historic two-stage system to a broader framework that incorporates tumor size and nodal status. This is of particular importance when considering candidacy for surgical resection.
Treatment: historical perspective
Chemotherapy and radiotherapy have served as the cornerstone of treatment for both limited and extensive stage SCLC for decades. This dogma is largely based upon the findings of three randomized controlled trials across a 20-year period. The first study by the Medical Research Council published in 1973 compared radiotherapy (N=73) versus surgery (N=71) for primary treatment of patients with SCLC tumors that appeared “technically resectable” (5). Comparison of study groups at 2-, 5- and 10-year follow-ups consistently found inferior survival among patients randomized to surgery (4% vs. 10%, 1% vs. 4%, and 0% vs. 4% at 2, 5 and 10 years, respectively). However, several limitations of this study question its applicability and relevance to current practice. First, only patients with proximal lesions diagnosed by rigid bronchoscopy were included. Therefore, patients with small, peripheral tumors that may have been surgically accessible and appropriate for resection were excluded. Additionally, staging of patients was likely inaccurate or incomplete without the existence of computed tomography (CT), positron emission tomography (PET), and mediastinoscopy. Finally, a microscopically margin-negative resection, or an R0 resection, was achieved in only 48% of patients undergoing surgery.
The second study by Lad et al. in 1994 randomized 146 patients with limited stage SCLC to surgery plus adjuvant thoracic and whole brain radiotherapy (N=70) versus thoracic and whole brain radiotherapy alone (N=76) (6). All patients received induction chemotherapy prior to randomization. The authors found no difference in median survival time between nonsurgical and surgical treatment groups (18.6 vs. 15.4 months, log rank P=0.78), and as a result, concluded that the addition of surgery as a component of multimodal therapy provided no benefit. However, patients without lymph node metastasis accounted for only 41% of patients randomized to surgery. In current practice, patients without lymph node involvement comprise the majority of candidates considered appropriate for oncologic surgical resection.
Finally, a study performed by Liao et al. in 1995 in China randomized 40 patients with stage III SCLC to either surgery and chemotherapy (N=20) versus radiotherapy and chemotherapy (N=20). All patients received induction chemotherapy prior to either surgery or radiotherapy. While this study demonstrated an improved survival rate in the group of patients receiving neoadjuvant chemotherapy followed by surgery, this difference in survival was not statistically significant at 2-year follow-up (7). Overall, while encouraging, the results are difficult to interpret given the small sample size as well as the relatively advanced stage of patients included in this study.
The aforementioned studies were critical in shaping treatment recommendations for SCLC. As a result, combination chemotherapy and radiotherapy became the treatment of choice for patients with SCLC. However, as discussed, overall advancements in diagnostic imaging and surgical technique call into question the applicability of these studies to modern practice.
Treatment: current perspective
Although no randomized trials exist confirming the benefit of combined surgery and chemotherapy, multiple retrospective and observational studies suggest an advantage of surgery for local control in appropriately chosen patients with early stage disease, specifically T1-2 and N0 disease (Figure 1). The University of Toronto Lung Oncology Group prospectively evaluated 38 patients undergoing chemotherapy followed by surgical resection (8). This group found that the combination of preoperative chemotherapy and surgery resulted in a 5-year survival rate of 51% for patients with stage I disease (N=7). No significant survival benefit was noted for patients with stage II (N=9) or stage III disease (N=22). An analysis of the Surveillance, Epidemiology, and End Results (SEER) registry investigated 14,179 patients presenting with localized (T1-2N0) or regional (T3-4N0) disease, of which 863 patients underwent surgery (9). Patients undergoing surgical resection had both improved 5-year survival (34.6% vs. 9.9%) and median survival (28 vs. 13 months, P<0.001) compared to patients undergoing non-surgical treatment. In a subgroup analysis, patients with localized disease were noted to have a 5-year survival rate of 44.8% compared to 26.3% in patients with regional disease. Another study of the SEER database found that patients with stage I and II disease who underwent surgical resection alone had improved median survival compared to those treated with radiotherapy alone (50 vs. 20 months, P<0.0001) (10). Importantly, authors found that the addition of radiotherapy to surgical resection did not result in a statistically significant difference in median survival time for patients undergoing lobectomy (50 months with radiotherapy vs. 37 months without radiotherapy, P=0.201) or sublobar resection (30 months with radiotherapy vs. 28 months without radiotherapy, P=0.585). These findings suggest that surgery alone may be adequate to achieve local control.
