Assessment of the usefulness of video-assisted thoracoscopic surgery in patients with non-tuberculous thoracic empyema
Introduction
The pneumonia is one of the common medical illnesses, and the economic burden of pneumonia is high in Korea (1). Parapneumonic effusion or empyema is associated with pneumonia. It typically occurs with 20-40% of pneumonia (2). Empyema, the purulent pleural manifestation of complicated pneumonia, is a difficult clinical problem with a mortality rate that is close to 20% (2). Classic management of empyema consists of early pleural drainage via chest tube and the use of antibiotics (3). Tube thoracostomy is the general treatment approach for patients with early-stage empyema, whereas patients with advanced empyema often experience a high rate of treatment failure because of the failure to drain loculated areas (2). Ideally, patients with advanced empyema should then undergo pleural decortication (4). More recently, however, the technique of minimally invasive video-assisted thoracoscopic surgery (VATS) has become the gold standard for thoracic empyema because of the low operative morbidity and fewer complications associated with the procedure (5). The purpose of this retrospective study was to investigate the effectiveness and the outcomes of VATS treatment compared with medical therapy (chest tube drainage, antibiotic treatment) in non-tuberculous thoracic empyema patients. Additionally, the study investigated the possibility of early operation group outcomes being superior to late operation group outcomes in surgical treatment of empyema.
Materials and methods
Patients were identified during the study period that extended from January 2006 through June 2012 at the Hallym University Kangnam Sacred Hospital, in Seoul, in the Republic of Korea. Thirty-two patients who met the criteria of empyema were selected for and included in this study. Patients that were <18 years of age, experienced malignant pleural effusions or tuberculosis pleural effusions, or had chronic empyema were excluded.
Thoracentesis was first performed in order to diagnose empyema. The following criteria were used to establish a diagnosis of empyema: septations or loculations identified in the pleural space by computed tomography (CT), existence of gross pus or organisms demonstrated by Gram stain or culture, or positive biochemical methods. The stage of empyema was determined using the classification of parapneumonic effusion described by the American College of Chest Physicians (ACCP) (6). Surgical decortication using the VATS procedure was attempted as soon as possible in cases of diagnosed empyema. Chest tube drainage was performed in cases where patients refused surgery, or in the event that the team was unable to operate. VATS decortication was performed under general anesthesia, using a double-lumen endotracheal tube for selective ventilation. After debridement was completed, one or two chest tubes (28F) were inserted into the lower part of the cavity through separate incisions using two ports. Follow-up consisted of a chest X-ray, a chest CT, or both, in addition to a clinical investigation. Patient symptoms including fever or chest pain or laboratory data such as a leucocyte count, a C-related protein level, preoperative bacteriology results, the duration of chest tube drainage, the duration of hospital stay, the duration of antibiotic use, and postoperative complications were recorded, in addition to radiographic findings. The operation group was defined as patients who were treated with VATS decortication as the first line of treatment for pleural empyema. The drainage group was defined as patients who were treated with simple drainage (insertion of a 28F large-bore chest tube). Antibiotic therapy duration was defined as the interval between the dates of administration of intravascular antibiotics. The drainage period was defined as the interval between the first chest tube drainage and the day drainage tubes were removed. The hospital stay was defined as the interval between the date of admission and the date of hospital discharge. Variation in patient outcomes, the duration of hospital stay, the length of the tube insertion period and the duration of antibiotic therapy between the operation group and the drainage group were analyzed. The operation group was divided into two groups for analysis: the early operation group, which was diagnosed with empyema and received an operation before 2 days had elapsed, and the late operation group, which was operated on 2+ days after diagnosis. The study also worked to verify the operation duration and identify the possibility of a transfusion.
Continuous statistical analysis variables were assessed with the 2-sided Student’s t-test. A P value of <0.05 was considered statistically significant. The difference between the early operation group and the late operation group was assessed using a non-parametric test. The data in this retrospective study are expressed as mean ± standard deviation. The study protocol was reviewed and approved by the institutional ethics committee (Ethical Committee of Kangnam sacred heart hospital, approval number 2013-03-25). Written informed consent was obtained from all participants.