In 2015, Combs et al. performed an investigation of SCLC patients undergoing surgical resection from the National Cancer Data Base (NCDB). For patients undergoing surgery plus chemotherapy, authors demonstrated 5-year survival rates of 51% for stage I disease (N=275), 25% for stage II disease (N=75), and 18% for stage IIIA disease (N=53) (11). Another NCBD analysis demonstrated a 47% 5-year survival rate for patients with pathologic T1-2N0 tumors who underwent complete oncologic resection (12). Furthermore, this analysis found that patients who underwent surgical resection plus adjuvant chemotherapy with or without radiation had improved median overall survival (66.0 vs. 42.1 months, log-rank P<0.01) and 5-year overall survival (52.7% vs. 40.4%, log-rank P<0.01) compared to patients who underwent surgery without adjuvant therapy. Most recently, Wakeam et al. identified 2,619 patients with SCLC from the NCDB who underwent surgery. This group performed a propensity-matched analysis of survival comparing surgical versus non-surgical treatment. Surgical resection conferred improved median overall survival for stage I disease (38.6 vs. 22.9 months, P<0.0001). Although surgery was also associated with improved median overall survival for stage II and IIIA disease, survival differences decreased with increased stage (stage II 23.4 vs. 20.7 months, P=0.06; stage IIIA 21.7 vs. 16.0 months, P<0.0001) (13). In comparison, estimates based on the SEER database regarding overall 5-year survival rates for all patients presenting with SCLC managed with surgical and nonsurgical therapy between 2009 and 2015 were 27%, 16%, and 3% for localized disease, regional disease, and distant disease, respectively (14). These survival estimates are notably lower compared to surgery-specific outcomes previously discussed, supporting a role for surgical intervention in the treatment of SCLC.
Extent of surgical resection
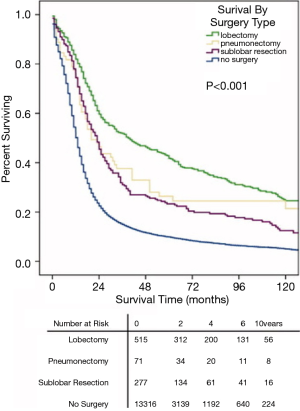
For the highly select cohort of patients eligible for surgical resection, lobectomy has been shown to be superior to other forms of lung resection. In the aforementioned NCDB analyses, authors consistently demonstrated a survival benefit for patients undergoing lobectomy compared to other procedures (11-13). Specifically, Combs et al. reported a 5-year survival rate of 40% for lobectomy compared to 21% for sublobar resection and 22% for pneumonectomy. Similarly, an analysis of the SEER registry demonstrated improved median time to death for patients with SCLC treated with lobectomy (40 months) compared to sublobar resection (23 months), pneumonectomy (20 months), and no surgery (13 months) (Figure 2) (9). In addition to lobectomy, a mediastinal lymph node dissection should be performed for surgical evaluation of lymph nodes, consistent with current surgical management of NSCLC.
Discussion
Evidence supports the consideration of initial surgery for early stage SCLC, specifically in patients with small tumors without lymph node involvement. Clinical guidelines now reflect this paradigm shift. The National Comprehensive Cancer Network (NCCN) Clinical Practice Guidelines in Oncology now recommend surgery for patients with stage I-IIA (T1-2bN0M0) disease followed by adjuvant chemotherapy (15). Similarly, guidelines by the American College of Chest Physicians (ACCP) state that in patients with stage I (T1-2aN0M0) SCLC who have undergone invasive mediastinal staging and thorough evaluation for distant metastasis, surgical resection is recommended (16). ACCP guidelines also endorse adjuvant chemotherapy following curative-intent surgical resection.
The survival advantage associated with surgical resection drastically declines in the setting of non-localized disease. Therefore, pathologic mediastinal staging is required preoperatively to rule out occult nodal disease and prevent unwarranted surgical intervention. Additionally, given the aggressive nature of SCLC, clinical staging may underestimate the extent of disease based on imaging alone. Studies suggest that pathological upstaging following surgical resection from clinical stage I disease to stage II or III disease occurs in 23–25% of patients (17,18). A study by Vallières et al. of the International Association of the Study of Lung Cancer (IASLC) database demonstrated that clinical and pathologic TNM stage were concordant in only 58% of cases (3). Furthermore, the University of Toronto Lung Oncology Group evaluated 57 patients undergoing surgical resection and found that the number of patients diagnosed with N2 disease increased from 14 patients preoperatively to 22 patients following pathologic examination (8). Therefore, preoperative invasive mediastinal lymph node sampling is imperative prior to pursuing surgery. Per the NCCN guidelines, invasive mediastinal staging can be performed with conventional mediastinoscopy or by alternative techniques such as transesophageal endoscopic ultrasound-guided fine-needle aspiration (EUS-FNA), endobronchial ultrasound-guided transbronchial needle aspiration (EBUS-TBNA), or video-assisted thoracoscopic surgery (VATS) (15). Further studies are needed to compare the relative accuracy of each of these staging methods specifically in patients with SCLC. A patient with positive lymph nodes found on preoperative mediastinal staging should no longer be considered a candidate for surgery.
As previously discussed, evidence suggesting a benefit of surgical resection for early stage SCLC is based on retrospective and observational studies. Consequently, these studies may be confounded by selection bias. That is, healthier patients with fewer comorbid conditions may be disproportionally selected over sicker patients to undergo surgical resection. This potential bias and the lack of prospective randomized trials must be considered when interpreting the results of the aforementioned studies.
Identifying patients with early stage disease that will benefit most from surgical resection remains an ongoing clinical challenge. Clinical risk factors, tumor biomarkers, and pathological characteristics all serve as potential predictors of survival outcomes and disease recurrence in patients eligible for resection. Several studies have explored factors predictive of recurrence following an R0 resection in patients with NSCLC (19-24). Kawachi et al. demonstrated that preoperative carcinoembryonic antigen (CEA) level, pathologic T stage, and lymphatic, vascular, and pleural invasion all serve as predictors of early recurrence in patients with resected stage I NSCLC (19). However, a paucity of similar data exists in patients with SCLC. In a multivariable analysis by Yang et al., increasing age [per year, hazard ratio (HR) 1.04, P<0.01], tumor size (per centimeter, HR 1.10, P<0.01), and number of comorbid conditions (Charlson/Deyo score ≥2, HR 1.54, P<0.01) were associated with decreased survival following an R0 resection for patients with pathologic T1-2N0M0 SCLC (12). Yet, with hazard ratios only slightly above 1, the associations between these variables and survival are not compelling. Further research is needed in this area to identify predictive factors of recurrence in early stage disease as a means of providing guidance to both patient and surgeon.
Conclusions
Treatment recommendations for limited stage SCLC have shifted significantly over the last decade. While historical trials supported primary chemotherapy and radiotherapy, recent studies suggest a role for surgery in early stage disease (Box 1). Current data demonstrate reasonable survival rates following surgical resection for small, localized tumors. Yet, less than 5% of patients with SCLC are diagnosed with potentially resectable stage I-IIA disease. However, with improving lung cancer awareness and increasing availability of screening modalities, it is likely that a greater number of patients with potentially resectable, early stage SCLC will be detected over the coming years. For this select group of patients, the preferred operation is lobectomy with mediastinal lymph node dissection. Further prospective randomized studies are necessary to fully evaluate the role and benefit of surgery in SCLC. Until that time, curative-intent surgical resection should be offered to patients with early stage SCLC without evidence of nodal involvement or distant metastasis, as available evidence suggests improved survival compared to prior treatment strategies utilizing chemotherapy and radiotherapy alone.

Full table
Acknowledgments
Funding: None.
Footnote
Provenance and Peer review: This article was commissioned by the Guest Editor (Jose M. Pacheco) for the series “Small Cell Lung Cancer”, published in Journal of Thoracic Disease. This article has undergone external peer review.
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/jtd.2020.03.79). The series “Small Cell Lung Cancer” was commissioned by the editorial office without any funding or sponsorship. JDM serves as the unpaid editorial board member of Journal of Thoracic Disease. Dr. JDM reports personal fees from Medtronic and Intuitive. The other authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Janssen-Heijnen ML, Karim-Kos HE, van der Drift MA, et al. Modest improvements of survival for patients with small cell lung cancer aged 45 to 59 years only, diagnosed in the Netherlands, 1989 to 2008. J Thorac Oncol 2012;7:227-32. [Crossref] [PubMed]
- Micke P, Faldum A, Metz T, et al. Staging small cell lung cancer: Veterans Administration Lung Study Group versus International Association for the Study of Lung Cancer—what limits limited disease? Lung Cancer 2002;37:271-6. [Crossref] [PubMed]
- Vallières E, Shepherd FA, Crowley J, et al. The IASLC Lung Cancer Staging Project: proposals regarding the relevance of TNM in the pathologic staging of small cell lung cancer in the forthcoming (seventh) edition of the TNM classification for lung cancer. J Thorac Oncol 2009;4:1049-59. [Crossref] [PubMed]