Results
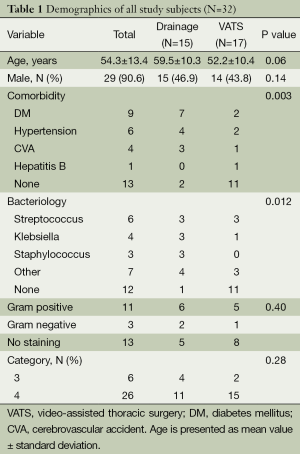
The research subjects consisted of 29 males and 3 females with a mean age of 54 years (range 19 to 72 years). A total of 32 patients underwent surgical decortication or tube drainage for pleural empyema between January 2006 and July 2011. Patient characteristics are shown in Table 1. Comorbidity was found in 20 of the 32 patients (62.5%) (Table 1) and fever, chest pain, and dyspnea were the most frequently encountered symptoms. Preoperative pleural fluid culture was positive in 20 of the 32 patients (62.5%) with Streptococcus findings in six cases, Klebsiella in four cases, Staphylococcus in three cases, and other bacteria found in seven cases. Bacteria were isolated on PF culture in 20 of 32 patients (62.5%), and 14 out of 32 (43.8%) of the PF cultures that were Gram-stained revealed bacterial isolation. The majority of patients had clinically advanced empyema and, out of 32 enrolled patients, 6 patients (18.8%) were staged as category 3 and 26 patients (81.2%) were staged as category 4. There was no significant difference in the initial disease severity index between the operation groups and the drainage group. The duration of hospital stay for the patients was 19.1±8.3 days, the length of antibiotic therapy was 16.6±7.7 days, and the number of days required for drainage was 11.1±9.1 days. Postoperative complications were not observed in all cases. There were five hospital deaths in the drainage group; however, there was no incidence of hospital death for patients in the operation group. Of 32 patients, 27 (67.6%) showed treatment success after the first intervention. The surgical group showed a 0% mortality rate (0/17 patients, Figure 1), whereas the drainage group showed a 33.3% success rate (5/15 patients) (P<0.001, Table 2). No significant difference was noted between groups with respect to age, APACHE II, or SAPS II scores. The duration of antibiotic therapy was 18.4±8.6 days for the drainage group and 15.2±6.7 days for the operation group, and no significant difference in the average durations of antibiotic treatments was observed between the two groups (P=0.25). In addition, the number of days required for drainage was 15.1±11.3 days for the drainage group and 8.3±4.6 days for the operation group, and the duration of hospital stay was 22.4±10.0 days for the drainage group and 16.3±5.0 days for the operation group, with observed significant differences of P=0.03 and 0.03, respectively.
Full table

Full table
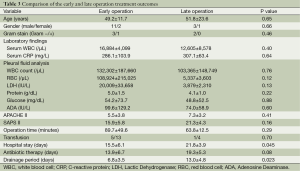
Out of the patients that received VATS therapy, 13 patients underwent VATS decortication directly, and four patients underwent VATS decortication later due to patient hesitancy to undergo the operation (Table 3). The mean operation duration was 89.7±49.6 minutes for the early operation group and 63.8±12.5 minutes for the late operation group. Of all 17 patients who underwent an operation, six received a blood transfusion. The results indicated that for the VATS group, the number of days required for drainage and the duration of hospital stay were significantly shorter for the early operation groups.
Full table
Discussion
Previously, thoracic tube insertion and post-operative follow-up were the standard treatment for empyema patients. However, the success rate was low for patients that were treated with this method, and there was a high rate of open decortication for patients with loculated empyema (7). After an initial diagnosis of empyema, rapid surgical treatment is needed to achieve a good prognosis (8). Even if there is prompt treatment with antibiotics and chest tube drainage, surgical intervention is often required if the lung does not expand (9). Since the 1990s, VATS has become widely used for several types of thoracic surgery (10). Although many retrospective findings have suggested that VATS is useful for fibropurulent (stage II) thoracic empyema, an evidence-based guideline assumes its clinical benefit at a level C (historically controlled series and case series) (6) because there are few prospective randomized control studies. In this study, however, VATS provided both an excellent surgical view and operationality for a complicated empyema cavity, thus making it possible to perform a sufficient evacuation of all empyema membranes and fluids and sufficient removal of fibrous peel in the same manner as an open surgery. The duration of chest drainage was 8.3±4.6 days in the surgical cases, and there was no irreversible morbidity. However, the duration of chest drainage was 15.1±11.3 days in the drainage cases, and there were five cases of mortality, three sepsis and two pneumonia. In this study, 17 of 17 cases (100%) were successfully cured with VATS, and in the instance of an open thoracotomy, surgery was not performed because of the possibility of a failure of reexpansion. This study did not have any incidences of surgery-related mortality and there were no complications except that 6 out of 17 patients needed blood transfusions as a result of the operation. The operation group in this study experienced better prognoses including a shorter length of hospitalization and a shorter chest tube insertion period.
In addition to VATS, intrapleural instillation of fibrinolytic drugs has been observed to have positive outcomes for lysing fibrin adhesions. Numerous case studies and controlled trials have reported the beneficial effects of intrapleural fibrinolysis for avoiding surgery, promoting catheter drainage, and improving the radiographic appearance of loculated effusions (11-13). When this procedure was studied, there was no significant difference in mortality, the need for surgery, the radiographic outcome or the length of hospitalization (14,15). For these reasons, intrapleural fibrinolysis was not performed in this study. Surgery is often selected as a treatment option when other medical treatments fail because of its invasiveness and the severity of potential complications; thus, surgery should only be recommended in patients with a residual pleural collection and persistent sepsis despite antibiotic therapy and drainage (16).
Two randomized clinical trials in adults that compared first line VATS with medical treatment (chest tube drainage with or without fibrinolytics and antibiotics) did not show a survival advantage from early surgical intervention (17,18). According to a meta-analysis which followed ACCP guidelines, surgical treatment for empyema, regardless of whether it was via VATS access or open access, showed a superior success rate when compared with therapeutic thoracocentesis, tube thoracostomy, and fibrinolysis. In addition, surgical treatment was found to be superior to simple drainage modalities in reducing total mortality (6). In this study, four cases from the drainage group required surgical intervention when lung expansion by the chest tube insertion procedure was unsuccessful. Prior to operation, clinical manifestations, such as the duration of the preoperative period or a chest CT evaluation, failed to determine these four cases as potentially stage III thoracic empyema. The data also showed that groups that received early surgical treatment had better prognoses and a shorter length of hospitalization. These results indicate that with tube insertions it is better to have an early operation than a delayed operation.
There were several limitations to this retrospective study. The cohort may be biased by patient selection and the small number of patients that were examined.
In summary, in patients with early stage multiloculated non-tuberculous thoracic empyema, VATS decortication is superior to tube thoracostomy alone, and has been shown to reduce the time required for drainage and the duration of hospital stay. VATS also has a relatively high success rate without significant morbidity. Therefore, VATS decortication might be recommended as a first-line therapy in non-tuberculous thoracic empyema.
Acknowledgements
Disclosure: The authors declare no conflicts of interest.
References
- Yoo KH, Yoo CG, Kim SK, et al. Economic burden and epidemiology of pneumonia in Korean adults aged over 50 years. J Korean Med Sci 2013;28:888-95. [PubMed]
- Light RW. Parapneumonic effusions and empyema. Proc Am Thorac Soc 2006;3:75-80. [PubMed]
- Davies HE, Rosenstengel A, Lee YC. The diminishing role of surgery in pleural disease. Curr Opin Pulm Med 2011;17:247-54. [PubMed]
- Tong BC, Hanna J, Toloza EM, et al. Outcomes of video-assisted thoracoscopic decortication. Ann Thorac Surg 2010;89:220-5. [PubMed]
- Shin JA, Chang YS, Kim TH, et al. Surgical decortication as the first-line treatment for pleural empyema. J Thorac Cardiovasc Surg 2013;145:933-9. [PubMed]
- Colice GL, Curtis A, Deslauriers J, et al. Medical and surgical treatment of parapneumonic effusions: an evidence-based guideline. Chest 2000;118:1158-71. [PubMed]
- Yamaguchi M, Takeo S, Suemitsu R, et al. Video-assisted thoracic surgery for fibropurulent thoracic empyema: a bridge to open thoracic surgery. Ann Thorac Cardiovasc Surg 2009;15:368-72. [PubMed]
- Lackner RP, Hughes R, Anderson LA, et al. Video-assisted evacuation of empyema is the preferred procedure for management of pleural space infections. Am J Surg 2000;179:27-30. [PubMed]
- Lewis RJ, Caccavale RJ, Sisler GE. Imaged thoracoscopic surgery: a new thoracic technique for resection of mediastinal cysts. Ann Thorac Surg 1992;53:318-20. [PubMed]
- Anstadt MP, Guill CK, Ferguson ER, et al. Surgical versus nonsurgical treatment of empyema thoracis: an outcomes analysis. Am J Med Sci 2003;326:9-14. [PubMed]
- Davies RJ, Traill ZC, Gleeson FV. Randomised controlled trial of intrapleural streptokinase in community acquired pleural infection. Thorax 1997;52:416-21. [PubMed]
- Misthos P, Sepsas E, Konstantinou M, et al. Early use of intrapleural fibrinolytics in the management of postpneumonic empyema. A prospective study. Eur J Cardiothorac Surg 2005;28:599-603. [PubMed]
- Skeete DA, Rutherford EJ, Schlidt SA, et al. Intrapleural tissue plasminogen activator for complicated pleural effusions. J Trauma 2004;57:1178-83. [PubMed]
- Maskell NA, Davies CW, Nunn AJ, et al. U.K. Controlled trial of intrapleural streptokinase for pleural infection. N Engl J Med 2005;352:865-74. [PubMed]
- Tokuda Y, Matsushima D, Stein GH, et al. Intrapleural fibrinolytic agents for empyema and complicated parapneumonic effusions: a meta-analysis. Chest 2006;129:783-90. [PubMed]
- Brixey AG, Luo Y, Skouras V, et al. The efficacy of chest radiographs in detecting parapneumonic effusions. Respirology 2011;16:1000-4. [PubMed]
- Bilgin M, Akcali Y, Oguzkaya F. Benefits of early aggressive management of empyema thoracis. ANZ J Surg 2006;76:120-2. [PubMed]
- Wait MA, Sharma S, Hohn J, et al. A randomized trial of empyema therapy. Chest 1997;111:1548-51. [PubMed]