- Shepherd FA, Crowley J, Van Houtte P, et al. The International Association for the Study of Lung Cancer lung cancer staging project: proposals regarding the clinical staging of small cell lung cancer in the forthcoming (seventh) edition of the tumor, node, metastasis classification for lung cancer. J Thorac Oncol 2007;2:1067-77. [Crossref] [PubMed]
- Fox W, Scadding JG. Medical Research Council comparative trial of surgery and radiotherapy for primary treatment of small-celled or oat-celled carcinoma of bronchus. Ten-year follow-up. Lancet 1973;2:63-5. [Crossref] [PubMed]
- Lad T, Piantadosi S, Thomas P, et al. A prospective randomized trial to determine the benefit of surgical resection of residual disease following response of small cell lung cancer to combination chemotherapy. Chest 1994;106:320s-3s. [Crossref] [PubMed]
- Liao M, Zhao J, Zhou Y. Multimodality therapy of late stage lung cancer. Zhonghua Zhong Liu Za Zhi 1995;17:384-6. [PubMed]
- Shepherd FA, Ginsberg RJ, Patterson GA, et al. A prospective study of adjuvant surgical resection after chemotherapy for limited small cell lung cancer. A University of Toronto Lung Oncology Group study. J Thorac Cardiovasc Surg 1989;97:177-86. [Crossref] [PubMed]
- Schreiber D, Rineer J, Weedon J, et al. Survival outcomes with the use of surgery in limited-stage small cell lung cancer: should its role be re-evaluated? Cancer 2010;116:1350-7. [Crossref] [PubMed]
- Varlotto JM, Recht A, Flickinger JC, et al. Lobectomy leads to optimal survival in early-stage small cell lung cancer: a retrospective analysis. J Thorac Cardiovasc Surg 2011;142:538-46. [Crossref] [PubMed]
- Combs SE, Hancock JG, Boffa DJ, et al. Bolstering the case for lobectomy in stages I, II, and IIIA small-cell lung cancer using the National Cancer Data Base. J Thorac Oncol 2015;10:316-23. [Crossref] [PubMed]
- Yang CFJ, Chan DY, Speicher PJ, et al. Role of Adjuvant Therapy in a Population-Based Cohort of Patients With Early-Stage Small-Cell Lung Cancer. J Clin Oncol 2016;34:1057-64. [Crossref] [PubMed]
- Wakeam E, Acuna SA, Leighl NB, et al. Surgery Versus Chemotherapy and Radiotherapy For Early and Locally Advanced Small Cell Lung Cancer: A Propensity-Matched Analysis of Survival. Lung Cancer 2017;109:78-88. [Crossref] [PubMed]
- Howlader N, Noone AM, Krapcho M, et al. SEER Cancer Statistics Review, 1975-2016, National Cancer Institute. Available online: [Accessed 15 Feb 2020].https://seer.cancer.gov/csr/1975_2016/
- Small Cell Lung Cancer (Version 3.2020). NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines). Available online: [Accessed 15 Feb 2020].https://www.nccn.org/professionals/physician_gls/pdf/sclc.pdf
- Jett JR, Schild SE, Kesler KA, et al. Treatment of Small Cell Lung Cancer: Diagnosis and Management of Lung Cancer, 3rd ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest 2013;143:e400S-19S.
- Takei H, Kondo H, Miyaoka E, et al. Surgery for small cell lung cancer: a retrospective analysis of 243 patients from Japanese Lung Cancer Registry in 2004. J Thorac Oncol 2014;9:1140-5. [Crossref] [PubMed]
- Thomas DC, Arnold BN, Rosen JE, et al. Defining outcomes of patients with clinical stage I small cell lung cancer upstaged at surgery. Lung Cancer 2017;103:75-81. [Crossref] [PubMed]
- Kawachi R, Tsukada H, Nakazato Y, et al. Early recurrence after surgical resection in patients with pathological stage I non-small cell lung cancer. Thorac Cardiovasc Surg 2009;57:472-5. [Crossref] [PubMed]
- Maeda R, Yoshida J, Hishida T, et al. Late Recurrence of Non-Small Cell Lung Cancer More Than 5 Years After Complete Resection: Incidence and Clinical Implications in Patient Follow-up. Chest 2010;138:145-50. [Crossref] [PubMed]
- Maeda R, Yoshida J, Ishii G, et al. Risk Factors for Tumor Recurrence in Patients With Early-Stage (Stage I and II) Non-small Cell Lung Cancer: Patient Selection Criteria for Adjuvant Chemotherapy According to the Seventh Edition TNM Classification. Chest 2011;140:1494-502.
- Pasini F, Pelosi G, Valduga F, et al. Late events and clinical prognostic factors in stage I non small cell lung cancer. Lung Cancer 2002;37:171-7. [Crossref] [PubMed]
- Shiono S, Abiko M, Sato T. Positron Emission Tomography/Computed Tomography and Lymphovascular Invasion Predict Recurrence in Stage I Lung Cancers. J Thorac Oncol 2011;6:43-7. [Crossref] [PubMed]
- Shoji F, Haro A, Yoshida T, et al. Prognostic Significance of Intratumoral Blood Vessel Invasion in Pathologic Stage IA Non-Small Cell Lung Cancer. Ann Thorac Surg 2010;89:864-9. [Crossref] [PubMed]


